We possess a comprehensive module delivery system, allowing us to provide tailored solutions from concept to preclinical validation.
Lung Adenocarcinoma Targeting Module Development Service
Accelerate Your Targeted Drug Delivery Research!
Are you currently facing challenges in developing highly specific and effective therapies for lung adenocarcinoma, struggling with off-target toxicity or limited drug delivery to tumor sites? Our Lung Adenocarcinoma Targeting Module Development services at Creative Biolabs help you accelerate the creation of novel, precise therapeutic agents by leveraging advanced targeted delivery systems, biomimetic nanoparticles, and innovative ferroptosis induction strategies. We provide tailored solutions to enhance therapeutic efficacy and minimize adverse effects.
Contact our team to get an inquiry now!
Overview
As the predominant malignancy among respiratory disorders and a foremost global mortality factor, lung cancer is histologically classified into small-cell (SCLC) and non-small-cell carcinomas (NSCLC). Adenocarcinoma, the most prevalent NSCLC subtype, arises in peripheral pulmonary regions from airway-lining secretory epithelia. While smoking-associated, it represents the predominant form among never-smokers. Therapeutic management of respiratory malignancies requires sustained high drug concentrations achieved via pulmonary or systemic delivery. Conventional antineoplastic regimens suffer from suboptimal tumor bioavailability leading to dose-limiting toxicity due to poor target specificity. Site-directed pharmaceutical deployment to pulmonary lesions enhances therapeutic precision, amplifying local efficacy while curtailing systemic drug exposure.
Delivery System Targeting Lung Adenocarcinoma
Recent advances have yielded multi-scale pulmonary delivery platforms engineered for oncological pulmonary intervention. These include nanoscale to microscale vehicles such as polymeric micelles, lipid-polymer hybrids, inhalable architectures (solid lipid nanocomposites, nanocapsules, core-shell configurations), gene therapy vectors, and particulate systems spanning liposomal carriers, polymer conjugates, and structured lipid matrices—all optimized for localized therapeutic action.
Precision-engineered drug delivery platforms exhibit superior tumor localization, diagnostic visualization, and extended in vivo therapeutic durability compared to passive systemic alternatives. Surface modification engineering optimizes critical bioperformance metrics: circulatory longevity, cellular internalization efficiency, lesion-specific payload accumulation, endolysosomal evasion, and stimulus-activated therapeutic release at disease loci
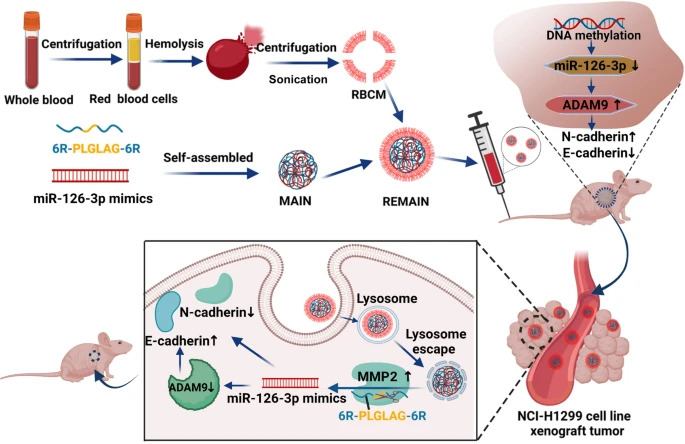 Fig 1. The reversion of DNA methylation-induced miRNA silence via biomimetic nanoparticles-mediated gene delivery for efficient lung adenocarcinoma therapy.1
Fig 1. The reversion of DNA methylation-induced miRNA silence via biomimetic nanoparticles-mediated gene delivery for efficient lung adenocarcinoma therapy.1
SP5-2, a synthetic peptide targeting NSCLC, demonstrates selective binding to carcinoma cells. Conjugation to liposomal nanocarriers amplifies its receptor-mediated intracellular uptake efficiency. SP5-2-functionalized Lipo-Dox exhibits enhanced tumor-targeting efficacy, reducing systemic toxicity while improving survival rates in metastatic, synergistic, and orthotopic murine models. Data revealed SP5-2-conjugated liposomal doxorubicin (SP5-2-LD) achieved 11.2-fold greater tumor accumulation versus free doxorubicin, with a 159.2-fold increase in plasma AUC. Bioavailability assessments validated SP5-2's role in promoting tumor-selective nanocarrier retention. Pharmacokinetic optimization via SP5-2 conjugation further enabled controlled biodistribution and sustained release kinetics, demonstrating superior therapeutic index.
