Background Service Technical Information Featured Services Highlights FAQs Contact
Creative Biolabs' CAR IVT products are specifically designed to enable the direct, in vivo reprogramming of T cells, bypassing the costly and complex ex vivo manufacturing process inherent to conventional cell therapies. We provide a comprehensive and customizable suite of high-quality IVT solutions, including CAR-encoding plasmids, modified mRNAs, circular RNAs, and LNP delivery systems. Our service integrates optimized product design, scalable manufacturing, and rigorous quality control into a seamless, end-to-end workflow. This integrated approach ensures researchers receive reliable, functional reagents that accelerate preclinical development, reduce time-to-validation, and support the advancement of next-generation, off-the-shelf immunotherapies.
Introduction
Chimeric antigen receptor (CAR) T-cell therapy is limited by costly and complex ex vivo manufacturing. To overcome this, research now focuses on reprogramming T cells directly within the body using synthetic nucleic acids, particularly in vitro-transcribed (IVT) mRNA. This strategy utilizes targeted nanocarriers to deliver transient, non-integrating CAR or TCR genes, enabling scalable, repeatable lymphocyte engineering. This approach shows promise in inducing robust anti-tumor responses against both blood and solid cancers, paving the way for accessible, off-the-shelf immunotherapies.
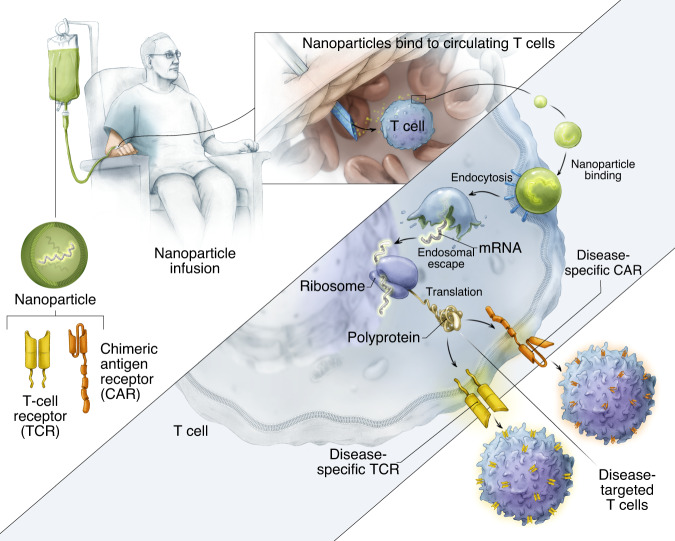 Fig.1 IVT mRNA-mediated in vivo T cell reprogramming.1
Fig.1 IVT mRNA-mediated in vivo T cell reprogramming.1
CAR IVT Products at Creative Biolabs
Creative Biolabs' CAR IVT platform offers an integrated solution designed to overcome the constraints of conventional viral CAR-T development. Our platform accelerates CAR candidate screening by enabling efficient delivery of high-purity mRNA into primary cells or via in vivo administration. Through controlled, transient expression, we support rapid assessment of novel binders' safety profiles and help mitigate off-tumor, on-target toxicity risks.
