Glyco-engineered Pichia pastoris Expression System
Therapeutic glycoproteins have occupied an extremely important position in the market of biopharmaceuticals. Due to the ability of performing posttranslational modifications (PTM) and secreting the target protein to the cultivation broth, the methylotrophic yeast Pichia pastoris is widely used for glycoprotein production in biotechnology and biopharmaceutical industry. With Ph.D. level scientists and over a decade of experience in recombinant protein production, Creative Biolabs has successfully constructed a glyco-engineered P. pastoris to produce high-quality glycoproteins, which are similar to human.
Introduction of P. pastoris
P. pastoris, which belongs to the genus Komagataella spp, is an organism commonly employed to produce a variety of active proteins with N- /O-linked glycans. This expression system has the characteristics of fast growth, easy cultivation, simple genetic manipulation, and the ability to perform space folding and glycosylation of proteins. The glycans of the P. pastoris-produced proteins is high-mannose type without core fucose, leading to reduced in vivo half-life and biological activity, and besides, carrying high mannose chains, glycoproteins cause an immune response in the body. Due to the shortcomings of excessive glycosylation modification of this expression system, it cannot replace mammalian cells to produce various glycoproteins. Therefore, solving the problem of high mannose chain is the core step of glycosylation engineering of P. pastoris.
 Distributed under Open Access license CC BY 4.0, from Wiki, without modification.
Distributed under Open Access license CC BY 4.0, from Wiki, without modification.
Glyco-engineered P. pastoris for Glycoprotein Production at Creative Biolabs
Based on our cutting-edge genetic engineering technology, our scientists have modified the glycosylation pathway of P. pastoris by knocking out the och1 gene and introducing mannosidase and successfully constructed a glycoengineered P. pastoris expression system, with obvious advantages, including short production period, low cost, high yield, easy operation and scaling up. This expression system overcomes the disadvantage of excessive glycosylation of proteins to form high mannose chains in wild-type P. pastoris, allowing the production of glycoproteins with uniform low molecular sugar chains. This new type of expression system combines the advantages of Escherichia coli, mammalian cells, and yeast, and overcomes their shortcomings, making it a good choice for glycoprotein production.
Our Services Including but Not Limited to:
-
Sequence design of the gene sequence of target glycoprotein, codon optimization, and gene synthesis;
-
Expression vector construction;
-
Transformation and identification of positive strains by PCR;
-
Select positive strains for protein expression identification;
-
Large-scale culture and purification.
Highlights
-
Production of glycoproteins with uniform low molecular glycan chains
-
Production of glycoproteins with the natural state biological activity
-
Short production period, high yield, easy operation and scaling up
-
Ensuring the amount and purity of glycoprotein delivered
-
High flexibility and cost-effectiveness
With abundant experience and comprehensive powerful platforms for glycoprotein production, Creative Biolabs is a perfect partner to offer our clients high-quality glycoproteins to facilitate our customers’ research and project development. For more detailed information, please feel free to contact us or directly send us an inquiry.
Published data
P. pastoris is widely applied in the production of recombinant proteins, which can grow in specific media and has high protein productivity. In this study, the authors used microscopy and flow cytometry to study the differences in cell morphology and recombinant protein production of three differently glycosylated P. pastoris strains (recombinant P. pastoris wild type (wt), recombinant P. pastoris och1 knockout strain (∆och1 ), and recombinant Man5GlcNAc2 glycosylated strain (SuperMan5)). The results showed that the cell morphology of the two glycoengineered strains (∆och1 and SuperMan5) was similar, their single cells and budded cells were larger than the cell morphology of the wt strain, and there was a strong production of cell clumps. This was caused by shorter glycan structures on the cell surface and disturbed budding process, and the viability of the inner nuclei in these clumps was impaired. Despite this cell aggregation phenomenon, the SuperMan5 strain showed superior growth, physiology and recombinant protein productivity at 30 °C compared with other glycosylated strains.
