Podocyte dysfunction underlies multiple inherited nephropathies including Alport syndrome, membranous nephropathy, and focal segmental glomerulosclerosis. The podocyte-specific αvβ3 integrin facilitates viral entry into kidney cells while functioning as a molecular gateway for enhanced therapeutic agent internalization.
Kidney Targeting Module Development Service
Accelerate Your Targeted Drug Delivery Research!
Are you currently facing challenges such as systemic toxicity, off-target effects, and inadequate drug concentrations at the disease site when developing pulmonary therapeutics? Our Lung Targeting Module Development service helps you overcome these limitations, ensuring precise and effective delivery of therapeutic agents directly to the lung through innovative targeting modules and advanced delivery systems. Creative Biolabs is pioneering precision in pulmonary therapeutics.
Contact our team to get an inquiry now!
Overview
The kidney system maintains fluid-ion balance and systemic metabolic equilibrium. Nephron impairment underlies chronic kidney disorders constituting a worldwide healthcare challenge. Existing therapies such as dialysis or transplant procedures are economically restrictive and pose substantial treatment-related complications. These limitations emphasize the urgent need for innovative kidney preservation strategies. Kidney-targeted pharmacotherapy employing molecular guidance systems may improve intrakidney drug bioavailability while reducing off-organ adverse effects through optimized tissue distribution.
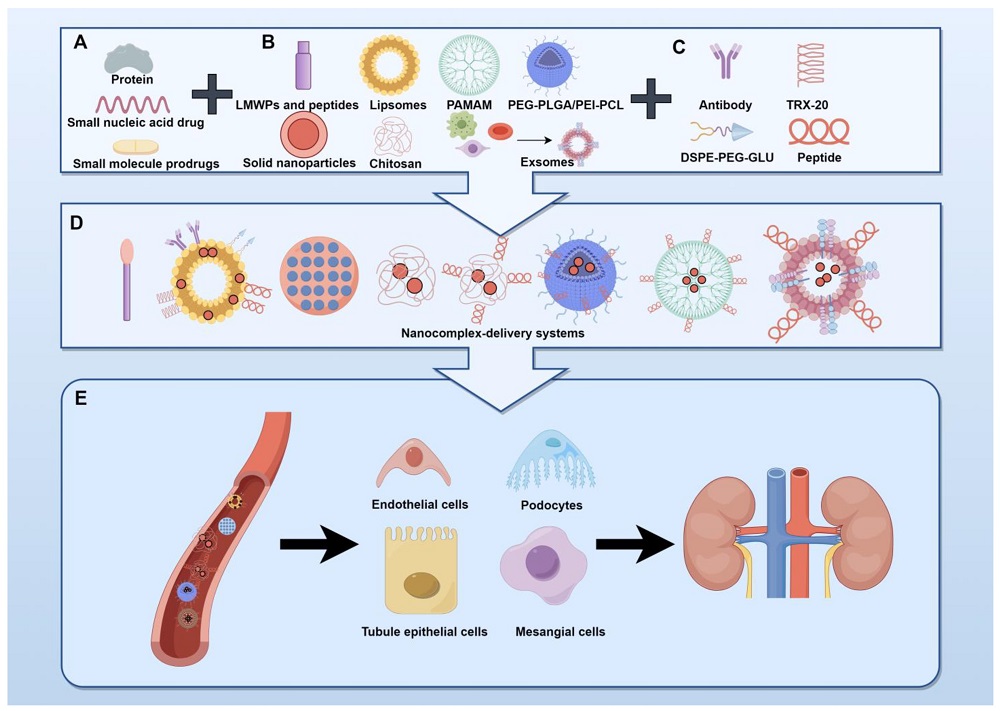 Fig.1 Delivery System Targeting Lung.1,3
Fig.1 Delivery System Targeting Lung.1,3
Delivery System Targeting Kidney
Cell-selective kidney delivery enables precision therapeutic interventions. Cell-type-specific vectorization approaches provide diverse molecular toolkits for developing diagnostic and treatment modalities addressing pathologies affecting distinct nephron populations.