What We can Offer?
At Creative Biolabs, our expertise in Lung Adenocarcinoma Targeting Module Development translates into a suite of tangible products and services designed to empower your research and therapeutic goals. We offer:
Complete Module Delivery System
Individual Targeting Modules
We can provide custom-designed individual targeting modules engineered for high specificity to lung adenocarcinoma cells.
Module-Payload/Carrier Complexes
We specialize in developing various types of module-payload/carrier complexes, enabling precise delivery of therapeutic agents to specific subcellular organelles within cancer cells.
Extensive Product Portfolio
We offer a wealth of corresponding products for you to choose from, providing flexibility and diverse options to meet your specific project requirements.
In vitro and in vivo evaluation of targeting module efficacy
Comprehensive assessment of your targeting modules' performance in both controlled laboratory settings and relevant animal models, providing robust data on their therapeutic potential.
Experience the Creative Biolabs Advantage - Get a Quote Today
Why Choose Us?
Creative Biolabs offers a unique combination of expertise, cutting-edge technology, and a steadfast customer focus that distinguishes us in the field of Lung Adenocarcinoma Targeting Module Development. We are committed to providing our clients with the highest quality research services and innovative solutions to accelerate their drug discovery and development efforts for lung cancer.
- Core Competencies: Our multidisciplinary team includes specialists with deep domain knowledge in lung adenocarcinoma biology, therapeutic vector engineering, and advanced drug delivery systems.
- Advanced Technology: We utilize state-of-the-art technologies and methodologies to design, synthesize, and evaluate targeting modules. This includes access to advanced chemical synthesis platforms, cell culture facilities, in vitro microfluidic systems, and comprehensive in vivo animal models.
- Precision Module Development: Leveraging bespoke project frameworks, we co-develop precision targeting modules aligned with client-specific molecular targets and clinical translation roadmaps through iterative design-test-optimize cycles, ensuring optimal performance for lung adenocarcinoma.
- Quality and Reliability: Stringent quality assurance protocols ensure batch-to-batch consistency and reproducibility in our module development. Our rigorous analytical methods and preclinical validation guarantee the high quality and reliability of our services.
