Complement C5a Receptor 2 (C5aR2)
In the process of complement activation, complement C5 is cleaved by C5 convertases into C5b to form the membrane attack complex (MAC), releasing an anaphylatoxin product C5a. C5a is a potent chemotactic agent and an anaphylatoxin mediating multiple inflammatory reactions by binding to C5a receptors. There are two C5a receptors, C5aR1 (CD88) and C5aR2.
C5a anaphylatoxin chemotactic receptor 2 (C5aR2), also known as C5L2 and GPR77, is a G-protein coupled receptor (GPCR) composed of a single polypeptide chain. The total 337 amino acid residues fold into seven-transmembrane α-helices and an extracellular N-terminus. Similar to C5aR1, at the N-terminus Asn3 of C5aR2 lies a single N-linked glycosylation site, which is of great significance for the ligand binding and functioning. Different from C5aR1, the intracellular C-terminus of C5aR2 contains several serine residues and phosphorylation sites for signaling and desensitization. And C5aR2 has a different DRY motif and lacks NPXXY intracellular motifs, both of which are necessary for G-protein coupling and signal transduction for GPCRs.
C5aR2 is an enigmatic receptor functioning by binding to its ligands complement C5a and C5a des-arginine, although the exact action in immune response has been controversial.
-
Serve as a modulator of C5aR1 signaling: C5aR2 may compete with C5aR1 for binding to C5a and C5a-desArg, thereby reducing the concentrations of C5a and C5a-desArg and C5aR1 signaling.
-
Modulatory activities on other receptors: C5aR2 also has been demonstrated to mediate inflammatory reactions through affecting some innate immune receptors, such as Toll-like receptors, and NLRP3 inflammasome system.
-
Functions in adaptive immunity: C5aR2 has a potential role in inducing the generation and development of Treg cells while inhibiting the development and differentiation of Th17 and Th1 cells.
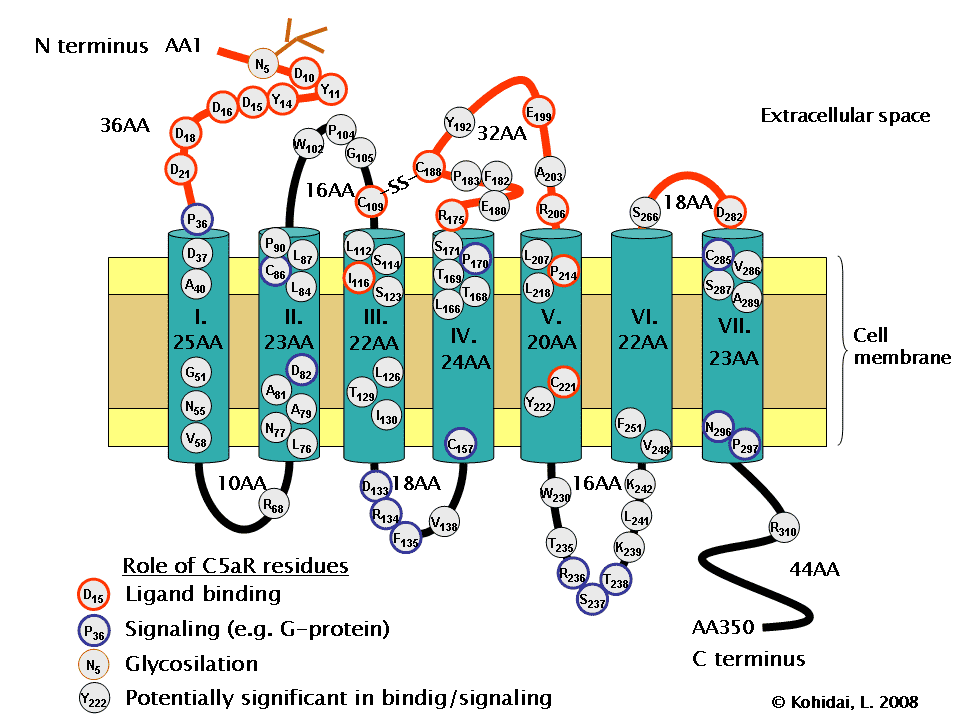 Fig.1 Protein structure of the complement C5aR2. (Li, 2019)
Fig.1 Protein structure of the complement C5aR2. (Li, 2019)
Reference
-
Li, X.X.; et al. The Complement Receptor C5aR2: A Powerful Modulator of Innate and Adaptive Immunity. The Journal of Immunology. 2019, 202: 3339-3348.
For Research Use Only.

 Fig.1 Protein structure of the complement C5aR2. (Li, 2019)
Fig.1 Protein structure of the complement C5aR2. (Li, 2019)