Technical Information
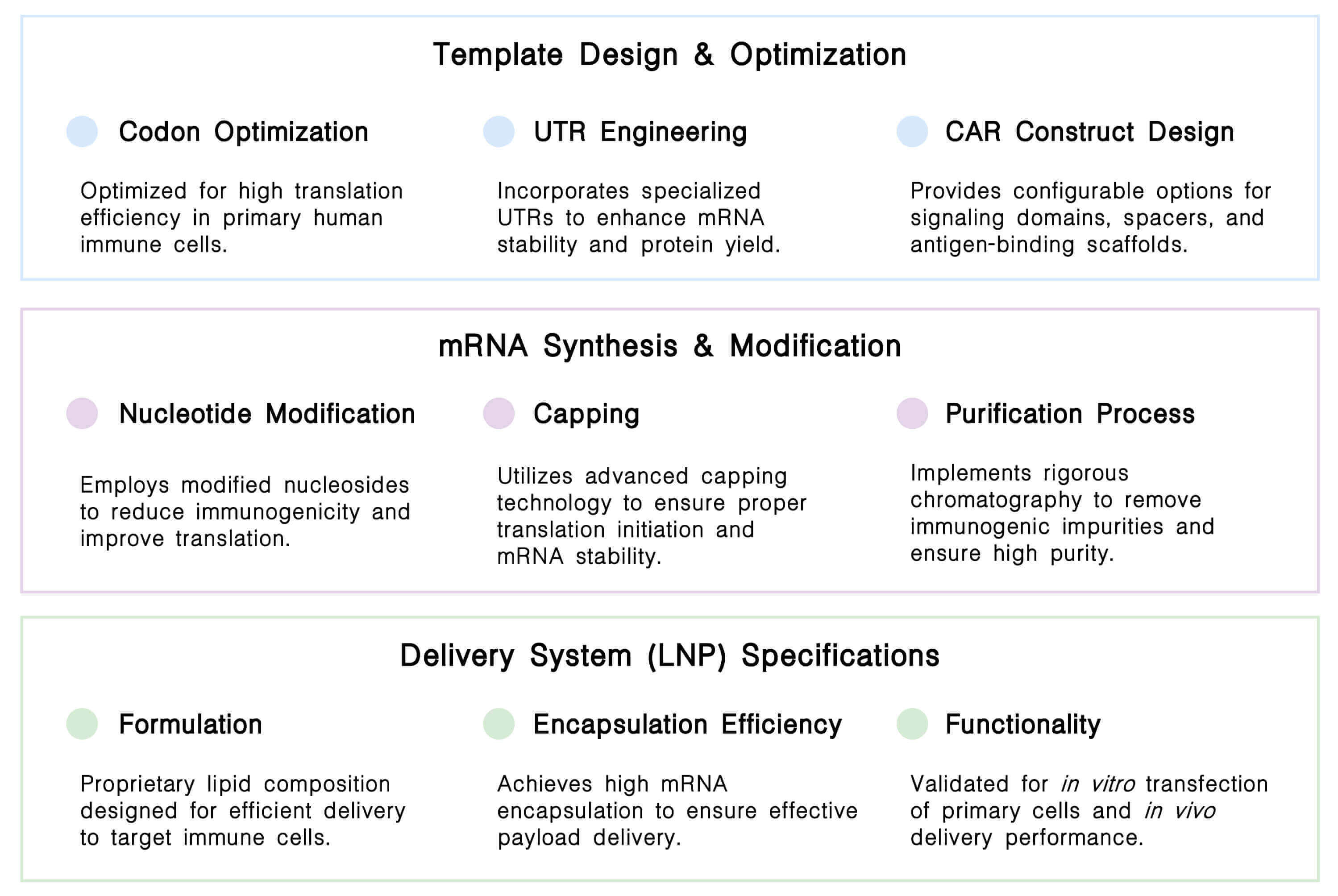
Our CAR IVT platform integrates a complete, controlled workflow, from optimized template design and modified mRNA synthesis through validated LNP delivery, ensuring high-purity, functional, and stable reagents for advanced cell engineering.
Featured Services
Our complete CAR IVT platform delivers an integrated workflow of specialized products, from plasmid design through final delivery, engineered to support robust and scalable cell therapy development across all key modalities.
Our CAR IVT plasmid portfolio provides linearized DNA templates optimized for in vitro transcription, enabling rapid, scalable production of CAR-encoding RNA. These high-purity plasmids support seamless development of mRNA, circRNA, and saRNA therapeutics for cell engineering applications, including CAR-T, CAR-NK, and CAR-Macrophage research. They are engineered with robust promoters and precise restriction sites to ensure consistent, high-yield transcription.
We offer research-grade IVT mRNA designed for transient CAR expression in primary immune cells. These products incorporate nucleotide modifications and optimized UTRs to enhance stability and translational efficiency, making them ideal for preclinical proof-of-concept, functional screening, and rapid CAR candidate validation in both in vitro and in vivo models.
