Glycopeptide-based Cancer Vaccine Development Service
Anticancer immunotherapy has emerged as a new exciting area for controlling tumors. Particularly, vaccination using synthetic tumor-associated carbohydrate antigens (TACAs)-based vaccine hold promise for generating a specific antitumor response by targeting cancer cells. As a leading specialist in glycodesign and synthesis of complex glycopeptide for vaccine development, Creative Biolabs is committed to providing high-quality service in the synthesis of glycopeptide-based vaccine candidates to promote the progress of cancer vaccine development.
Background of TACAs as Potential Vaccine Antigens
A defining feature of cancer is altered glycosylation, including increased expression of certain TACAs. Commonly, changes in glycosyltransferase expression levels can lead to an increase in the size and branching of N-linked glycans. Overexpression of glycosyltransferases leads to the overexpression of certain terminal glycan epitopes on tumors, such as sLeX, sLea, sTn, LeY, Globo H and PSA. An expanding body of preclinical and clinical research shows that Abs against TACA can eliminate circulating tumour cells and micrometastases. Although TACAs are ‘self’ glycans, they are generally poorly expressed or inaccessible on normal, healthy tissues, these glycans may serve as potential vaccine Ags.
Certain glycoproteins, such as mucin, is expressed in almost all epithelial tissues and densely covered with complex O-linked carbohydrates. It is a membrane located glycoprotein which is often overexpressed on tumor cells. Mucin serves as a scaffold for several of the aforementioned TACAs, and glycolipids, such as gangliosides, a glycosphingolipid-containing sialic acid. In its large extracellular portion, it contains a variable number of tandem repeats of the sequence HGVTSAPDTRPAPGSTAPPA. One tandem repeat contains five potential O-glycosylation sites. MUC-1 mucin displays distinct carbohydrate structures, such as ThomsenFriedenreich (TF) and sialyl-Tn, the presence of which is attributed to reduced glycosylation activity. A vaccine based on synthetic sialyl-Tn has proven to be highly target-specific in human trials, and the induction of high anti-STn antibody titers correlated with prolonged survival of breast cancer patients.
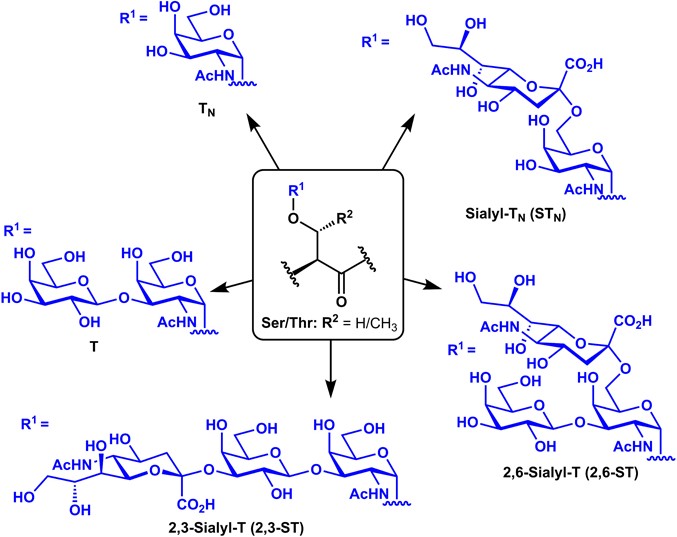 Fig.1 The structures of common TACAs.1, 2
Fig.1 The structures of common TACAs.1, 2
Synthesis of Glycopeptide-based Cancer Vaccine Candidate at Creative Biolabs
To trigger an efficient immune response, synthetic antigens are commonly linked to an immunogenic carrier protein. Since tetanus toxoid contains potent T-helper cell epitopes, which initiate T-helper cell stimulation and widely used in human medicine, it is a suitable carrier for the glycopeptide conjugate. Besides, to stimulate effective T-helper cell responses in human, Creative Biolabs has engineered a synthetic non-natural T-helper epitope, which can bind to a wide range of HLA class II molecules. Linear peptides linked to this carrier protein are found to be potent immunogens in vivo. Both two carrier proteins above can be used in the development of cancer vaccine candidates.
Creative Biolabs has successfully designed and synthesized a vaccine containing a synthetic MUC1 glycopeptide B-cell epitope covalently linked to a carrier protein corresponding to the T-helper epitope. The antibodies induced by this construct can efficiently recognize human tumor cell lines. The rational design and synthesis of glycopeptide constructs can be potentially used as cancer vaccine candidates for human use.
We have accumulated extensive experience in offering high-quality synthesis of diverse TACA glycopeptide B-cell epitopes, including Tn, T, sialyl-Tn, sialyl-T antigens, Lewis series antigens, and glycolipid-derived antigens, covalently linked to a carrier protein or peptide (according to every requirement of our clients) to induce T-helper epitope cell response.
Highlights
-
TACA glycopeptide B-cell epitopes, mainly including Tn, T, sialyl-Tn, and sialyl-T antigens.
-
Carrier proteins including tetanus toxoid and engineered peptide corresponding to T-helper epitope
-
Allowing an effective population coverage
-
Improved the therapeutic efficacy
For more detailed information in synthetic glycopeptide-based cancer vaccine development, please feel free to contact us and communicate with us about your specific puzzles. Our Ph.D. scientists will offer you professional solutions as soon as possible.
References
-
McDonald, David M., Scott N. Byrne, and Richard J. Payne. "Synthetic self-adjuvanting glycopeptide cancer vaccines." Frontiers in chemistry 3 (2015): 60.
-
Under Open Access license CC BY 4.0, without modification.
For Research Use Only.
Related Services

 Fig.1 The structures of common TACAs.1, 2
Fig.1 The structures of common TACAs.1, 2