- Proven Track Record: Our work is supported by extensive Published Data and successful case studies demonstrating the effectiveness and safety of our lung adenocarcinoma targeting strategies.
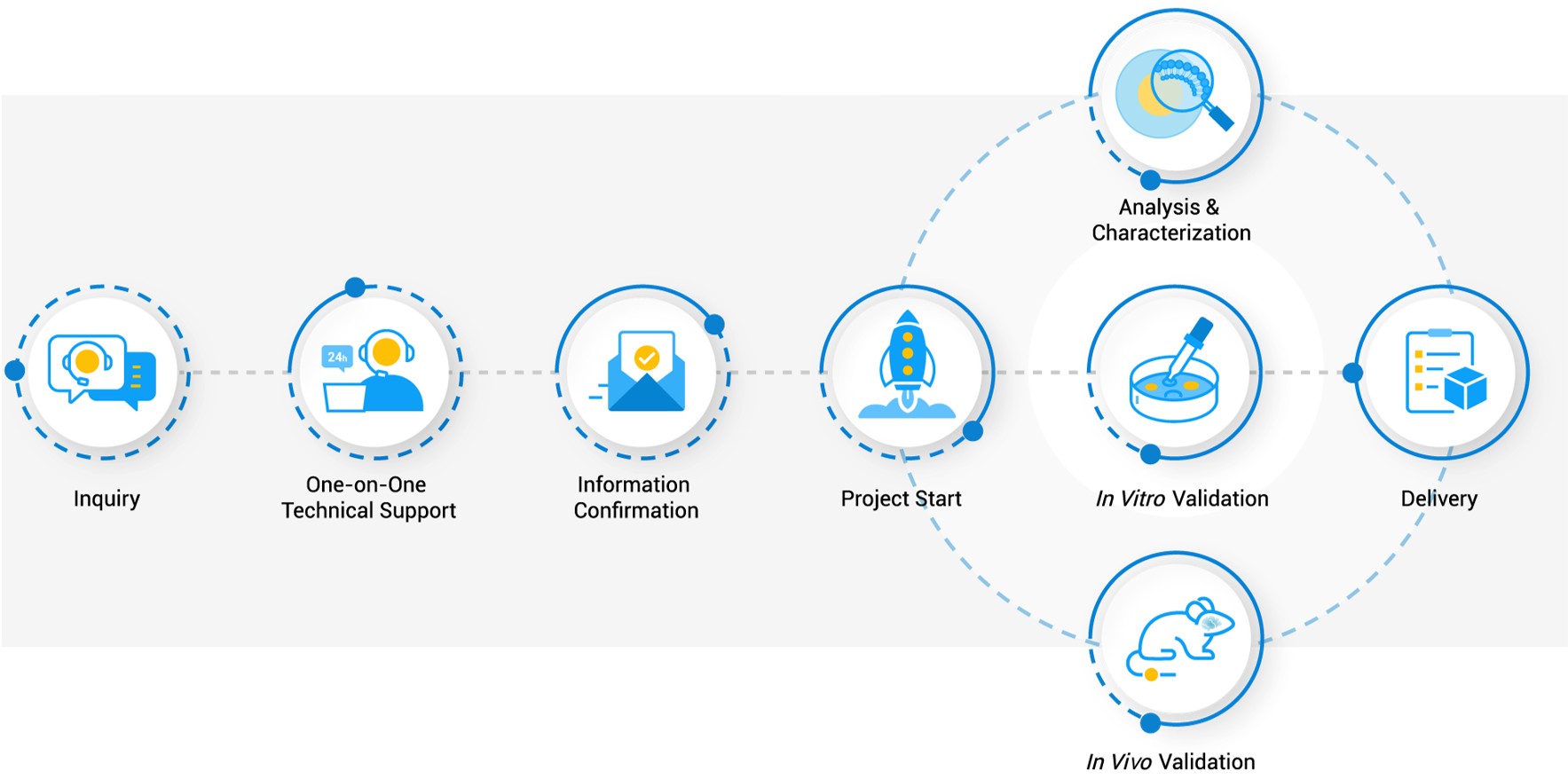
Workflow
FAQs
How does Creative Biolabs ensure the specificity of its Lung Adenocarcinoma Targeting Modules?
Our approach to specificity is multi-faceted. We begin with rigorous target validation, focusing on biomarkers highly expressed in lung adenocarcinoma cells. Our module design incorporates advanced engineering principles, such as antibody-drug conjugates or biomimetic nanoparticles, which are designed to selectively bind to these targets. We then perform extensive in vitro and in vivo characterization to confirm precise tumor accumulation and minimal off-target interaction.
What types of therapeutic payloads can be incorporated into Creative Biolabs' targeting modules?
Creative Biolabs' platform is highly versatile, capable of incorporating a wide range of therapeutic payloads. This includes small molecule drugs, nucleic acids (e.g., miRNA for gene therapy), proteins, and even agents designed to induce specific cell death pathways like ferroptosis. Our team will work with you to select and optimize the best payload for your specific therapeutic goal.
How does Creative Biolabs address the challenge of drug resistance in lung adenocarcinoma?
We address drug resistance by designing targeting modules that can overcome common mechanisms of drug resistance. This includes developing modules that target alternative pathways, utilizing novel mechanisms of action like ferroptosis induction, or engineering systems for intracellular delivery that bypass efflux pumps. Our preclinical models also include drug-resistant cell lines to validate the module's efficacy against challenging phenotypes.
Creative Biolabs provide tailored targeted delivery solutions addressing unique research and therapeutic requirements. To explore these capabilities, please contact us for more information.
Reference
- Liang, Lu et al. "The reversion of DNA methylation-induced miRNA silence via biomimetic nanoparticles-mediated gene delivery for efficient lung adenocarcinoma therapy." Molecular cancer vol. 21,1 186. 28 Sep. 2022. doi:10.1186/s12943-022-01651-4. Distributed under Open Access license CC BY 4.0, without modification.



