Tailored development of modules for specific drug payloads and ocular targets, including nanocarrier-based systems (polymeric nanoparticles, liposomes, niosomes, micelles, microparticles), and advanced formulations for SCS delivery.
Eye Targeting Module Development Service
Accelerate Your Targeted Drug Delivery Research!
Are you currently facing challenges with systemic toxicity, poor ocular bioavailability, or frequent, invasive drug administration in your ophthalmic programs? Our Eye Targeting Module Development services at Creative Biolabs help you overcome these critical hurdles, achieve highly specific drug delivery to ocular tissues, and enhance therapeutic outcomes through advanced nanotechnology, novel anatomical targeting, and smart drug release systems.
Contact our team to get an inquiry now!
Overview
The visual system exhibits acute vulnerability to inflammatory, infectious, and degenerative conditions provoking discomfort, visual anomalies, functional deficits, or irreversible sight loss. Anterior segment pathologies—spanning allergic conjunctivitis, aqueous-deficient dry eye, microbial keratitis, corneal erosions, aberrant angiogenesis, stromal thinning pathologies, and lysosomal storage defects—differ fundamentally from posterior conditions like choroidal neovascularization, hyperglycemia-driven retinopathy, glaucomatous neurodegeneration, inherited photoreceptor dystrophies, retinal inflammation, posterior uveitis, and intraocular tumors. Demographic analyses confirm escalating incidence strongly linked to population aging trends. Lifestyle factors, particularly rising diabetes incidence driving retinopathy rates, significantly influence disease burden. Furthermore, environmental pollutants and sustained digital device exposure are implicated in anterior segment pathologies, notably dry eye syndrome.
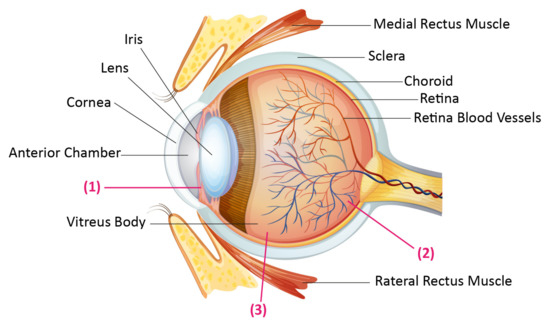 Fig.1 Human eye anatomy, and the drug administration routes.1,3
Fig.1 Human eye anatomy, and the drug administration routes.1,3
Delivery System Targeting Eye
While topical ophthalmic solutions remain the most convenient delivery modality for anterior segment disorders, their therapeutic efficacy is limited by rapid precorneal clearance and poor corneal permeability. Recent advances have yielded engineered nanocarriers and bioadhesive matrices specifically addressing these pharmacokinetic challenges. Poly(lactic-co-glycolic acid) nanoparticles encapsulating flurbiprofen demonstrated sustained release kinetics in preclinical models. This nanoparticulate system exhibited superior anti-inflammatory responses versus conventional eyedrop formulations in comparative studies. Similarly, α-cyclodextrin-integrated polymeric micelle hydrogels fabricated using amphiphilic copolymers demonstrated enhanced transcorneal permeation and anterior chamber retention of diclofenac in experimental models.
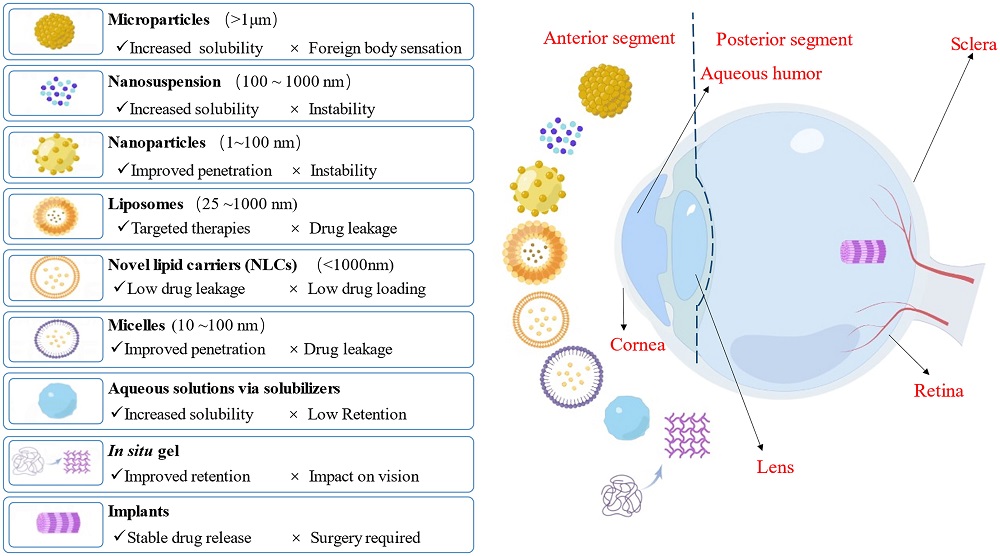 Fig.2 Schematic representation of novel ophthalmic dosage forms.2,3
Fig.2 Schematic representation of novel ophthalmic dosage forms.2,3
Posterior segment ophthalmic pathologies predominantly affect retinal and choroidal tissues. Significant research efforts have focused on engineering advanced delivery platforms to address these posterior ocular disorders. Core to these advancements are extended-release systems that reduce dosing frequency through biodegradable scaffolds (hydrogels), nanovehicles (nanoparticles, implants), and liposomal constructs. As an example, single-layer nanocarriers loaded with pressure-lowering therapeutics achieved prolonged ocular hypertension mitigation in animal studies, preserving controlled drug release profiles for four months following a single periocular injection. These advancements in depot-forming delivery systems are catalyzing progress in precision ophthalmic therapeutics.
