Lymphoma Targeting Module Development Service
Accelerate Your Targeted Drug Delivery Research!
Are you currently facing challenges in developing targeted therapies for lymphoma, such as limited drug delivery efficiency and potential off-target effects? Creative Biolabs' Lymphoma Targeting Module Development service helps you enhance drug delivery and improve treatment efficacy through advanced targeting strategies.
Contact our team to get an inquiry now!
Overview
Lymphoma, a hematopoietic system cancer originating in lymphocytes, manifests diagnostic indicators such as indolent lymph node enlargement, febrile episodes, unintentional weight depletion, cutaneous pruritus, persistent asthenia, and nighttime diaphoresis. The disease encompasses a heterogeneous classification spectrum, chiefly stratified into Hodgkin lymphoma (HL) and non-Hodgkin lymphoma (NHL), with NHL comprising approximately nine-tenths of all confirmed cases. Rarer classifications encompass plasma cell dyscrasias and immune hyperproliferative disorders. These conditions fall within hematolymphoid neoplasms, pathobiologically aligned with leukemias. Contemporary management employs cytotoxic agents, ionizing radiation protocols, molecularly targeted biologics, and selective surgical resection.
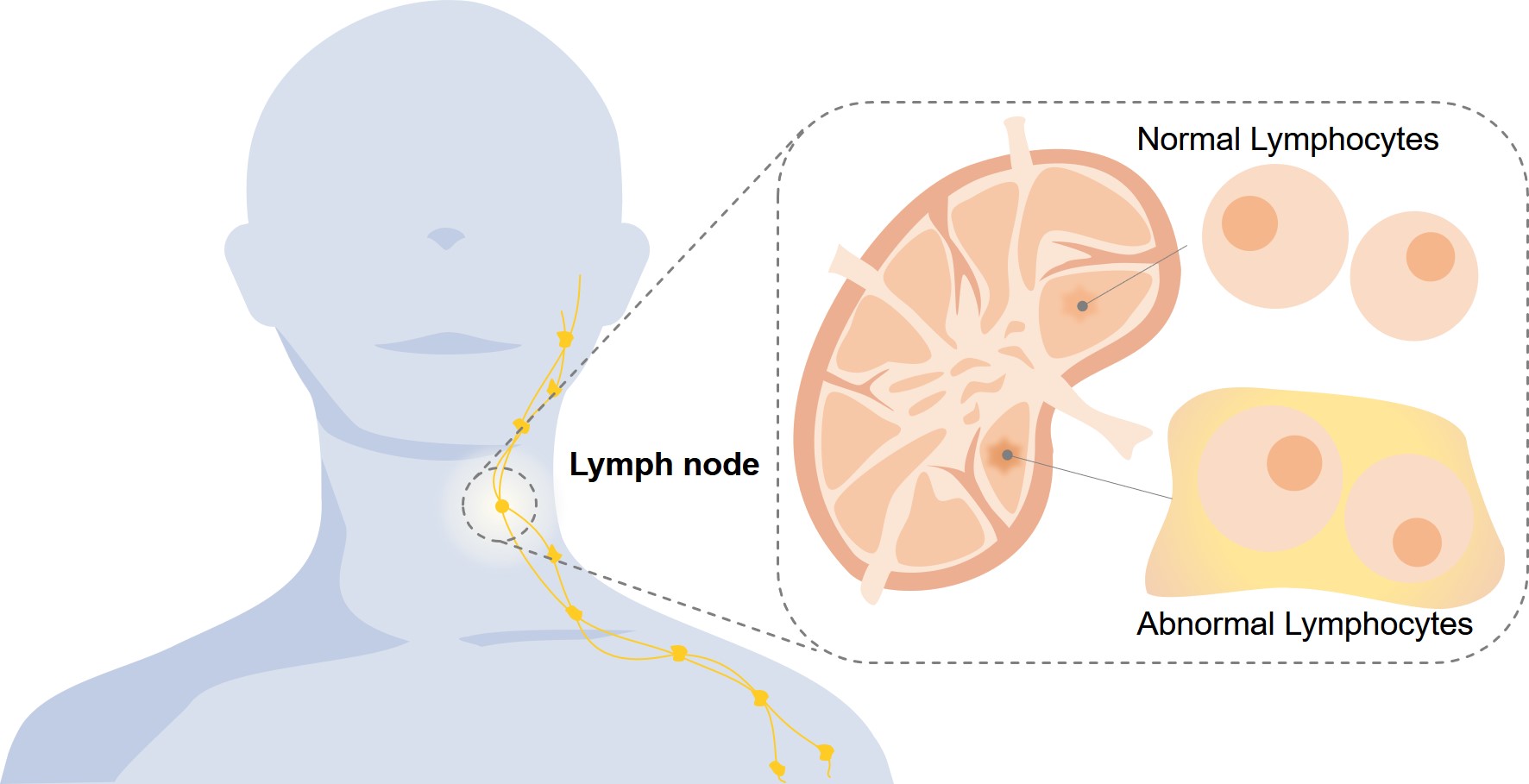 Fig.1 Lymphoma.
Fig.1 Lymphoma.
Delivery System Targeting Lymphoma
Contemporary lymphoma management increasingly incorporates advanced nanotherapeutic strategies. Folate receptor (FR)-targeted delivery platforms demonstrate particular promise, leveraging FR overexpression in malignant B-cell lineages for preferential cellular uptake. Researchers have engineered folate-conjugated lipid-polymer nanoparticles (LPNs) loaded with vincristine (VCR) to enhance chemotherapeutic precision. Comprehensive preclinical assessments—spanning cellular assays and murine xenograft models—validated these FR-directed LPNs for site-specific accumulation, achieving potent antitumor responses in lymphoma tissue while sparing healthy systems. Experimental outcomes highlighted significant tumor regression in vivo and markedly reduced off-target toxicity compared to conventional delivery methods. These findings position folate-functionalized LPNs as a paradigm-shifting approach for FR-dependent malignancies, merging high therapeutic precision with minimized systemic adverse effects.
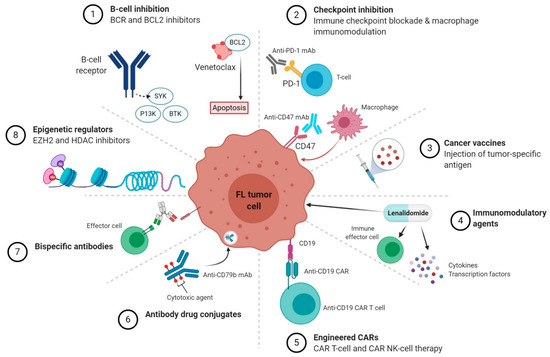 Fig 2. Novel targeted therapeutic classes in follicular lymphoma (FL).1,3
Fig 2. Novel targeted therapeutic classes in follicular lymphoma (FL).1,3
Recent pharmacological innovations highlight polyethylene glycol-functionalized NLG copolymers (PEG-Fmoc-NLG) as precision vehicles for doxorubicin (DOX) delivery in leukemia intervention. These nanocarriers self-assemble into monodisperse micellar structures (~120 nm diameter) with stable drug encapsulation. In vitro analyses using A20 leukemic cells revealed micellar DOX cytotoxicity comparable to free drug counterparts. In vivo evaluation in lymphoma models demonstrated superior tumor regression with PEG-Fmoc-NLG/DOX versus unformulated DOX. Parallel research leveraged platelet-based delivery for lymphoid malignancies, achieving exceptional drug payload retention. Platelet-vectored DOX significantly suppressed Raji cell proliferation, positioning thrombocytes as biomimetic carriers for targeted hematological oncology applications.
What We can Offer?