Podocytes
Glomerular endothelial cell barrier and basement membrane
Glomerular endothelial cells and podocytes collaboratively synthesize a specialized extracellular matrix layer—the glomerular basement membrane—primarily consisting of collagen IV, laminin, and proteoglycans. Computational models have predicted peptide sequences capable of recognizing kidney endothelial biomarkers. Suzuki et al. engineered glycosylated arginine vasopressin (AVP) derivatives (CYFQNCPRG) conjugated to multiple saccharide moieties via C8 alkyl linkers to enhance cellular binding specificity. Experimental data demonstrate that these conjugates undergo kidney internalization via basolateral membrane transport following circulatory delivery.
Tubular cells
Kidney tubular epithelia demonstrate heightened vulnerability to damage, positioning them as critical therapeutic targets for inflammatory and fibrotic nephron injury. Bidwell’s team engineered an elastin-like polypeptide (ELP) vector system for precise tubular addressing. Phage-derived ligands including the G3-C12 motif exhibited rapid kidney accumulation through proximal tubule endocytic uptake within three minutes post-administration.
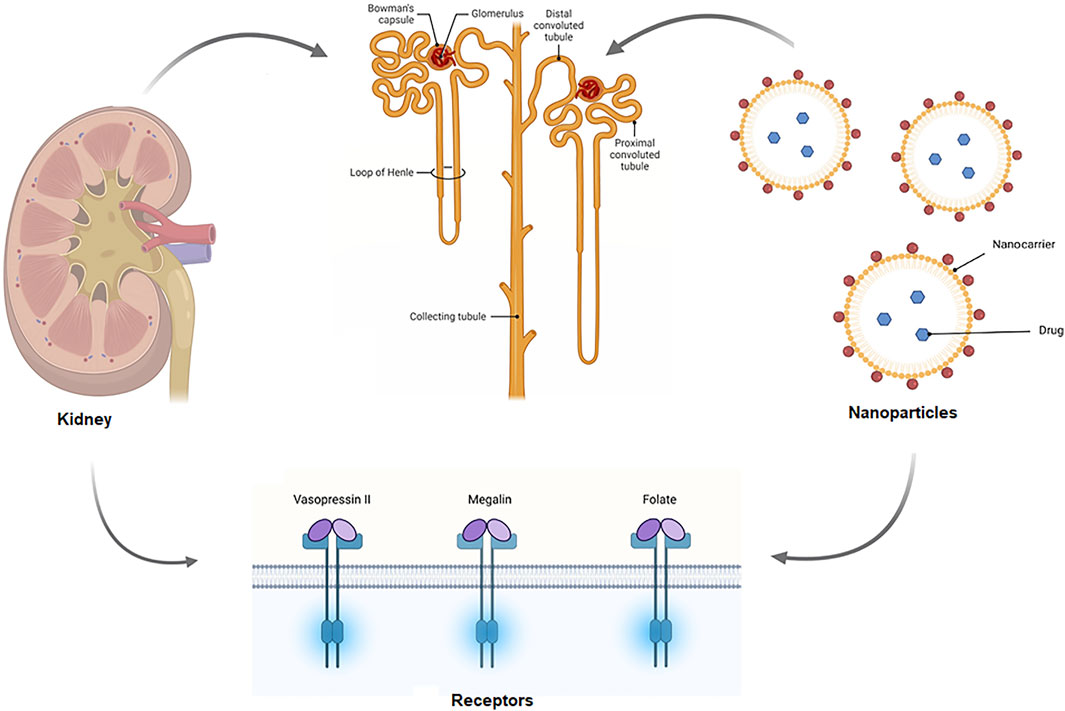 Fig.2 Schematic of the mechanism of targeted drug delivery in the kidney.2,3
Fig.2 Schematic of the mechanism of targeted drug delivery in the kidney.2,3
What We can Offer?
We offer a robust portfolio of products and services designed to meet diverse research and development needs:
Customized Kidney Targeting Module Design and Synthesis
Development of novel peptides, antibody fragments, aptamers, or small molecule ligands with high specificity for kidney-specific targets.
