Introduction of CATSPERB
CATSPERB, a novel transmembrane protein in the CatSper1 complex, was identified from data base searches and reverse transcription PCR. CATSPERB contains two clear transmembrane domains (6-22 of the NH2 terminus, and 1060-1076 at the end of the COOH domain). A large extracellular domain (∼1000 amino acids) precedes the carboxyl terminal transmembrane segment. The predicted mouse intracellular carboxyl terminus is only 27 amino acids in length. Eight N-glycosylation sites are predicted in the polypeptide. The overall 2-TM topology is reminiscent of that of the large conductance K+ channel (BK) β subunits and the P2X receptor (ATP-gated channel), but CATSPERB (∼126 kDa) is significantly larger (BK β subunits <30 kDa).
| Basic Information of CATSPERB | |
| Protein Name | Cation channel sperm-associated protein subunit beta |
| Gene Name | CATSPERB |
| Aliases | CatSper-beta |
| Organism | Homo sapiens (Human) |
| UniProt ID | Q9H7T0 |
| Transmembrane Times | 4 |
| Length (aa) | 1,116 |
| Sequence | MESPLIYVSVLLLNIFEFSSGIVYNKDDTEKRFACSNKGFPQENEIIKLYLFLENLKIQCFFQTENEIASKAMLSVFTSGGLAPSLGIMNSTYNGIFHFNLTLFSDRILWLVDIPRENITQSTDIAAVEEWLVRITLHHGLNIYATEGTLLDVIREPILQWTPGDVIPESEISKLYPHVVDLKVTKCPCANDVALLGFIVDTIVDGVYIGITFGGFWHDYDTTWFNMTQTIYSQLQEEYEDLSLVDMVLTNHFLVILTSLGLFVSEDLRYPSRHSLSFSRADFCGFERVDYVKGKLWYNERCFANREHFEVDYVTVTFERNRTLSESSSCFYSQEPFLEWVPCLPHIFKGIKIFPTVLTFLVDQERGTGVYLFYNKVRKTAIASVSTLRNNEPNSQSKFPIFRFPSSFSSPVGMVFHPRSHFLYAYGNQIWLSVDGGNTFQLIANFHDDIIKKTFHSFYTSAITFVSQRGKVYSTKAGMGRYSAVGSVTERIFTLYYDHLGFLHKLTLGRFEASGPPTAFGNSRNLFGQPPDMGFETALAPQHTSLDEIIFFAYVPENEPQETIYSKKFGNIHYGKVIHSGKTGRAYIRKVLQHTTPKGFLSSVIAEMKEPFGLEEVNESSCLSSSLLINKAGNVYKLTLDSQVVQALFEDTDIEKTVVLPGYSSFLITSILDNKNALAIATMPESAPNNMTFLKSTWFLYNFGQRNGRTWKIYSKPCNYWFQHDDSPSLNIVKYIDLGNSYVLKAKVIRNAKGFRMLEIPLLTVFVGNPNLLEVTAEVTFDDTDSYVITISAASKVLHQGSTSLAFIMWSASTECFVTTMVPTLKSSCSYLRSMHHIPSKFIPFEDWISGVHKDSQGFNLIKTLPINYRPPSNMGIAIPLTDNFYHADPSKPIPRNMFHMSKKTGKFKQCANVSTREECNCTKDQKFSHAVAFSDCREKVPRFKFPITQYPVSLEIINEDGRVPLQSPYLVTVTEVNMRHNWKLKHTVPENIKRMKQLVEPILGAAVYNPSGLNLSIKGSELFHFRVTVISGVTFCNLIEEFQIYVDEAPLPFPGHTLIAVATAVVLGGLIFIAFMFQLQGIHPWRTFQRWIRRNQEKFSSISLSELIHRSKSEE |
Function of CATSPERB Membrane Protein
Four CatSper ion channel subunit genes (CatSpers1-4) are required for sperm cell hyperactivation and male fertility. The four proteins assemble (presumably as a tetramer) to form a sperm-specific, alkalinization-activated Ca2+-selective channel. Like the CatSper ion channel subunits, CATSPERB is restricted to testis and localized to the principal piece of the sperm tail. CATSPERB protein is absent in CatSper1(-/-) sperm, suggesting that it is required for trafficking or formation of a stable channel complex. CATSPERB is the first identified auxiliary protein to the CatSper channel.