Creative Biolabs has a complete module delivery system and an experienced team of scientists. We can provide individual targeting modules or different types of module-payload/carrier complexes for specific lymphoma targets. We provide a wealth of corresponding products for you to choose from, including:
- Custom design and synthesis of lymphoma-targeting modules (aptamers, antibodies, peptides)
- Conjugation of targeting modules to therapeutic agents (drugs, genes, proteins)
- In vitro and in vivo evaluation of targeting module efficacy
- Optimization and scale-up of targeting module production
Experience the Creative Biolabs Advantage - Get a Quote Today
Why Choose Us?
Creative Biolabs offers a unique combination of expertise, technology, and customer focus that sets us apart in the field of targeted drug delivery. We are committed to providing our clients with the highest quality research services and innovative solutions to accelerate their drug discovery and development efforts.
- Core Competencies: Our multidisciplinary team includes specialists with deep domain knowledge in lymphoid oncogenesis, therapeutic vector engineering, and supramolecular chemistry. This expertise is demonstrated through clinically validated delivery vectors for diverse theranostic applications.
- Advanced Technology: We utilize state-of-the-art technologies and methodologies to design, synthesize, and evaluate targeting modules. This includes access to advanced chemical synthesis platforms, cell culture facilities, and animal models.
- Precision Biologics Development: Leveraging bespoke project frameworks, we co-develop precision biologics aligned with client-specific molecular targets and clinical translation roadmaps through iterative design-test-optimize cycles.
- Quality and Reliability: Stringent quality assurance protocols ensure batch-to-batch consistency, with GMP-grade manufacturing workflows and spatiotemporal biodistribution analytics guaranteeing clinical-grade reproducibility.
- Published Data: Our work is supported by extensive Published Data demonstrating the effectiveness of our targeting strategies.
Workflow
FAQs
What mechanisms confer cellular specificity in your ligand systems for lymphoid malignancies?
