Chimeric antigen receptor (CAR) T cells have shown great promise in the treatment of hematology and solid malignancies. However, despite the success in this field, there are still some major challenges, including accelerated T cell failure, potential toxicity, and insertional tumorigenesis. To overcome these limitations, the latest advances in clustering, regularly spaced, short palindromic repeat-related endonuclease 9 (CRISPR/Cas9) technology have achieved targeted interventions on endogenous genes in human CAR-T cells. The CRISPR/Cas9 tool provides a simple method to generate multiple gene modifications. So far, 11 clinical trials for gene-edited CAR-T cells have been registered. CRISPR-edited CAR engineering enables immune cells to detect tumor-specific antigens and activate them after binding to their cognate antigens.
At Creative Biolabs, we have established a powerful platform that combines CAR cell therapy with CRISPR technology, which unleashes the therapeutic potential of CAR cell therapy and enables our global customers to push their projects into clinical trials. Our CRISPR/Cas9 system has been used to knock out and knock into multiple CAR cell genome editing sequences, such as CAR-T, CAR-NK, and CAR-MA cells, as well as CAR-stem cells.
The use of CRISPR in genome editing is highly efficient and provides a simple and effective method to multiplex the processing of T cells. The CRISPR/Cas9 system has been used for sequence knockout and knock-in in mammalian genome editing (Figure 1). In principle, the deletion or insertion of the target gene is introduced by Cas9 nuclease guided by small RNA (sgRNA), which induces double-stranded DNA breaks, followed by non-homologous end joining (NHEJ) or homology-directed repair (HDR). In the NHEJ pathway, indels result in the deletion or insertion of nucleotides. In the HDR pathway, cofactors can promote genome recombination through two homology arms, leading to knock-in of the target gene.
Currently, several CRISPR-based strategies are being used to develop next-generation CAR-T cells through multiplexed genome editing. Such methods include knocking out endogenous genes (such as TCR, MHC, or autoantigens) to construct allogeneic universal CAR-T cells, destroying inhibitory receptors (such as CTLA-4, PD-1, or LAG-3), and integration of the CAR cassette into the endogenous TCR α constant locus (TRAC) or CC chemokine receptor type 5 (CCR5) locus.
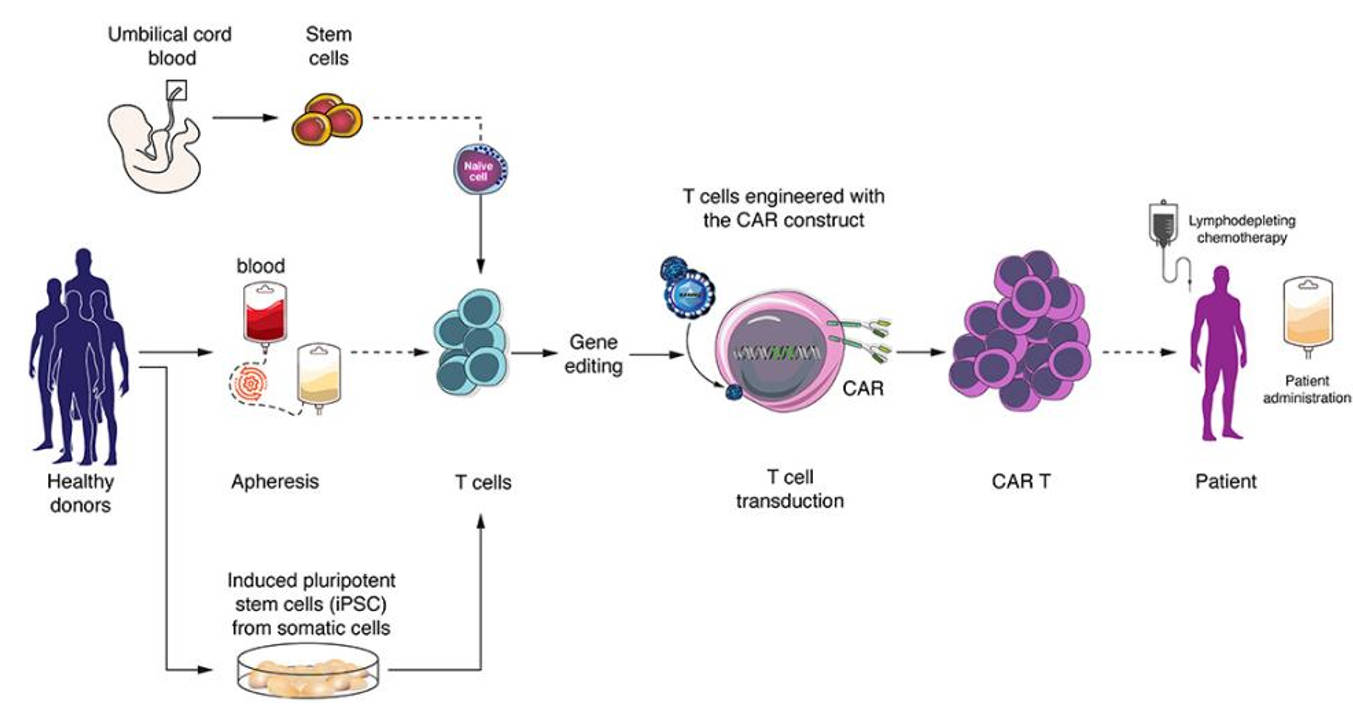 Fig.1 A schematic of applications of gene-editing technology in CAR-T cells.1
Fig.1 A schematic of applications of gene-editing technology in CAR-T cells.1
Creative Biolabs provides a comprehensive list of CRISPR-edited jurkat cells, please browse the table below to explore more details.
| KNOCK-OUT | |||||||||
| TRAC | TRBC | PD-1 | β2M | CD7 | LAG-3 | CD4 | CD5 | CD52 | Fas |
| CS-1 | CTLA-4 | PSCA | MHC | ||||||
| KNOCK-IN | |||||||||
| TRAC exon 1 | CCR5 | ||||||||
We also provide customized gene-editing services according to your interests and project needs. Please feel free to contact our team and get a complete solution about our CRISPR-edited CAR cell technology.
Reference
For any technical issues or product/service related questions, please leave your information below. Our team will contact you soon.
All products and services are For Research Use Only and CANNOT be used in the treatment or diagnosis of disease.
 NEWSLETTER
NEWSLETTER
The latest newsletter to introduce the latest breaking information, our site updates, field and other scientific news, important events, and insights from industry leaders
LEARN MORE NEWSLETTER NEW SOLUTION
NEW SOLUTION
CellRapeutics™ In Vivo Cell Engineering: One-stop in vivo T/B/NK cell and macrophage engineering services covering vectors construction to function verification.
LEARN MORE SOLUTION NOVEL TECHNOLOGY
NOVEL TECHNOLOGY
Silence™ CAR-T Cell: A novel platform to enhance CAR-T cell immunotherapy by combining RNAi technology to suppress genes that may impede CAR functionality.
LEARN MORE NOVEL TECHNOLOGY NEW SOLUTION
NEW SOLUTION
Canine CAR-T Therapy Development: From early target discovery, CAR design and construction, cell culture, and transfection, to in vitro and in vivo function validation.
LEARN MORE SOLUTION