To execute progression and metastasis, cancer cells undergo metabolic evolution to maximize nutrient utilization for bioenergetic and biosynthetic demands, survive harsh tumor microenvironment (TME), and escape immunosurveillance. The aberrant glucose metabolism is a major kind of metabolic reprogramming in cancer. A majority of human tumors exhibit significantly higher glucose flux compared with adjacent normal tissues, and the glucose metabolism is characterized by increased aerobic glycolysis in the tumorous tissues, known as the Warburg effect. In fact, significant metabolic heterogeneity exists among the cells in a tumor. In reverse Warburg effect, cancer-associated fibroblasts (CAFs) also metabolize glucose through anaerobic glycolysis and export lactate, which is then taken up and utilized by oxidative cancer cells. Moreover, impaired immune function due to metabolic competition between cancer and immune cells is one of the critical factors of an immunosuppressive TME.
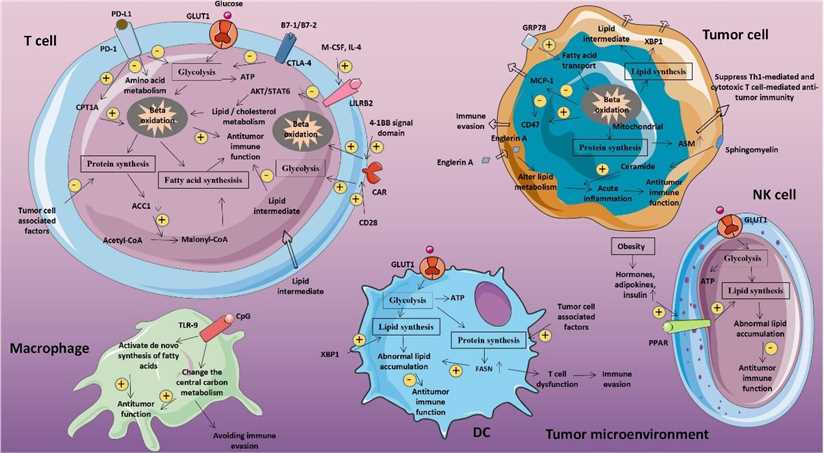 Fig. Abnormal glucose metabolism and tumors. (Wu, 2020)
Fig. Abnormal glucose metabolism and tumors. (Wu, 2020)
Tumor cell survival under aberrant metabolism of glucose is a vital step not only for the process of tumorigenesis but also to reshape TME. Nutrient competition and depletion of glucose in the TME by cancer cells have been proposed as a metabolic mechanism of immunosuppression.
The imbalance between glucose metabolism and cancer cell growth in TME is closely related to the occurrence and progression of cancer. Uptake and use of glucose can be controlled by a multitude of factors including enzymes (GLUT and hexokinase), mitochondrial defects, hypoxia-inducible factor, MYC, PI3K/Akt/mTOR, p53/TIGAR/PFKB3. Compared to conventional cytotoxic therapy, modulation of particular targets with altered glycolytic metabolism would reduce treatment toxicity. Targeting glucose metabolism becomes an attractive target to restore anti-tumor immunity and develop anticancer therapy.
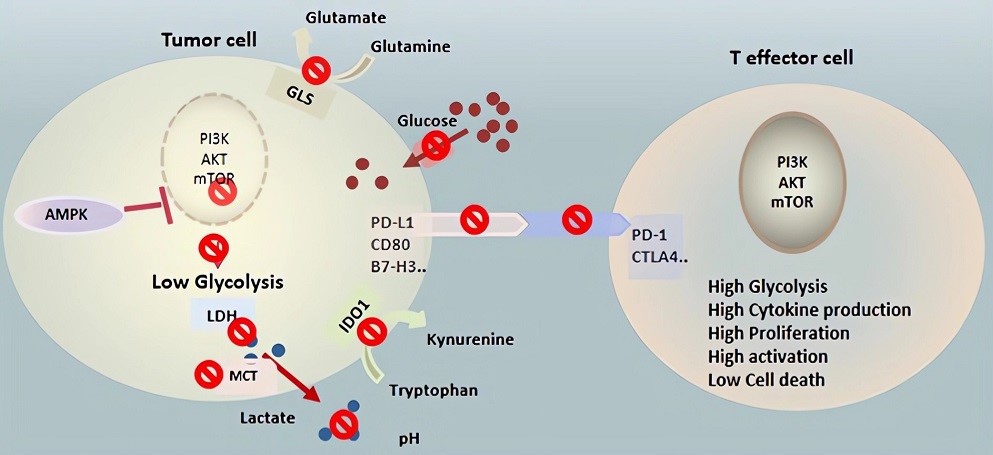 Fig.2 Therapeutic targeting cell metabolism in the TME. (Kouidhi, 2018)
Fig.2 Therapeutic targeting cell metabolism in the TME. (Kouidhi, 2018)
Several enzymes in the glycolytic pathway have been targeted, some showing tumoricidal effects in vitro and in vivo. Unfortunately, there has been little clinical success given that glycolysis is crucial to the glucose metabolism of normal cells as well.
2-DG competitively inhibits hexokinase through product inhibition due to the accumulation of 2-DG-6-phosphate (2-DG-6-P), which is not metabolized further causing the metabolic block. Numerous preclinical studies have demonstrated anti-proliferative effects of 2-DG but clinical success has been limited.
The AMPK pathway plays an important role in the TME. AMPK activation is thought to reduce tumor burden in part by slowing tumor growth, while concurrently supporting the expansion and survival of tumor-infiltrating lymphocytes in the TME. AMPK activity must be balanced, however, as excessive AMPK may inhibit effector T cell responses.
As PD-1 signaling inhibits glycolysis in T cells and PD-L1 in cancer cells stimulates aerobic glycolysis, this reciprocal glucose uptake associated with the PD-1-PD-L1 axis can be applied to metabolic modulation with immune checkpoint inhibitors as a new strategy for cancer immunotherapy.
Creative Biolabs provides high-quality services and helps you save precious time to focus on analyzing data and making project plan. With the help of our professional scientists, we are committed to developing services in targeting tumor metabolism.
References
For any technical issues or product/service related questions, please leave your information below. Our team will contact you soon.
All products and services are For Research Use Only and CANNOT be used in the treatment or diagnosis of disease.
 NEWSLETTER
NEWSLETTER
The latest newsletter to introduce the latest breaking information, our site updates, field and other scientific news, important events, and insights from industry leaders
LEARN MORE NEWSLETTER NEW SOLUTION
NEW SOLUTION
CellRapeutics™ In Vivo Cell Engineering: One-stop in vivo T/B/NK cell and macrophage engineering services covering vectors construction to function verification.
LEARN MORE SOLUTION NOVEL TECHNOLOGY
NOVEL TECHNOLOGY
Silence™ CAR-T Cell: A novel platform to enhance CAR-T cell immunotherapy by combining RNAi technology to suppress genes that may impede CAR functionality.
LEARN MORE NOVEL TECHNOLOGY NEW SOLUTION
NEW SOLUTION
Canine CAR-T Therapy Development: From early target discovery, CAR design and construction, cell culture, and transfection, to in vitro and in vivo function validation.
LEARN MORE SOLUTION