Our bioaffinity agents exploit disease-selective epitopes overexpressed on neoplastic B-cell membranes. Proprietary screening cascades combined with in silico-affinity maturation protocols ensure subnanomolar target avidity while achieving non-target tissue passivation.
Which bioactive payloads are compatible with your delivery platforms?
Our multivalent ligand platforms enable precision conjugation with diverse pharmacological compounds - spanning low-molecular-weight pharmaceuticals, biologics, gene therapeutics, and diagnostic contrast media.
How do you evaluate the efficacy of your targeting modules?
We employ a variety of in vitro and in vivo assays to evaluate the efficacy of our targeting modules, including cell binding assays, cytotoxicity assays, and animal studies. We can tailor our evaluation strategy to your specific requirements.
Published Data
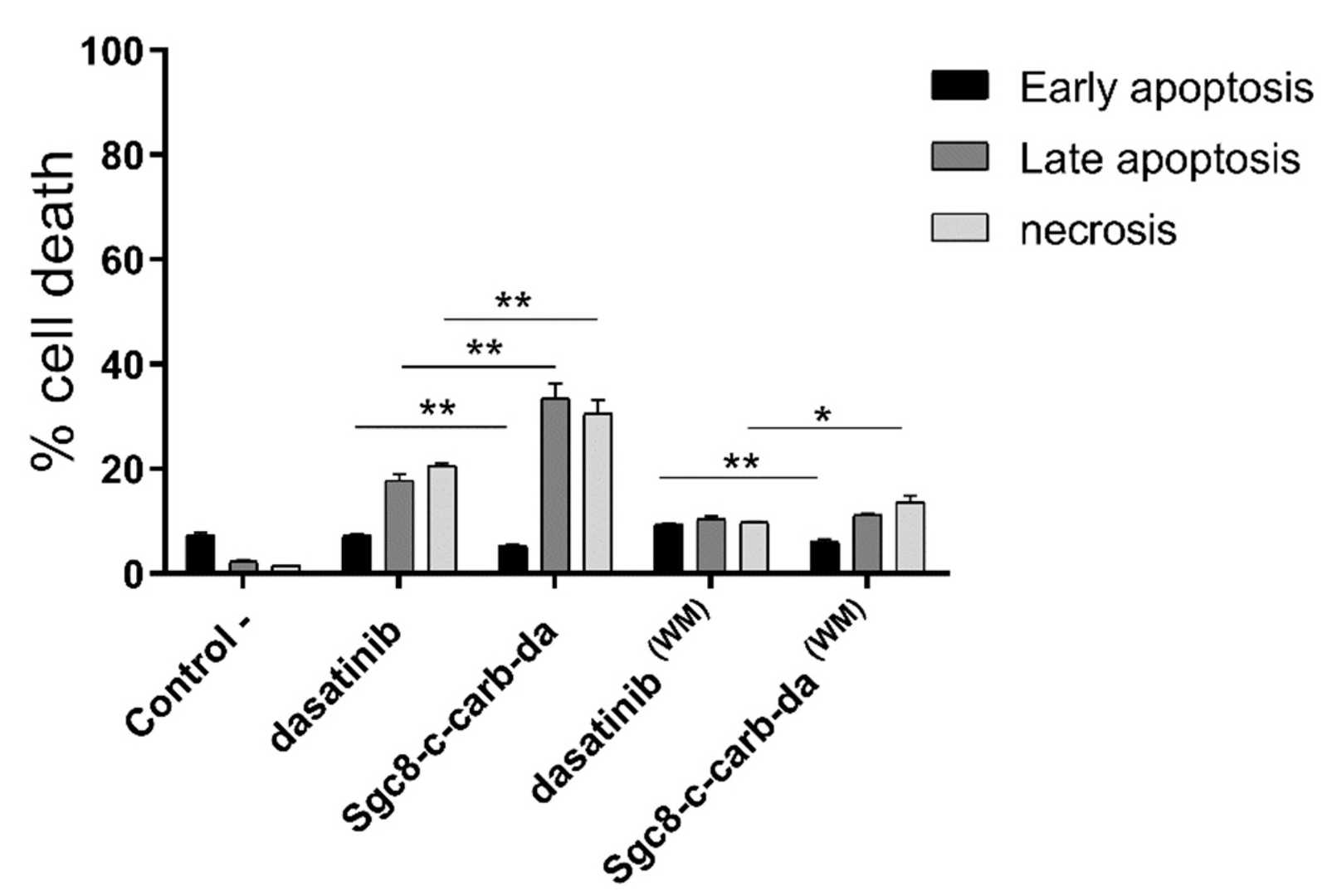 Fig 3. Percentage of cell death of A20 cells obtained after incubation with dasatinib or Sgc8-c-cab-da.2,3
Fig 3. Percentage of cell death of A20 cells obtained after incubation with dasatinib or Sgc8-c-cab-da.2,3
The study explores a targeted drug delivery system for lymphoma using the Sgc8-c aptamer. The researchers synthesized aptamer-drug conjugates (ADCs) by chemically linking Sgc8-c to dasatinib, a lymphoma drug. The experiment focused on evaluating the efficacy of this conjugate in inhibiting lymphoma cell growth. The results demonstrated that the Sgc8-c-dasatinib conjugate effectively and specifically inhibited lymphocyte growth and induced cell death in vitro. This finding suggests that the Sgc8-c aptamer can be used to target lymphoma cells and deliver therapeutic drugs.
Creative Biolabs maintains the life science sector's most comprehensive service array for lysosome targeting solutions. Please contact us for more information.
References
- Nath, Karthik, and Maher K. Gandhi. "Targeted treatment of follicular lymphoma." Journal of personalized medicine 11.2 (2021): 152.
- Sicco, Estefanía, et al. "Targeted-lymphoma drug delivery system based on the Sgc8-c aptamer." Cancers 15.3 (2023): 922.
- Distributed under Open Access license CC BY 4.0, without modification.



