Overview Activation Pathways Molecular Events Activation Regulation Complement Activation in Diseases Resources
Overview of the Complement System
The complement system is made up of more than 30 proteins which lie in an inactive proenzyme state in the blood. These elements can be broken down into the following groups.
Table. 1 Proteins and their functions of different pathways in the complement system.
|
Category
|
Components
|
Function
|
|
Classical Pathway
|
C1
|
Recognizes and binds IgG or IgM, initiates complement cascade activation
|
|
C2
|
Binds to C4b to form C4b2a
|
|
C4
|
Hydrolyzed to C4a and C4b
|
|
Lectin Pathway
|
MBL
|
Recognizes specific glycan structures on the surface of pathogenic microorganisms to initiate complement activation
|
|
MASP-1
|
Binds to pattern recognition molecules and initiates complement cascade
|
|
MASP-2
|
As effector enzymes and key mediators
|
|
Alternative Pathway
|
C3
|
Forms C3bBb , thereby initiating and amplifying the complement cascade
|
|
Factor B
|
Initiates and amplifies alternative pathways
|
|
Factor D
|
Initiates and amplifies alternative pathways
|
|
Terminal Pathway
|
Membrane attack complex (MAC)
|
Forms complement activation pores in membrane
|
|
Central Components
|
C3 and C5
|
For opsonization and MAC formation
|
|
C3 Convertase
|
Converts C3 into C3a and C3b
|
|
C5 Convertase
|
Converts C5 into C5a and C5b
|
|
Regulatory Proteins
|
Factor H
|
Prevents activating complement on host cells
|
|
C1 Inhibitor (C1-INH)
|
Inhibits C1 in the classical pathway
|
|
Decay-Accelerating Factor (DAF)
|
Dissociates C3/C5 convertases
|
|
Complement Receptors
|
CR1, CR2, CR3
|
Facilitate immune cell interaction and pathogen recognition
|
The complement system has diverse and vital roles in immunity, ranging from direct pathogen elimination to immune system regulation. These functions can be broadly categorized as follows:
-
Opsonization
-
Inflammation
-
Pathogen lysis
-
Immune complex clearance
-
Bridging innate and adaptive immunity
Pathways of Complement Activation
Inherent and acquired immunity involve both complement system activation. After activation of complement, complement can help antibodies and phagocytes rid the body of microbes and infected cells and inflame the body.
Central to complement system are the three primary complement pathways of complement activation: classical pathway, lectin pathway, and alternative pathway. Each pathway is distinct in its initiation mechanisms but converges in a common terminal pathway that results in MAC formation.
