Targeted delivery employs bioengineered transport mechanisms to concentrate pharmacological agents at pathological sites, enhancing therapeutic bioavailability while minimizing systemic dispersion. This advanced strategy utilizes molecularly designed carriers to address the non-specific distribution and suboptimal therapeutic indices inherent in traditional drug administration protocols.
 Conventional Drug Delivery
Conventional Drug Delivery
 Targeted Drug Delivery
Targeted Drug Delivery
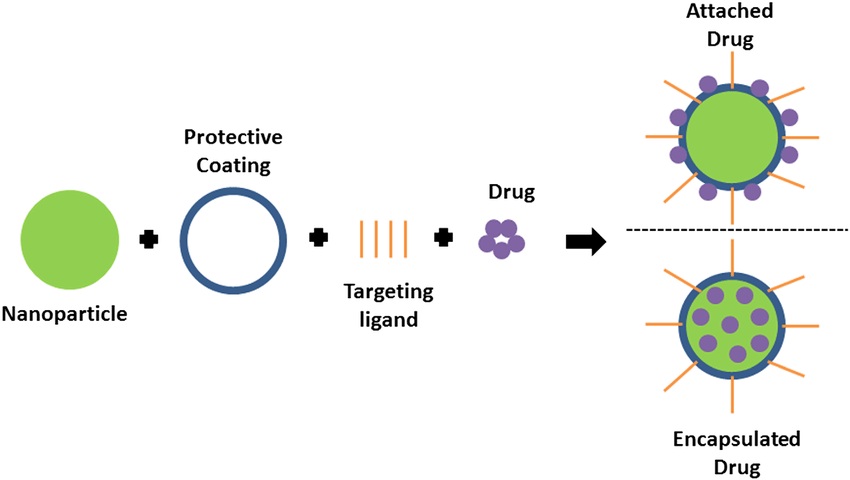 Fig.1 Scheme of drug loading options in targeted drug delivery.1
Fig.1 Scheme of drug loading options in targeted drug delivery.1
Creative Biolabs stands at the forefront of pharmaceutical innovation, offering unparalleled expertise in targeted drug delivery through a sophisticated combination of advanced Module Delivery Systems and highly specific Targeting Modules. Our comprehensive suite of delivery vehicles, including cutting-edge liposomes, Lipid Nanoparticles (LNPs), Lipid-Polymer Hybrid Nanoparticles, Exosomes, Virus-Like Particles (VLPs), Fc Fusion Proteins, and Albumin, ensures optimal payload encapsulation and transport. Complementing these systems, our meticulously engineered Targeting Modules—ranging from diverse peptides (including cell-penetrating peptides and homing peptides), antibodies, and aptamers, to Functionalized Lipids, Targeted Polymers, and Responsive Materials—enable precise recognition and accumulation at disease-specific sites, organs, or even subcellular organelles. This integrated approach allows us to overcome traditional delivery barriers, enhancing therapeutic efficacy while minimizing off-target effects, and ultimately accelerating the development of next-generation therapies for our clients.
Creative Biolabs is equipped with a complete module delivery system and an experienced team of scientists dedicated to advancing your research. We provide a wealth of corresponding products and services for you to choose from, tailored to your specific project needs.
Creative Biolabs is committed to providing a wide range of customized delivery systems development services and offering solutions tailored to our customers' unique specifications.
 Distributed under Freepik License, from Freepik.
Distributed under Freepik License, from Freepik.
Current drug delivery systems occasionally exhibit limited therapeutic efficacy despite tumor accumulation, as single-agent platforms struggle to concurrently address membrane-level and organelle-level biological barriers. To resolve this, next-generation bioresponsive vectors are being engineered for precise subcellular navigation. Creative Biolabs' solutions enable compartment-specific localization—including nuclear localization signals, cytosolic release mechanisms, and mitochondrial translocation pathways—or synergistic dual-addressing systems targeting both tumor surface markers and intracellular organelle vulnerabilities.
 Distributed under Freepik License, from Freepik.
Distributed under Freepik License, from Freepik.
Diverse pathogenic species present a significant global health challenge, necessitating persistent scientific innovation in microbial-targeting delivery systems. Recent innovations in targeted therapy facilitate enhanced treatment accuracy via microbe-directed delivery systems, ensuring superior pharmacological effectiveness with minimized collateral impact. These methodologies address urgent medical imperatives: heightened molecular targeting to reduce unintended harm, increased patient compliance through simplified dosing regimens, and optimized therapeutic frameworks enabling expanded clinical adoption.
 Distributed under Freepik License, from Freepik.
Distributed under Freepik License, from Freepik.
The development of precision vectors originated from the need for enhanced diagnostic accuracy and safer therapeutic interventions in human pathologies. Vector engineering has progressively advanced alongside deepening insights into disease-associated molecular pathways. Through specific engagement of pathognomonic biomarkers, these ligand-payload conjugates optimize subcellular localization of pharmacological or imaging payloads, significantly reducing off-target sequelae. Numerous bioengineered targeting platforms have now successfully completed investigational stage testing in disease models.