Targeted Nanoparticle and Liposome Development
Design and synthesis of various nanoparticle platforms and liposomes, functionalized with our proprietary kidney targeting modules for enhanced drug delivery.
Module-Payload Conjugation Services
Expert conjugation of your therapeutic payload (small molecules, proteins, nucleic acids, gene therapies) to our targeting modules or functionalized carriers, ensuring optimal stability and biological activity.
Preclinical Evaluation of Targeting Efficacy
Comprehensive in vitro and in vivo studies to rigorously validate the performance of your kidney-targeted drug delivery system.
Consultation and Project Management
Expert guidance from our seasoned scientists at every stage of your project, from target identification to preclinical validation, ensuring seamless execution and successful outcomes.
Experience the Creative Biolabs Advantage - Get a Quote Today
Why Choose Us?
Creative Biolabs stands at the forefront of Kidney Targeting Module Development, offering unparalleled expertise and cutting-edge solutions. Our distinct advantages ensure your project's success:
- Proven Expertise: Over two decades of experience in specialized biology and targeted delivery systems, ensuring deep scientific understanding and innovative problem-solving.
- Comprehensive Module Portfolio: Access to a diverse range of pre-validated and custom-designed targeting modules, including peptides, antibody fragments, and small molecules, tailored for various kidney cell types and pathologies.
- Nanoscale Delivery Mastery: Expertise in multifunctional nanovector systems (polymeric/lipid-based architectures) enabling pharmacokinetic optimization and tissue-selective biodistribution, with efficacy benchmarks established through peer-reviewed validation.
- Rigorous Validation: Our commitment to rigorous in vitro and in vivo evaluation guarantees the specificity, efficacy, and safety of developed modules, providing robust data to support your preclinical and clinical stages.
- Custom Engineering: Precision-formulated development pipelines engineered to your clinical objectives and technical specifications through adaptive design paradigms, eschewing standardized methodologies.
- Accelerated Development: Our streamlined workflow and experienced team significantly reduce development timelines, bringing your targeted therapies to fruition faster.
Workflow
FAQs
Here are some frequently asked questions from potential clients interested in Kidney Targeting Module Development:
What therapeutic payload classes integrate with your renal-targeted delivery systems?
Our modular platform supports multifunctional payload classes—low-molecular-weight agents, peptidic therapeutics, protein-based biologics (catalytic enzymes/antibody derivatives), nucleic acid modalities (siRNA/mRNA platforms), and viral/synthetic gene delivery systems. We co-develop nanostructured carriers with payload-customized biophysical parameterization during partner-driven development cycles.
How do you ensure the specificity of kidney targeting and minimize off-target effects?
We employ a multi-faceted approach to ensure specificity. This includes rigorous selection of highly selective targeting ligands, optimization of linker chemistry for stable conjugation, and comprehensive in vitro (e.g., cell-specific binding assays) and in vivo (e.g., biodistribution studies) evaluations to confirm targeted accumulation and assess any off-target interactions.
Which kidney molecular targets are addressable through your delivery platforms?
Our targeting portfolio spans specialized nephron populations (podocyte/mesangial in glomerular compartments; proximal/distal tubular epithelial subtypes) and pathology-specific biomarkers, along with dysregulated molecular signatures—receptors/transporters exhibiting kidney tissue-selective overexpression during disease progression.
Creative Biolabs provide tailored targeted delivery solutions addressing unique research and therapeutic requirements. To explore these capabilities, please contact us for more information.
References
- Huang, Li-Feng, et al. "Research Progress of Drug Delivery Systems Targeting the Kidneys." Pharmaceuticals 17.5 (2024): 625. doi:10.3390/ph17050625
- Merlin, JP Jose, and Xiaogang Li. "Role of nanotechnology and their perspectives in the treatment of kidney diseases." Frontiers in Genetics 12 (2022): 817974. doi:10.3389/fgene.2021.817974
- Distributed under Open Access license CC BY 4.0, without modification.