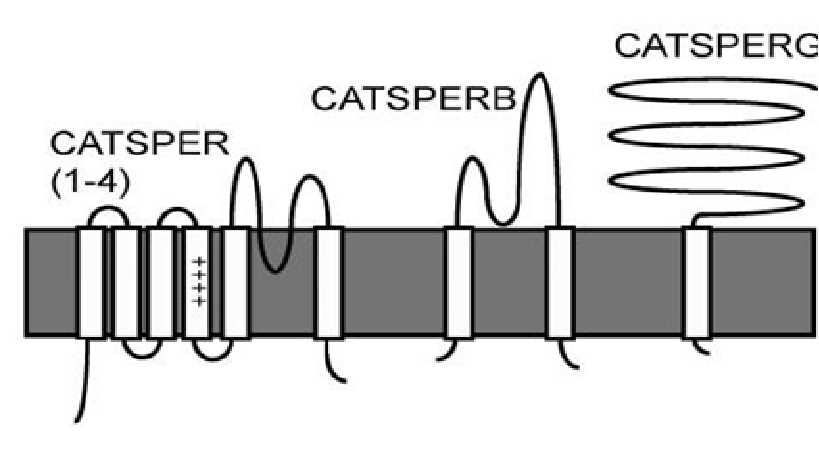 Fig.1 A model depicting the composition of CATSPER channel complex and the putative membrane topology for CATSPERs (CATSPER1-4), CATSPERB and CATSPERG. (Wang, 2009)
Fig.1 A model depicting the composition of CATSPER channel complex and the putative membrane topology for CATSPERs (CATSPER1-4), CATSPERB and CATSPERG. (Wang, 2009)
Application of CATSPERB Membrane Protein in Literature
In this article the authors predict that the CatSperβ protein has a large extracellular fragment (∼1000 amino acids) with several putative N-glycosylation sites, and they speculate that these extracellular domains could be “sensors” for sperm interactions with other cell types or surfaces.
This study demonstrated that the polymorphisms of the CATSPERB (rs1298989) and NR5A2 (rs3762397) are associated with Peak bone mineral density (PBMD) both in Han and Uyghur subjects. PBMD, in Xinjiang, appears to be associated with several known factors that are well described in the literature. While the genotypes of CATSPERB and NR5A2 do not appear have a strong effect on the PBMD.
This article has carried out a comprehensive evolutionary genomics study of CatSper and CatSperβ proteins that constitutes the Ca2+ channel complex critical for sperm Ca2+ hyperactivation in mammals. With continued advances in genome biology, the evolutionary genomics approaches undertaken here will further illuminate the lineage-specific distribution of Ca2+ channels and transporters at the genomic scale and greatly facilitate deciphering the Ca2+ signaling codes in a species- and/or tissue-specific manner.
CATSPERB Preparation Options
We provide custom membrane protein preparation services for worldwide customers. Leveraging by our advanced Magic™ membrane protein production platform, we are able to present target membrane protein in multiple active formats. Our professional scientists are happy to help you find an ideal method and make your project a success. Aided by our versatile Magic™ anti-membrane protein antibody discovery platform, we also provide customized anti-CATSPERB antibody development services.
Creative Biolabs provides high-quality membrane protein preparation service to facilitate the development of worldwide customer’s research. During the past years, we have successfully established a powerful Magic™ membrane protein platform which enables us to provide a series of membrane protein preparation services. For more detailed information, please feel free to contact us.
Reference
All listed services and products are For Research Use Only. Do Not use in any diagnostic or therapeutic applications.