 Distributed under Freepik License, from Freepik.
Distributed under Freepik License, from Freepik.
Pathological and physiological states frequently exhibit distinct cellular phenotypes across tissues. Ligand systems engineered for organ-specific cellular signatures (e.g., vascular endothelia, mononuclear phagocytes) can be functionalized with diagnostic probes to track biomarker dynamics during therapeutic interventions. When coupled with therapeutic payloads through precision conjugation of cell-selective ligands, these platforms enable spatiotemporal precision in pharmacologic agent deployment, optimizing biodistribution while minimizing non-specific exposure.
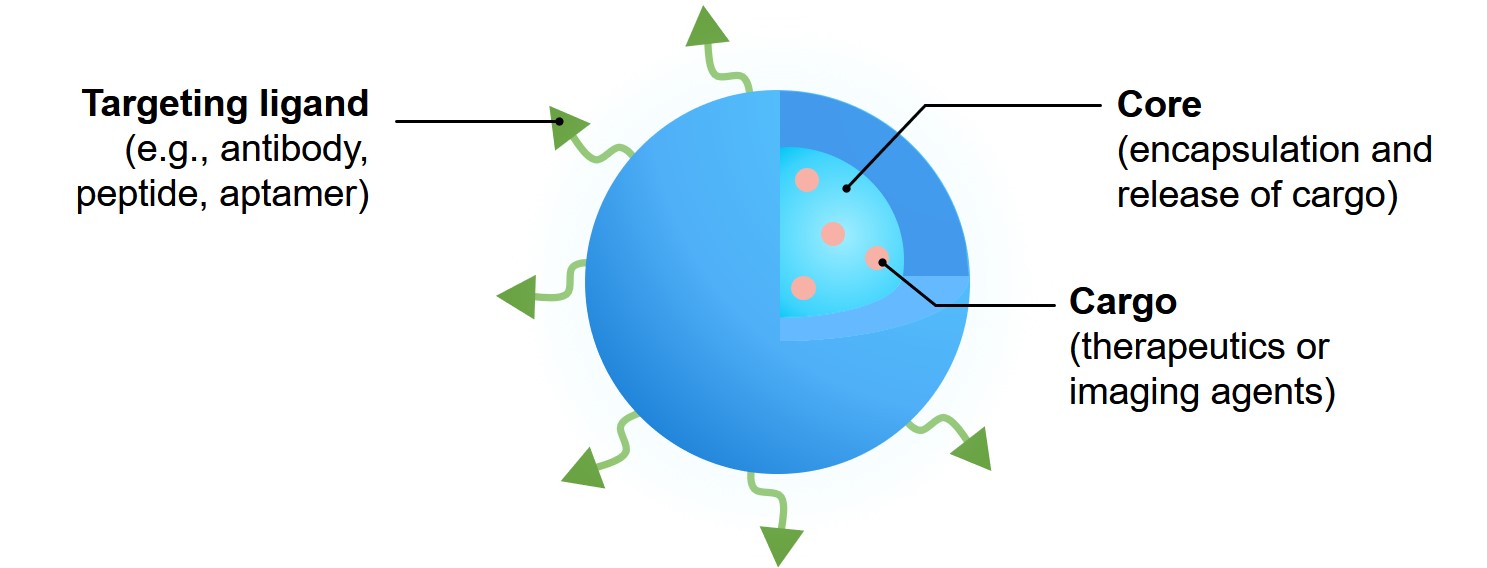 Fig 6. Nanoparticle platform for drug delivery.
Fig 6. Nanoparticle platform for drug delivery.
Targeting modules enable precise addressing of pathological entities across tissue, cellular, and microbial scales. These bioaffinity agents are integral to precision delivery platforms and molecular imaging diagnostics. With over a decade of expertise in protein engineering and peptide optimization, Creative Biolabs develops tailored conjugation architectures for diverse biomedical applications. Our cutting-edge bioengineering infrastructure supports comprehensive molecular homing solutions for oncological therapeutics and diagnostic innovations. Partnering with researchers, we provide end-to-end strategic guidance throughout the innovation pipeline—translating conceptual frameworks into validated clinical-stage biotherapeutics.
