Oncolytic Viruses in Breast Cancer Treatment
Introduction of Breast Cancer
Breast cancer is a common malignant tumor in women. Breast cancer mainly starts in three parts of the breast, including lobules, ducts, and connective tissue. In general, the lobules and ducts play an important role in mediating the production and transportation of milk in humans. In the past few years, different kinds of breast cancer have been defined based on cells that can be converted to cancer, such as invasive ductal carcinoma and invasive lobular carcinoma. Meanwhile, pilot studies have demonstrated that breast cancer can also spread to many tissues by blood vessels and lymphatics. The main symptoms of breast cancer consist of a lump near your breast, nipple discharge, as well as the size and shape change of breast or nipple.
Standard Therapy for Breast Cancer
Nowadays, some options for the treatment of breast cancer have been developed to remove the tumor and prevent tumor recurrence. Among them, surgery and chemotherapy are widely used for cutting out or killing cancer cells. Hormonal therapy and radiation therapy have been regarded as replacement therapy for blocking the growth of cancer cells in the human body. Furthermore, recent studies have revealed that novel immunotherapy is a powerful tool for triggering enough immune responses against cancer cells without causing any side effects in clinical use.
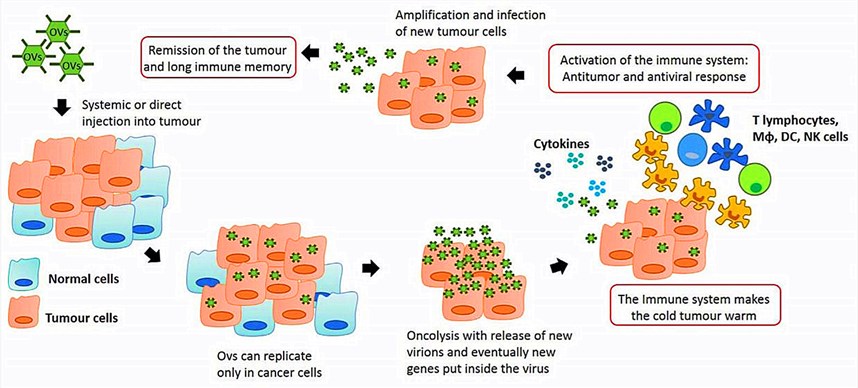 Fig.1 Anti-tumor immunity by oncolytic virus therapy. (Marelli, 2018)
Fig.1 Anti-tumor immunity by oncolytic virus therapy. (Marelli, 2018)
Oncolytic Viral Therapy for Breast Cancer
Oncolytic viruses are a group of natural and engineered viruses that have been used for inhibiting the replication of tumor cells and killing cancer cells, especially for breast cancer cells. Previous researches have illustrated that oncolytic viral therapy is capable of activating the immune system suppressed by the tumor microenvironment without harming normal cells and tissues. Macrophages and T lymphocytes are essential to produce a variety of cytokines responsible for recruiting immune cells to kill cancer cells. In this process, oncolytic viruses enable to create a long-term immunological memory that avoids tumor progression.
Currently, many kinds of viruses, including but not limited to, reovirus, oncolytic adenovirus (oAd), poliovirus, oncolytic measles virus (oMV), herpes simplex virus type 1 (HSV-1), Newcastle disease virus (NDV), and coxsackievirus, have been exclusively studied to evaluate the potential oncolytic efficacy in the clinic. For example, a highly attenuated HSV-1 clone has been used to infect human and murine breast cancer cells, and assess its safety profile in virotherapy. The data have shown that virus can prevent tumor growth and improve the long-term survival rates, suggesting its significant role as cancer therapeutic agents.
Clinical Trials for Breast Cancer Treatment
The feasibility of oncolytic viruses has been broadly utilized in numerous tumor models. Many results derived from phase I and phase II clinical trials have also proven its therapeutic effectiveness for different tumor types, including breast cancer. Several small early-phase clinical trials have been conducted on patients with breast cancers to test the safety and toxicity of oncolytic viral therapy. Furthermore, a series of genetic engineering technology has been generated to design novel oncolytic viral vectors for improving their overall performance in the clinic. For example, phase II clinical trials of a modified HSV-1 have been conducted against breast cancers. Patients have been recruited to receive intratumoral injections with doses of virus every two weeks. The objective response rate (ORR), partial response (PR) rate, adverse events (AEs) and the overall survival rate will be fully assessed. The data have indicated that the ORR and CR rates are 36% and 18%, respectively. Also, a phase I trial investigated reovirus in combination with another strain of oncolytic reovirus has been completed in patients with metastatic breast cancer. This trial has proven that the combination therapy of oncolytic viruses and the therapeutic drug is more effective than conventional treatment alone.
Reference
- Marelli, G.; et al. Oncolytic viral therapy and the immune system: a double-edged sword against cancer. Frontiers in immunology. 2018, 9: 866. Distributed under Open Access license CC BY 4.0, without modification.
