Oncolytic Viruses in Ovarian Cancer Treatment
Introduction to Ovarian Cancer
Ovarian cancer is a malignant disease that begins in the ovaries. In general, the main symptoms of ovarian cancers include appetite loss, bloating, vaginal bleeding, breathlessness, and pain in the pelvis and back. The female reproductive system consists of vagina, womb, fallopian tubes, as well as two ovaries. The ovary plays an important role in producing eggs, estrogen, as well as progesterone. In the past few decades, four types of ovarian cancer, including epithelial ovarian cancer, germ cell ovarian tumors, sex cord-stromal tumors, and borderline ovarian tumors, have been confirmed and evaluated by both in vitro and in vivo studies. Among them, epithelial ovarian cancer is the most common type and accounts for 90% of all cases. Furthermore, many experts have reported that a series of factors have been identified that are associated with a high risk of ovarian cancers, such as age, family and reproductive history, obesity, and overweight. Besides, recent studies have revealed that breast cancer gene (BRCA) should be a significant biomarker for various cancers, especially for breast cancer and ovarian cancer. In this case, patients with breast cancer may also have a great risk of ovarian cancer.
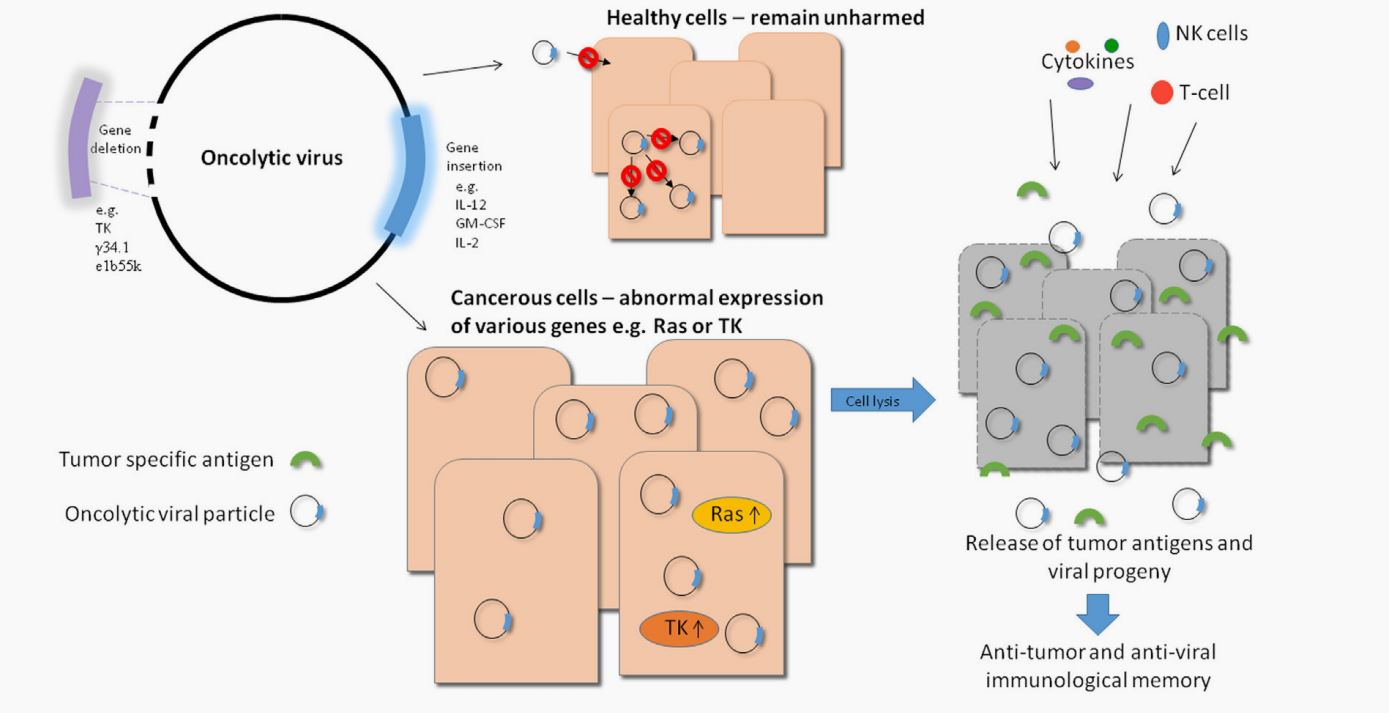
The Oncolytic Viruses in Ovarian Cancer Treatment
Ovarian cancer has become the fifth common cancer in women between the ages of 35 and 70. A wide variety of methods have been developed for treating ovarian cancer, such as surgical resection, chemotherapy, and radiotherapy. Meanwhile, pilot studies have shown that immunotherapy should be a promising strategy for ovarian cancer treatment. For instance, adoptive cell therapy has been developed by using engineered T cells to express ovarian tumor-specific T cell receptors or a chimeric antigen receptor (CAR) in patients.
In recent studies, oncolytic viruses have been used as an alternative therapy for ovarian cancer and many attempts have been made to develop their therapeutic potential. As a result, novel oncolytic virotherapy has also been designed for specifically and efficiently infect and replicate in ovarian cancer cells without causing any side effects. Additionally, combining oncolytic virotherapy with CAR-T therapies has been considered as an attractive way to improve adenovirus transduction in ovarian cancer cells. Also, various kinds of oncolytic viruses have been used as candidate therapeutic agents and several clinical trials have been conducted to test the safety and efficacy profiles. Among them, adenovirus and measles virus are broadly used as potential tools for delivering specific genes or candidate drugs to target ovarian cancer cells in phase I clinical trials. However, no oncolytic viruses have been approved by the Food and Drug Administration (FDA) for treating ovarian cancer. Therefore, many efforts still need to be made to improve the safety and efficacy of oncolytic virus-based therapy in clinical use.
Reference
- Howells, A.; et al. Oncolytic Viruses-Interaction of Virus and Tumor Cells in the Battle to Eliminate Cancer. Front Oncol. 2017, 7: 195.