 Targeting Module
Targeting Module
Targeted drug delivery enhances therapeutic agent accumulation at disease sites through engineered transport mechanisms. Our targeting module can bind to the target through receptor-ligand binding, thereby delivering drugs to specific locations. Here is the comment targeting module we provide.
| Peptides | Peptides represent biopolymeric structures formed through covalent amide linkages between amino acid residues. As targeting modules, these biomolecules, including cell-penetrating peptides (CPPs), homing peptides (HPs), etc., have emerged as promising pharmacotherapeutics due to their enhanced conformational versatility relative to antibody-based treatments. |
| Antibodies | Antibodies achieve antigen-specific binding through variable fragment regions, either neutralizing pathogenic cascades or orchestrating macrophage-mediated opsonization of pathological entities. Antibodies now constitute frontline biopharmaceutical interventions in clinical oncology and immune disorder management. |
| Aptamers | Aptamers are used as targeting modules in targeted drug delivery due to their high specificity and ability to bind to specific targets with high affinity. They can be conjugated to module delivery systems, enabling selective delivery to cells or tissues expressing the aptamer's target. It mainly includes the nucleic acid aptamer and peptide aptamer. |
| Functionalized Lipid | Functionalized lipids have emerged as a promising targeting module for targeted delivery in various biomedical applications. These lipids are designed to possess specific biological functions or interactions, allowing them to selectively bind to and accumulate in specific cells, tissues, or organs. It includes DSPE-PEG-Folate,DSPE-PEG-Biotin,DSPE-PEG-COOH, etc.. |
| Targeted Polymer | Targeted polymers have emerged as a promising class of materials for targeted delivery of therapeutics, diagnostics, and imaging agents. They are designed to accumulate selectively in specific cells or tissues, reducing side effects and enhancing efficacy. |
| Responsive Materials | Responsive materials, also known as smart materials, are designed to change their properties in response to specific stimuli, enabling targeted drug delivery and release. These materials can be responsive to a variety of stimuli, including temperature, pH, light, ROS, glucose and magnetic fields. |
 Module Delivery Systems
Module Delivery Systems
A number of model candidates that are highly potent and have low therapeutic indications can be targeted to the required diseased site using the appropriate delivery system. For instance, some drugs encapsulated in liposomes can have a significantly altered pharmacokinetics. Creative Biolabs has pioneered the development of advanced delivery strategies, including liposomes, LNPs (with a focus on liposomes and LNPs), lipid-polymer hybrid nanoparticles, exosomes, virus-like particles (VLPs), Fc fusion proteins, albumin, and more.
Liposomes constitute sophisticated drug delivery platforms, structured as nanoscale vesicles with amphiphilic phospholipid membranes. These systems enable temporally and spatially regulated therapeutic payload release, enhancing bioavailability through physiological synchronization during treatment while facilitating site-specific biodistribution to designated cellular or anatomical sites.
Lipid nanoparticles (LNPs) are precision-engineered for tissue-specific biodistribution through advanced molecular architecture. Their proprietary bilayer configuration enables optimized cellular internalization and intracellular payload release via endosomal bypass. This organotropic delivery paradigm demonstrates clinical potential across oncology, inherited metabolic pathologies, and infectious agent management. By compartmentalizing therapeutic cargo within LNPs, researchers achieve enhanced pharmacodynamic profiles with minimized off-target effects, positioning these nanovehicles as transformative tools in next-generation biotherapeutics.
Lipid-polymer hybrid nanoparticles (LPHNPs) are core-shell architectures merging lipid and polymer matrices, harnessing synergistic advantages of both vesicular and polymeric nanovectors. These systems enable superior payload retention, temporal release modulation, extended systemic persistence, and programmable biodirectionality—positioning them as transformative platforms across oncology, nucleic acid transport, and next-generation immunization strategies.
Exosomes—naturally occurring endogenous nanovesicles—are emerging as novel therapeutic platforms owing to their intrinsic tropism for molecularly defined cellular niches. By encasing pharmacological payloads within lipid bilayer membranes, these vesicles achieve directional cargo transfer to pathological loci, maximizing therapeutic indices while minimizing systemic toxicity. Their inherent biosafety profile and natural degradability further position them as preferred biocarriers for precision medicine applications.
Virus-Like Particles (VLPs) are non-pathogenic nanostructures derived from viral capsid components, functioning as versatile carriers for site-specific therapeutic payload delivery. These particles can be engineered via surface functionalization to direct cargo to designated cellular or anatomical regions, amplifying treatment precision while curtailing off-target effects.
Fc fusion proteins represent bioengineered constructs that integrate the immunoglobulin-derived Fc segment with therapeutic payloads, augmenting delivery efficiency and pharmacodynamic performance. The Fc region—a conserved antibody component—is strategically utilized for its Fc receptor binding capacity on immune effector cells, extending circulatory persistence of the conjugated biomolecule and enabling directional transport to pathological sites.
Albumin emerges as a transformative therapeutic carrier, leveraging its status as the body's primary circulatory transporter of nutrients via polyvalent binding domains and extended plasma persistence. Rational conjugation strategies involve covalently linking biologics to this endogenous protein through site-specific bioconjugation at its amino- or carboxyl-terminal regions, capitalizing on albumin's structural versatility to enhance drug stability and systemic retention.
Experience the Creative Biolabs Advantage - Get a Quote Today
Creative Biolabs is confident in offering customized targeted delivery products and services to meet clients' specific needs. If you are interested in our service, please do not hesitate to contact us for more details.
Reference