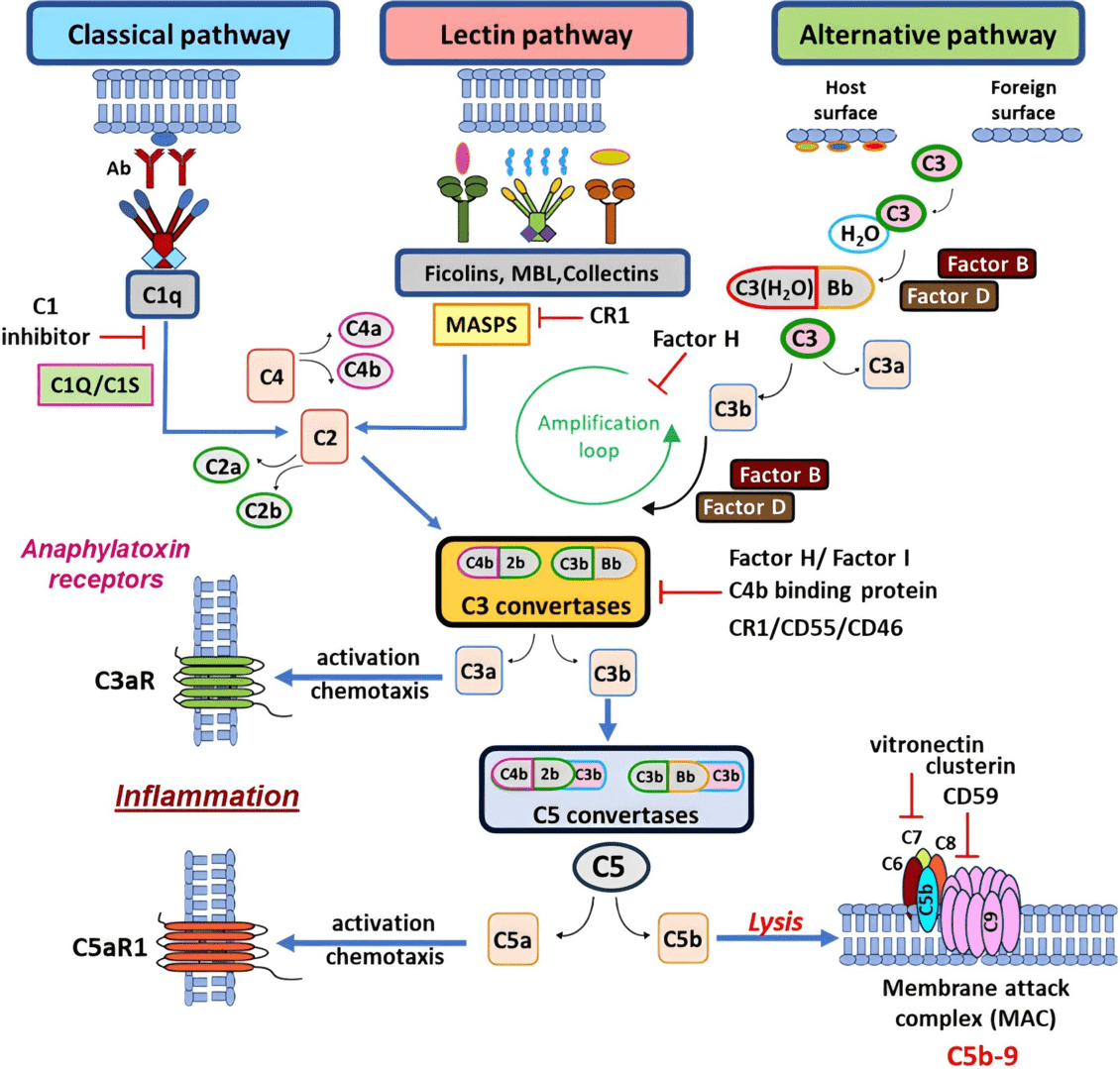
Fig. 1 Complement pathway diagram.1, 2
The classical pathway for complement activation is initiated by:
-
Initiation
-
C1 complex (C1q, C1r, C1s) binds to the Fc region of IgG or IgM in immune complexes.
-
C1q recognizes and binds to antibodies, triggering a conformational change.
-
Enzymatic activation
-
Activated C1r cleaves and activates C1s, which in turn cleaves C4 and C2.
-
C3 convertase formation
-
C4b binds C2a, forming the classical C3 convertase (C4b2a).
-
Amplification
-
The C3 convertase cleaves C3 into C3a (anaphylatoxin) and C3b, which opsonizes pathogens and facilitates phagocytosis.
The MBL lectin pathway of complement system is initiated independently of antibodies, relying on pattern recognition molecules to identify carbohydrate structures on microbial surfaces.
-
Recognition
-
MBL or ficolins bind specific carbohydrate patterns on pathogens.
-
Enzymatic activation
-
MBL-associated serine proteases (MASPs) are activated upon binding, cleaving C4 and C2.
-
C3 convertase formation
-
Similar to the classical pathway, C4b2a is formed and acts as the C3 convertase.
The alternative pathway of complement system is unique in its ability to spontaneously activate without specific triggers, relying instead on constant low-level hydrolysis of C3 in the plasma.
-
Spontaneous hydrolysis
-
C3 undergoes hydrolysis to form C3(H2O), which binds Factor B.
-
Amplification loop
-
Factor D cleaves Factor B into Bb, forming the C3 convertase (C3bBb) stabilized by Properdin.
-
C3 convertase action
-
Cleavage of additional C3 molecules amplifies the response.
-
C5 convertase formation
-
Addition of another C3b to C3bBb forms the C5 convertase (C3bBbC3b).
Activated complement involves a very strong degree of amplification. Serine proteases play an important role in human physiology and pathology, activating each other to promote initiation and amplification of the complement cascade. They have a common domain that contains the catalytic triad of histidine, aspartic acid and serine residues. The serine proteases of the complement system include C1r, C1s, MASPs, C2 of the classical/lectin complement pathway, and Factor B, Factor D, and Factor I.
Key Molecular Events in Complement Activation Pathways
The complement pathway activation involves precisely coordinated molecular events that culminate in pathogen elimination, inflammation modulation, and immune complex clearance.
Table. 2 Key molecular components and their functions.