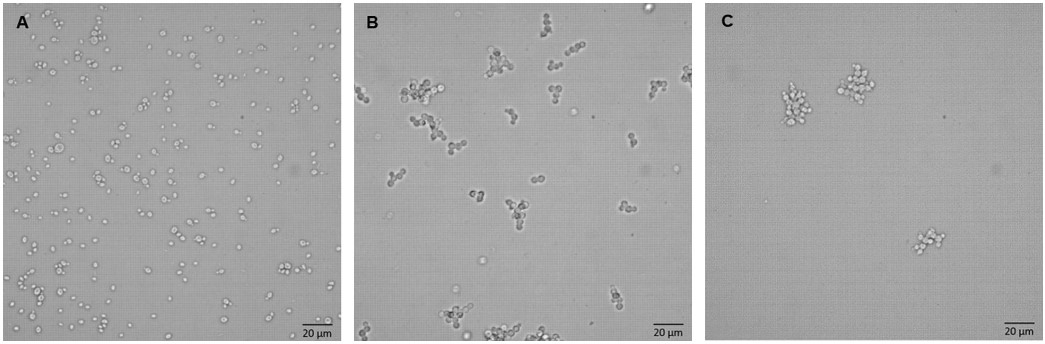 Fig.1 Microscopic results of yeast cells of different strains, A wt, B ∆och1, C SuperMan5.1
Fig.1 Microscopic results of yeast cells of different strains, A wt, B ∆och1, C SuperMan5.1
FAQs
Q1: Can you elaborate on the genetic modification of P. pastoris to reduce high mannose chains in glycoproteins?
A1: Absolutely, our glycoengineering approach focuses on two major modifications:
-
Knockout of the och1 gene: The och1 gene is responsible for initiating exo-link glycosylation, which results in high mannose glycan structures. By knocking out this gene, we prevent the addition of excessive mannose residues.
-
Introduction of mannosidase: We also incorporate mannosidase, which can influence the glycosylation pathway to produce glycoproteins with uniform low molecular weight glycans.
This dual approach minimizes immunogenic high mannose structures and helps produce glycoproteins with improved bioactivity.
Q2: How do you ensure the purity of the glycoproteins you produce?
A2: We employ multiple quality control measures to ensure the purity of our glycoproteins. For example, we use advanced chromatography techniques such as affinity, ion exchange, and size exclusion chromatography to purify glycoproteins and combine multiple mass spectrometry techniques to analyze glycan structures. In addition, we perform functional assays to confirm that the glycoproteins retain their biological activity in their native state.
Q3: What are the advantages of your system over traditional mammalian cell systems?
A3: Our glycoengineered P. pastoris expression system has the following advantages:
-
Short production cycle: P. pastoris grows faster and has a shorter production cycle than mammalian cells.
-
High yield: P. pastoris usually produces higher concentrations of target proteins than mammalian cells.
-
Easy to operate and scale up: P. pastoris culture conditions are simple and suitable for large-scale production. Moreover, our system overcomes the shortcomings of traditional P. pastoris over-glycosylation and will not cause immune response due to high mannose chains.
Customer Review
High Yield of Glycoproteins
"By collaborating with Creative Biolabs, we had successfully used their glyco-engineered P. pastoris expression system to produce the glycoproteins we need. This system not only greatly shortened the production cycle, but also had a very high yield, greatly improving our experimental efficiency. The gene synthesis and vector construction services were very good, and we were very satisfied with the purity and biological activity of the final protein."
Producing Uniform Low-Molecular-Weight Glycans
"The expertise and technical platform of the Creative Biolabs scientific team are impressive. They successfully knocked out the och1 gene and introduced mannosidase, so that the glycoproteins produced by P. pastoris had uniform low-molecular-weight glycans. This technical improvement significantly improved the in vivo half-life and biological activity of our products and avoided the occurrence of immune responses."
Reference
-
Pekarsky, Alexander, et al. "Production of a recombinant peroxidase in different glyco-engineered Pichia pastoris strains: a morphological and physiological comparison." Microbial cell factories 17 (2018): 1-15. Distributed under Open Access license CC BY 4.0, without modification.
For Research Use Only.
Related Services


 Fig.1 Microscopic results of yeast cells of different strains, A wt, B ∆och1, C SuperMan5.1
Fig.1 Microscopic results of yeast cells of different strains, A wt, B ∆och1, C SuperMan5.1

