SLC13A5 is also known as solute carrier family 13 member 5, Na⁺/citrate cotransporter, NaCT, sodium-coupled citrate transporter, and Sodium-dependent citrate transporter. It is a member of the mammalian solute carrier family 13 (SLC13), which belongs to the ion transporter superfamily, comprising 16 transporter families, with over 32,000 members identified so far. SLC13A5 possesses twelve transmembrane helices and two helix hairpins. It consists of two inverted 5-TMH repeats connected by a long cytoplasmic loop plus an additional N-terminal helix.
| Basic Information of SLC13A5 | |
| Protein Name | Solute carrier family 13 member 5 |
| Gene Name | SLC13A5 |
| Aliases | Na+/citrate cotransporter, NaCT, Sodium-coupled citrate transporter, Sodium-dependent citrate transporter |
| Organism | Homo sapiens (Human) |
| UniProt ID | Q86YT5 |
| Transmembrane Times | 12 |
| Length (aa) | 568 |
| Sequence | MASALSYVSKFKSFVILFVTPLLLLPLVILMPAKFVRCAYVIILMAIYWCTEVIPLAVTSLMPVLLFPLFQILDSRQVCVQYMKDTNMLFLGGLIVAVAVERWNLHKRIALRTLLWVGAKPARLMLGFMGVTALLSMWISNTATTAMMVPIVEAILQQMEATSAATEAGLELVDKGKAKELPGSQVIFEGPTLGQQEDQERKRLCKAMTLCICYAASIGGTATLTGTGPNVVLLGQMNELFPDSKDLVNFASWFAFAFPNMLVMLLFAWLWLQFVYMRFNFKKSWGCGLESKKNEKAALKVLQEEYRKLGPLSFAEINVLICFFLLVILWFSRDPGFMPGWLTVAWVEGETKYVSDATVAIFVATLLFIVPSQKPKFNFRSQTEEERKTPFYPPPLLDWKVTQEKVPWGIVLLLGGGFALAKGSEASGLSVWMGKQMEPLHAVPPAAITLILSLLVAVFTECTSNVATTTLFLPIFASMSRSIGLNPLYIMLPCTLSASFAFMLPVATPPNAIVFTYGHLKVADMVKTGVIMNIIGVFCVFLAVNTWGRAIFDLDHFPDWANVTHIET |
SLC13A5 is a member of the sodium dicarboxylate/sulfate cotransporter family. SLC13A5 recognizes and transports various dicarboxylate and tricarboxylate TCA intermediates with citrate as the predominant substrate. SLC13A5 is most abundantly expressed in the plasma membrane of hepatocytes and controls the uptake of citrate into hepatocytes from the bloodstream, where the citrate concentration (100–150 μm) is several fold greater than that of all other TCA intermediates combined, suggesting that SLC13A5 may play a key physiological role in facilitating the use of circulating citrate by the liver. At the same time, the human citrate transporter SLC13A5 plays a central role in fatty acid synthesis and glycolysis, and is a potential drug target against obesity and diabetes. SLC13A5 plays critical roles in fatty acid synthesis, cholesterol synthesis, glycolysis, and gluconeogenesis and may represent a novel therapeutic target for obesity and metabolic disorders.
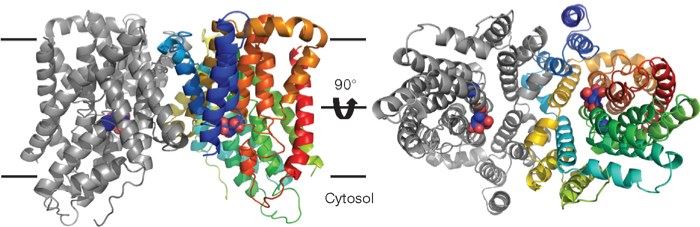 Fig.1 Structure of SLC13A5 membrane protein (Mancusso,2012).
Fig.1 Structure of SLC13A5 membrane protein (Mancusso,2012).
This article reports the structure and mechanism of a bacterial sodium-dependent dicarboxylate transporter, which is homologous fly gene of SLC13A5 and shares 26–33% sequence identity with the three human SLC13 transporters.
The results of this article expand the role of SLC13A5 from facilitating hepatic energy homeostasis to influencing hepatoma cell proliferation and suggest a potential role of SLC13A5 in the progression of human hepatocellular carcinoma.
The article demonstrates that SLC13A5 is involved in regulation of liver fat metabolism because siRNA-mediated SLC13A5 inhibition prevented neutral lipid storage and triglyceride accumulation in the liver and no effect on body weight.
The findings of this article reveal that SLC13A5 plays a potentially important role in the development and function of teeth and bone because SLC13A5 deficiency led to decreased bone mineral density and impaired bone formation in 13-week-old mice but not in older mice.
This article suggests that the inhibition of SLC13A5 may be an attractive therapeutic target to treat non-alcoholic fatty liver disease and type 2 diabetes because compound targeting SLC13A5 result in reducing hepatic glucose and lipid production.
To obtain the soluble and functional target protein, the versatile Magic™ membrane protein production platform in Creative Biolabs enables many flexible options, from which you can always find a better match for your particular project. Besides, aided by our versatile Magic™ anti-membrane protein antibody discovery platform, we also provide customized anti-SLC13A5 antibody development services.
Creative Biolabs is a leading custom service provider and has won good reputation among our worldwide customers in the field of membrane protein, we have accomplished numerous challenging projects including generation of many functional membrane proteins successfully. Please feel free to contact us for more information.
Reference
All listed services and products are For Research Use Only. Do Not use in any diagnostic or therapeutic applications.