In vivo immunotherapy currently confronts significant obstacles, including the intricate logistics, prohibitive costs, and scalability limitations inherent in ex vivo cell manufacturing. Creative Biolabs' CellRapeutics™ In Vivo B Cell Engineering Service enables the direct, systemic generation of persistent therapeutic cells within the patient. Our platform specializes in precisely reprogramming endogenous B cells into durable, self-renewing biologic factories. By leveraging proprietary viral vectors for targeted delivery and advanced synthetic gene circuits for controlled expression, we overcome traditional manufacturing bottlenecks to provide a scalable, long-lasting, and cost-effective solution for next-generation cell therapies.
In vivo B cell engineering is an advanced technology that enables the in situ genomic modification of B cells within a living organism by delivering gene editing systems and therapeutic genes directly into the body. This approach bypasses the complex and costly ex vivo cell manufacturing process and leverages the inherent longevity and memory functions of B cells to achieve sustained and stable secretion of therapeutic proteins, such as broadly neutralizing antibodies. It thereby offers a more scalable and cost-effective therapeutic paradigm for persistent treatment needs in areas including infectious diseases, oncology, and autoimmune disorders.
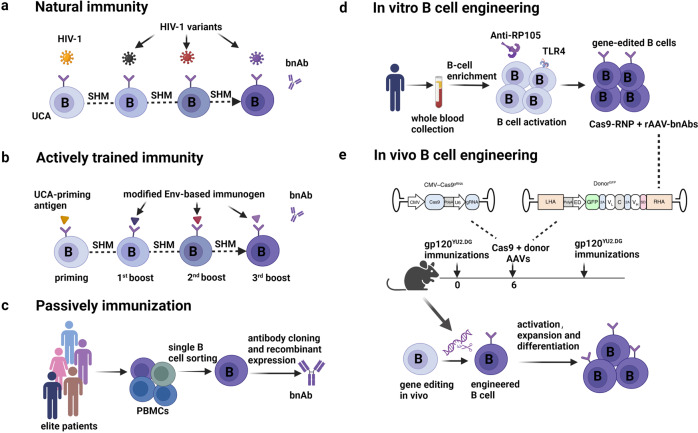 Fig.1 In vivo engineered B cells for enhanced anti-HIV-1 neutralizing antibody production.1
Fig.1 In vivo engineered B cells for enhanced anti-HIV-1 neutralizing antibody production.1
Creative Biolabs' CellRapeutics™ In Vivo B Cell Engineering Service directly reprogramming patients' own B cells in situ into durable, self-sustaining therapeutic protein factories, such as for bNAbs or custom biologics, we bypass costly ex vivo manufacturing. Our platform ensures sustained, targeted drug delivery, offering a precise and scalable solution for chronic infectious, oncological, and autoimmune disorders.
We offer a comprehensive in vivo B cell engineering platform that integrates targeted delivery, precision genome editing, programmable synthetic circuits, and optimized persistence strategies to enable the development of durable, self-sustaining cellular therapeutics.
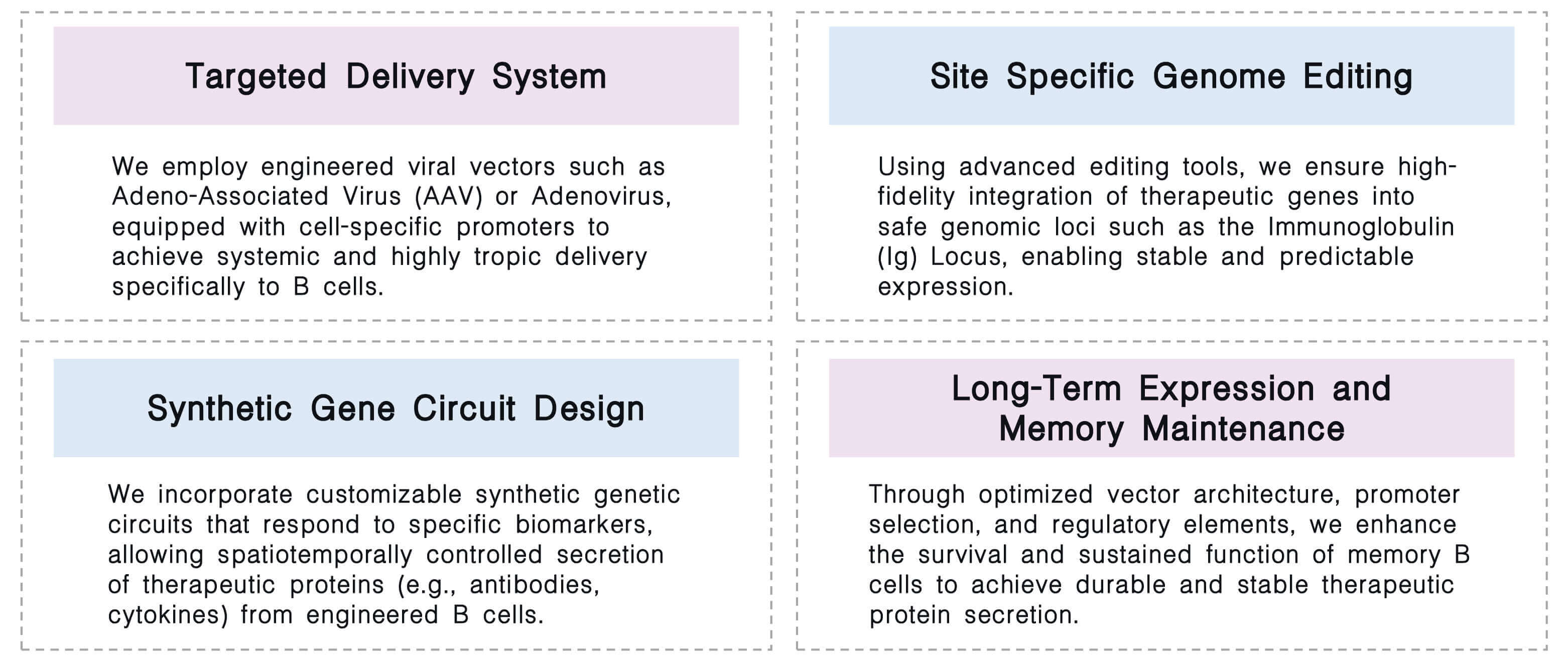
Required starting materials:
Key Steps:

Final Deliverables:
What is the primary advantage of In Vivo B Cell Engineering over traditional Ex Vivo CAR T/B therapies?
The main advantage is scalability and simplicity. By eliminating the need for patient apheresis, ex vivo manipulation in a cleanroom, and reinfusion, we dramatically reduce manufacturing complexity, cost, and time. Furthermore, the in vivo approach leverages the natural migration and long-term survival of B cells for more durable therapeutic effects.
How do you ensure the gene editing system is specific to B cells and avoids off-target effects?
