All products and services are For Research Use Only and CANNOT be used in the treatment or diagnosis of disease.
The outbreak of a novel coronavirus (COVID-19), which began in Wuhan, China, has spread rapidly and has now been confirmed in multiple countries. The International Virus Classification Commission has named this new coronavirus "SARS-CoV-2" (formerly: 2019-nCov) and identified it as a "sister virus" of SARS-CoV (Severe acute respiratory syndrome (SARS) coronavirus). SARS and COVID-19 have a common "hobby": they like to break into the same door, and this "door" is the receptor on the surface of infected cells - Angiotensin-converting enzyme 2 (ACE2). Numerous studies have fully demonstrated the important role of ACE2 in the pathogenesis of SARS infection. Existing studies have confirmed that the pathogenic mechanism of COVID-19 virus is highly similar to SARS in 2002-2003.
Empowered by decades of research experience in SARS-CoV in 2003, Creative Biolabs has built specific knowledge in the fields of virology and epidemiology. At present, our virologists have sufficient evidence to speculate that ACE2 - has great potential and is expected to be the next candidate for the treatment of COVID-19.
ACE2 is a zinc-containing metalloproteinase that can hydrolyze angiotensin I (angiotensin I) to Ang1-9, and hydrolyze angiotensin II (angiotensin II, Ang II) into vasodilating Ang1-7. Studies have shown that ACE2 is mainly expressed in alveolar epithelial cells and small intestinal epithelial cells, and is also expressed in vascular endothelial cells and arterial smooth muscle cells, and also exists in the basal cell layer of the epidermis.
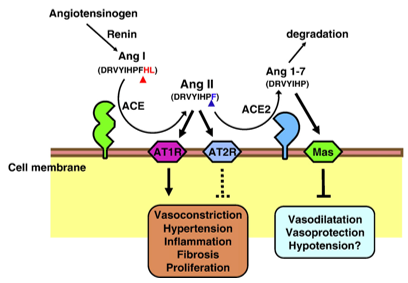 Fig.1 Schematic diagram of the role of ACE2 in the renin-angiotensin system. (Kuba, K, 2010)
Fig.1 Schematic diagram of the role of ACE2 in the renin-angiotensin system. (Kuba, K, 2010)
ACE2 has been identified as a receptor for SARS-CoV and human CoV-NL63 coronavirus. ACE2 is usually localized on the luminal surface of epithelial cells. When SARS coronavirus is infected through the luminal surface of ACE2-expressing cells, its infection efficiency is increased by 10 times.
A large number of studies have fully demonstrated that the COVID-19 virus also infects human alveolar epithelial cells through ACE2, and the pathogenic mechanism of COVID-19 is highly similar to SARS. Therefore, studying the action mechanism of ACE2 receptor and COVID-19 virus possess great significance for the development of drugs and vaccines for the treatment of diseases infected by the COVID-19 virus.
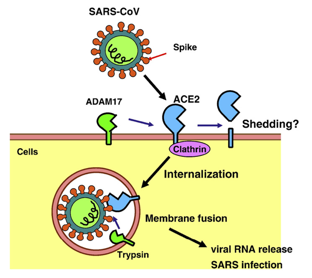 Fig.2 Mechanism of SARS-CoV infection. (Kuba, K, 2010)
Fig.2 Mechanism of SARS-CoV infection. (Kuba, K, 2010)
Creative Biolabs has been actively involved in the fight against the COVID-19 virus infection epidemic, hoping to utilize our strong technical strength, and research and development capabilities to contribute to the research of the COVID-19 epidemic. Powered by our proprietary Cre-In technology, we integrate human-derived ACE2 into the chromosome of CHO cells to obtain a cell model-ACE2-CHO that stably expresses ACE2 on the cell surface. This effective cell modeling tool helps researchers to further study the interaction between the receptor ACE2 and the COVID-19 virus, to explain the mechanism of virus invasion, and to develop drugs and vaccines to treat the COVID-19 virus. Welcome to consult us for more information.
The Cre-In method for rapid cell line preparation has the following advantages:
(1) Site-specific integration at high expression sites
The high expression "hot spots" were found in the CHO cell genome in advance. The Cre-LoxP homologous recombination method was used for site-specific integration, and the chromosome structure was open, and the expression was always constant.
(2) Simultaneous expression of multiple genes at a specific ratio (Stoichiometry)
The foreign genes were cloned into the pTOG3 and pTOG4 vectors. Through continuous targeting, the foreign genes were integrated into the designated high expression hotspots each time. The expression ratio of foreign genes is completely controllable.
(3) Only two antibiotic screening genes are needed, which can be recycled without limit.
(4) Almost 100% shooting efficiency. After 2 weeks of screening, the cell pool is ready for use.
For further details, please don't hesitate to contact us.
Reference
For any technical issues or product/service related questions, please leave your information below. Our team will contact you soon.
 NEWSLETTER
NEWSLETTER
The latest newsletter to introduce the latest breaking information, our site updates, field and other scientific news, important events, and insights from industry leaders
LEARN MORE NEWSLETTER NEW SOLUTION
NEW SOLUTION
CellRapeutics™ In Vivo Cell Engineering: One-stop in vivo T/B/NK cell and macrophage engineering services covering vectors construction to function verification.
LEARN MORE SOLUTION NOVEL TECHNOLOGY
NOVEL TECHNOLOGY
Silence™ CAR-T Cell: A novel platform to enhance CAR-T cell immunotherapy by combining RNAi technology to suppress genes that may impede CAR functionality.
LEARN MORE NOVEL TECHNOLOGY NEW SOLUTION
NEW SOLUTION
Canine CAR-T Therapy Development: From early target discovery, CAR design and construction, cell culture, and transfection, to in vitro and in vivo function validation.
LEARN MORE SOLUTION