Creative Biolabs now offers specialized membrane protein production based on baculovirus-insect cells. Aided by state-of-the-art technical platforms and vast expertise in the membrane protein production field, we are confident in tailoring our customers the most efficient and reliable service to accelerate their cutting-edge projects.
Baculovirus-insect recombinant protein expression system was first invented in 1983. Since then, thousands of recombinant proteins, including some difficult ones like GPCRs, ion channels, have been successfully produced in this expression system. This system is primarily based on the use of recombinant baculoviruses (insect viruses) and their ability to express biologically active proteins in cultured insect cells or insect larvae. These baculoviruses have very late genes (polyhedrin, P10), which are highly expressed driven by very strong promoters but are dispensable for virus formation. Deletion of P10 genes and replacement with other genes convert these viruses into expression vectors for the production of foreign proteins. The proteins expressed by this system can be easily purified using tags and affinity chromatography.
Briefly, the baculovirus system relies on the infection of insect cell lines (usually Sf9 or Sf21) by recombinant viruses encoding the target membrane proteins. Notably, Sf9, a clonal isolate of the Spodoptera frugiperda cell line IPLB-Sf21-AE, is the most widely used for baculovirus-based membrane protein production. Sf9 was originally established from ovarian tissue of the fall armyworm. The major feature of the recombinant baculovirus in vitro infection is that the naturally occurring polyhedrin gene within the wild-type baculovirus genome is replaced with a recombinant gene or cDNA, which is commonly under the control of polyhedrin and p10 promoters. In the late phase of in vitro infection, the virions are assembled and budded recombinant virions are released. However, during the very late phase of infection, the inserted heterologous genes are placed under the transcriptional control of the strong AcNPV polyhedrin promoter. Thus, recombinant membrane proteins are expressed in place of the naturally occurring polyhedrin protein. Usually, the recombinant proteins are processed, modified, and targeted to the appropriate cellular locations.
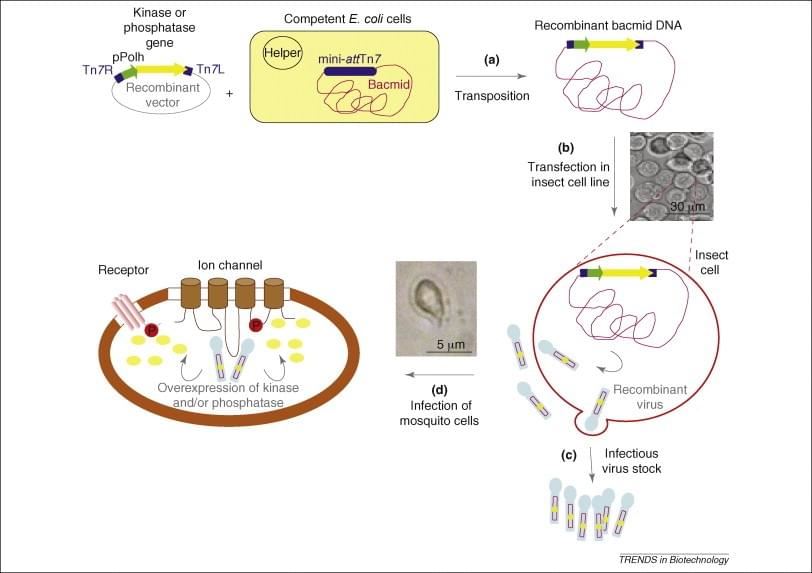 Figure 1. Heterologous protein expression in insect cells.
Figure 1. Heterologous protein expression in insect cells.
This system is well compatible to expressing many eukaryotic proteins with efficient folding and post-translational modifications (e.g. glycosylation). In contrast to other eukaryotic machinery, insect cell-based expression represents a promising alternative with lower cost and higher yield.
The advantages of our Magic™ protein expression in baculovirus-insect cells including:
Creative Biolabs offers professional membrane protein expression services to help our global clients with their cutting-edge projects. We also provide other cell-based membrane protein expression systems, including:
With our versatile platform, our guaranteed service can deliver the most satisfactory membrane protein products for antibody development, structural analysis, assays, etc. Please feel free to inquire us for more details.
References
All listed services and products are For Research Use Only. Do Not use in any diagnostic or therapeutic applications.