What We can Offer?
Creative Biolabs offers a comprehensive suite of products and services designed to support your ocular drug development needs through advanced targeting module development:
Custom Eye Targeting Module Design & Synthesis
Ligand Discovery & Conjugation Services
Identification and conjugation of highly specific ligands (antibodies, peptides, aptamers, carbohydrates, fatty acids) to your chosen delivery system for active, receptor-mediated targeting.
Stimuli-Responsive Drug Delivery System Development
Engineering "smart" modules that release drugs in response to specific physiological triggers within the ocular environment.
Comprehensive In Vitro Ocular Barrier Models
Advanced in vitro assays to predict drug permeability across corneal, conjunctival, and retinal barriers.
Robust In Vivo Preclinical Evaluation
Expertise in preclinical animal models for ocular drug delivery, assessing pharmacokinetics, biodistribution, efficacy, and safety in the eye.
Experience the Creative Biolabs Advantage - Get a Quote Today
Why Choose Us?
Choosing Creative Biolabs for your Eye Targeting Module Development ensures access to unparalleled expertise and cutting-edge solutions, delivering tangible benefits for your ophthalmic drug development programs.
- Unrivaled Expertise: Our team comprises seasoned biology specialists with over two decades of experience in ocular biology, drug delivery, and pharmaceutical development.
- Comprehensive Technology Platform: Our platform employs an advanced technological matrix spanning multimodal nanovehicles (polymeric NPs, lipid vesicles, non-ionic carriers, amphiphilic aggregates, microspheres), controlled-release depot devices, and thermos responsive hydrogels, enabling bespoke therapeutic configurations.
- Precision Anatomical Targeting: We pioneer innovative administration pathways like suprachoroidal targeting, enabling anatomically precise therapeutic deposition to chorioretinal tissues—critical for posterior ocular disease management.
- Active Targeting Proficiency: Our platform engineers and functionalizes diverse targeting moieties (immunoglobulins, recombinant proteins, bioactive peptides, lipid moieties, glycan structures, and oligonucleotide aptamers) to achieve cell/microbe-specific localization of therapeutic payloads.
- Innovative Stimuli-Responsive Systems: We pioneer the development of "smart" drug delivery systems that react to physiological cues within the eye, enabling controlled and on-demand drug release.
- Reduced Toxicity and Enhanced Safety: Our targeted approaches are designed to minimize systemic exposure and off-target ocular toxicity, leading to improved drug safety profiles.
Workflow
FAQs
Here are some common questions we receive about Eye Targeting Module Development at Creative Biolabs:
How do your ocular-targeting platforms optimize therapeutic precision compared to standard delivery methods?
Our technology enables localized containment of therapeutic agents at ocular disease sites, boosting therapeutic concentrations while minimizing systemic dispersion. This precision targeting increases therapeutic efficacy and lowers toxicity risks by overcoming drug delivery limitations, such as poor ocular retention and premature systemic clearance, typically associated with traditional methods.
What ocular conditions can your precision delivery systems treat?
Our platforms address multifaceted ophthalmic conditions across anatomical regions. We develop interventions for anterior/posterior axis disorders such as age-related macular degeneration, diabetic retinal complications, optic nerve degeneration, uveitic inflammation, infectious corneal pathologies, and transplant-related immune responses. This adaptable design architecture permits precise calibration of administration variables tailored to distinct pharmacological requirements across ocular disease categories.
Are your Ocular Targeting Systems compatible with diverse therapeutic modalities (small molecules, biologics, gene editing tools)?
Our platform features multi-agent compatibility, specifically engineered to transport and stabilize varied pharmacological cargo. From low-MW compounds to macromolecular biologics, nucleic acid vectors, or cellular therapeutics, our team configures nanostructured vectors with precision biofunctionalization protocols. This ensures optimized biodistribution and payload integrity for your specific therapeutic modality.
Creative Biolabs provide tailored targeted delivery solutions addressing unique research and therapeutic requirements. To explore these capabilities, please contact us for more information.
References
- Durak, Saliha, et al. "Niosomal drug delivery systems for ocular disease—Recent advances and future prospects." Nanomaterials 10.6 (2020): 1191. doi:10.3390/nano10061191
- Liu, Ye, et al. "Research progress of ophthalmic preparations of immunosuppressants." Drug Delivery 30.1 (2023): 2175925. doi:10.1080/10717544.2023.2175925
- Distributed under Open Access license CC BY 4.0, without modification.