Our LNP delivery systems are formulated for efficient in vivo or ex vivo transfection of CAR-encoding RNA into target immune cells. These customizable nanoparticles ensure high cellular uptake and endosomal escape, enabling robust in vivo T cell engineering and sustained functional CAR expression in preclinical animal studies and translational research programs.
Engineered for enhanced stability and prolonged protein expression, our circular RNA (circRNA) products provide a durable alternative to linear mRNA in CAR therapy development. They resist exonuclease degradation and enable extended CAR presentation in immune cells, supporting long-term functional studies and reducing dosing frequency in persistent in vivo models.
CAR IVT saRNA Products
The CAR IVT saRNA Products utilize self-amplifying RNA (saRNA) technology to deliver CAR-encoding constructs with high-level and prolonged antigen receptor expression. Derived from alphavirus replicons, saRNA contains an RNA-dependent RNA polymerase complex that drives intracellular RNA amplification, resulting in enhanced and durable CAR production at low RNA doses. This approach is ideal for generating potent and persistent CAR immune effector cells, especially when limited starting material or reduced transfection frequency is desired.
CAR mRNA-LNP conjugated Antibody Products
The core technology involves conjugating CAR mRNA-loaded Lipid Nanoparticles (LNPs) to specific monoclonal antibodies. These antibody-LNP conjugates systemically deliver the CAR genetic payload directly to predefined immune cell subsets in the patient's body, enabling the in vivo generation of functional CAR-equipped cells. This off-the-shelf approach is designed to bypass the complex ex vivo manufacturing of traditional CAR therapies, offering a rapid, flexible, and potentially safer strategy for treating cancers and other diseases.
Key Advantages
-
Enhanced Expression Design: Our proprietary codon and UTR optimization strategy maximizes protein expression in human or murine primary immune cells.
-
Scalable Synthesis Platforms: Equipped with high-yield production and purification systems capable of supporting studies ranging from murine proof-of-concept to multi-dose preclinical evaluations.
-
Quality-Driven Manufacturing: Production follows Quality-by-Design (QbD) guidelines and integrates process analytical technology to ensure transcript consistency and biological activity.
-
Stringent Quality Control: Rigorous analytical methods, including fragment analysis, endotoxin detection, and functional potency assays, provide quantitative quality verification.
FAQ
Why select IVT mRNA for CAR-T development?
IVT mRNA provides a non-integrating delivery mechanism, effectively avoiding insertional oncogenesis and enabling controlled, transient expression to improve therapeutic safety.
Are these products suitable for in vivo use?
Yes. Our LNP-formulated CAR IVT solutions are engineered for targeted or systemic delivery in preclinical models to facilitate direct in vivo assessment of T-cell engineering.
How is mRNA immunogenicity minimized?
Immunogenic potential is substantially reduced through chemically modified nucleotides and high-purity purification methods that eliminate dsRNA contaminants, thereby preventing unintended immune activation.
Why Choose Us?
We overcome CAR-T RNA instability with proprietary IVT systems that extend transcript half-life by 2-3 times. Our solutions achieve >90% T-cell transfection without compromising viability, delivering structurally optimized RNA for superior functional performance. Choose reliable, data-driven reagents to accelerate your cell therapy development.
How to contact us?
Enhance your CAR therapy development with our integrated IVT product platform. Consult our scientific team to explore tailored plasmid, mRNA, circRNA, and LNP solutions designed to advance your program. Connect with us to schedule a personalized discussion and receive a comprehensive project quotation.
Reference
-
Parayath, N N et al. "In vitro-transcribed antigen receptor mRNA nanocarriers for transient expression in circulating T cells in vivo." Nature communications vol. 11,1 6080. Distributed under Open Access License CC BY 4.0, without modification. https://doi.org/10.1038/s41467-020-19486-2.

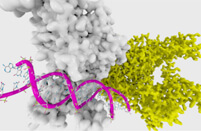
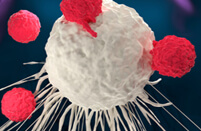



 Fig.1 IVT mRNA-mediated in vivo T cell reprogramming.1
Fig.1 IVT mRNA-mediated in vivo T cell reprogramming.1