We ensure high specificity through two mechanisms: 1) Vector Tropism: Selecting or engineering viral serotypes with a high affinity for B cell surface markers, and 2) Promoter Control: Using robust B cell specific promoters to drive the expression of genome editing components only within the targeted B cell population, significantly mitigating expression in non-target tissues.
Creative Biolabs' In Vivo B Cell Development platform bypasses the complexity of ex vivo processing by enabling direct, in situ genetic engineering. We deliver durable, scalable therapies through precise, locus-specific editing of B cells, leveraging their intrinsic memory and longevity for sustained therapeutic secretion. Our approach ensures antigen-restricted expression, reduced immunogenicity, and a streamlined path to clinical translation.
"The design of the custom B cell specific promoter was a game-changer. It eliminated the off-target editing and payload expression we previously observed with non-specific vectors, providing the clean safety profile needed to progress our autoimmune therapeutic."— Ay Js, Director of Immunology Research.
"Using Creative Biolabs' In Vivo B Cell Engineering in our research has significantly improved the longevity of our antibody-based antiviral therapy. The sustained, high serum titers achieved in our NHP model were simply not possible with conventional protein injections or ex vivo CAR therapies."— Dr. Dd Hn.
"Creative Biolabs' commitment to in vivo AAV-based delivery simplified our supply chain immensely. We were able to move from a highly specialized, multi-site ex vivo protocol to a single, scalable intravenous administration method, dramatically reducing our projected cost."— P. Ls Sa, Ph.D.
To harness the power of in vivo B cell engineering for simpler, more accessible therapies, partner with our experts. We offer comprehensive scientific support and project-specific consultation designed to accelerate your path to the clinic. Contact our team to initiate a dialogue and receive a detailed proposal.
Reference
For any technical issues or product/service related questions, please leave your information below. Our team will contact you soon.
All products and services are For Research Use Only and CANNOT be used in the treatment or diagnosis of disease.
 NEWSLETTER
NEWSLETTER
The latest newsletter to introduce the latest breaking information, our site updates, field and other scientific news, important events, and insights from industry leaders
LEARN MORE NEWSLETTER NEW SOLUTION
NEW SOLUTION
CellRapeutics™ In Vivo Cell Engineering: One-stop in vivo T/B/NK cell and macrophage engineering services covering vectors construction to function verification.
LEARN MORE SOLUTION NOVEL TECHNOLOGY
NOVEL TECHNOLOGY
Silence™ CAR-T Cell: A novel platform to enhance CAR-T cell immunotherapy by combining RNAi technology to suppress genes that may impede CAR functionality.
LEARN MORE NOVEL TECHNOLOGY NEW SOLUTION
NEW SOLUTION
Canine CAR-T Therapy Development: From early target discovery, CAR design and construction, cell culture, and transfection, to in vitro and in vivo function validation.
LEARN MORE SOLUTION