|
Component
|
Molecular Weight (kDa)
|
Primary Function
|
Activation Products
|
Key Interactions
|
Regulatory Proteins
|
|
C1q
|
460
|
Pattern recognition, immune complex binding
|
N/A
|
IgG, IgM, CRP, PTX3
|
C1-INH, calreticulin
|
|
C4
|
205
|
Opsonization, C3 convertase formation
|
C4a (Anaphylatoxin)
C4b (Opsonin)
|
C2, C1s, MASP-2
|
C4BP, Factor I
|
|
C2
|
102
|
Enzymatic activity, C3 convertase component
|
C2a (Enzymatic)
C2b (Unknown)
|
C4b, MASP-2
|
DAF (CD55)
|
|
C3
|
185
|
Central component, amplification loop
|
C3a (Anaphylatoxin)
C3b (Opsonin)
|
Factor B, Factor H
|
Factor H, MCP, CR1
|
|
Factor B
|
93
|
Alternative complement pathway C3 convertase
|
Ba (Regulation)
Bb (Enzymatic)
|
C3b, Properdin
|
Factor H
|
|
MBL
|
96
|
Pattern recognition, lectin pathway initiation
|
N/A
|
MASPs, Carbohydrates
|
MASP-3
|
|
Factor D
|
24
|
Alternative pathway complement activation
|
Activated Factor D
|
Factor B
|
None known
|
|
C5
|
190
|
Terminal pathway initiation
|
C5a (Anaphylatoxin)
C5b (MAC)
|
C6-C9
|
CD59
|
|
Properdin
|
220
|
Alternative pathway stabilization
|
N/A
|
C3bBb
|
Factor H
|
|
Factor H
|
155
|
Alternative pathway regulation
|
N/A
|
C3b, Surface GAGs
|
Factor I
|
Regulation of Complement Activation
Precise regulation of complement activation is essential, as inappropriate activation can lead to host tissue damage and various pathological conditions.
Table. 3 Complement regulation pathways.
|
Pathway
|
Key Regulations
|
Primary Location
|
|
Classical Pathway
|
C1-INH, C4bp, Factor l, CD55,CD59
|
Plasma/Cell Surface
|
|
Lectin Pathway
|
C1-INH, MASP-3, Factorl, CD55, CD59
|
Plasma/Cell Surface
|
|
Alternative Pathway
|
Factor H, Factor l, CD55, CR1, CD59
|
Plasma/Cell Surface
|
Fluid-Phase Regulators
The regulation of complement activation in plasma involves several key proteins.
Membrane-Bound Regulators
Cell surfaces are protected by membrane-bound complement regulatory proteins.
Table. 4 Membrane-bound complement regulatory proteins.
|
Regulator
|
Primary Function
|
Location
|
|
CD46 (MCP)
|
Cofactor for Factor I
|
All nucleated cells
|
|
CD55 (DAF)
|
Accelerates convertase decay
|
Most blood cells
|
|
CD59
|
Prevents MAC formation
|
Widespread distribution
|
|
CR1
|
Multiple regulatory functions
|
Erythrocytes, leukocytes
|
Molecular Mechanisms of Regulation
Decay-accelerating factors promote the dissociation of C3/C5 convertases, preventing amplification of the complement cascade. This mechanism is particularly important in protecting host cells from complement-mediated damage.
The regulation of complement activation heavily relies on specific enzymatic processes. The regulation of C3 and C5 convertases represents a critical control point in the complement cascade pathways.
Several regulatory proteins serve as cofactors for Factor I-mediated cleavage of C3b and C4b, including Factor H, MCP (CD46), CR1 (CD35), C4BP.
Complement Activation in Diseases
The complement system is the centre of the immune system. When this system is engaged, its effects are profound for health and illness.
-
Complement activation in autoimmune diseases
In autoimmune diseases, abnormal complement activity drives tissue damage and disease. Complement activation causes immune complexes to cling to tissues and induce chronic inflammation and cell death.
-
Complement activation in infectious diseases
Over-activation or unsupervised stimulation can cause complement-induced tissue destruction. That's especially true in sepsis, where high complement levels cause endothelial cell destruction, vascular collapse and organ failure.
-
Complement Activation in Cancer Immunotherapy
Both hyperacute rejection and acute rejection are complement-mediated damage. The transplantation application of complement inhibitors - C1 esterase inhibitors for example - is designed to prevent rejection and prolong the life of grafts.
-
Complement Activation in in Transplantation Medicine
Hyperacute rejection and acute rejection are both associated with complement-mediated damage. The use of complement inhibitors in transplantation, such as C1 esterase inhibitors, aims to reduce the risk of rejection and improve graft survival.
-
Complement Activation and Therapeutic Approaches
It is one of the new potential therapeutic targets for the complement system pathways. Drugs inhibit complement activation at various points, particularly C5, preventing the formation of the MAC and reducing downstream tissue damage.
Table. 5 Current complement-targeted therapies.
|
Target
|
Indication
|
Mechanism of Action
|
|
C5
|
PNH, aHUS
|
Terminal complement inhibition
|
|
C5
|
PNH, aHUS
|
Long-acting C5 inhibition
|
|
C3
|
PNH
|
Proximal complement inhibition
|
|
Factor D
|
PNH
|
Alternative pathway inhibition
|
Creative Biolabs provides comprehensive complement-related products and complement testing services, including but not limited to:
Resources
References
-
Detsika, M. G., et al. "The complement cascade in lung injury and disease." Respiratory Research 25.1 (2024): 20.
-
under Open Access license CC BY 4.0, without modification
For Research Use Only.
Related Sections: