Transmembrane protein 184A (TMEM184A) is a transmembrane protein composed of 413 amino acids with seven transmembrane domains. TMEM184A is encoded by the TMEM184A gene, which spans the 9.3 kb region on chromosome 5 and was originally identified in mouse cDNA library screen. Mouse TMEM184A has orthologs with many mammalian species, including humans, chimpanzees, dogs and rats, with homology among these homologs exceeding 80%. TMEM184A is predicted to have kinase activity; however, its amino acid sequence does not align to any known domains. Predictive analysis of the TMEM184A protein sequence reveals that the C-terminal region is rich in basic amino acids that can participate in heparin binding. In other regions of the TMEM184A protein, although there are no multiple basic residues in the transmembrane segment, it may also be involved in heparin binding.
| Basic Information of TMEM184A | |
| Protein Name | Transmembrane protein 184A |
| Gene Name | TMEM184A |
| Organism | Homo sapiens (Human) |
| UniProt ID | Q6ZMB5 |
| Transmembrane Times | 7 |
| Length (aa) | 413 |
| Sequence | MSNVSGILETAGVPLVSANWPQPSPPPAVPAGPQMDHMGNSSQGAPWLFLTSALARGVSGIFVWTALVLTCHQIYLHLRSYTVPQEQRYIIRLLLIVPIYAFDSWLSLLLLGDHQYYVYFDSVRDCYEAFVIYSFLSLCFQYLGGEGAIMAEIRGKPIKSSCLYGTCCLRGMTYSIGFLRFCKQATLQFCLVKPVMAVTTIILQAFGKYHDGDFNVRSGYLYVTLIYNASVSLALYALFLFYFTTRELLRPFQPVLKFLTIKAVIFLSFWQGLLLAILERCGVIPEVETSGGNKLGAGTLAAGYQNFIICVEMLFASVALRYAFPCQVYAEKKENSPAPPAPMQSISSGIRETVSPQDIVQDAIHNFSPAYQHYTQQATHEAPRPGTHPSGGSGGSRKSRSLEKRMLIPSEDL |
TMEM184A is present in vascular smooth muscle cells (VSMCs) and endothelial cells (ECs) and is recently identified as the heparin receptor in vascular ECs and VSMCs. In VSMCs, TMEM184A is required for potent anti-proliferative and anti-inflammatory signaling cascades induced by heparin. Heparin binds specifically to TMEM184A and decreases growth-factor-induced MAPK signaling and proliferation. In ECs, TMEM184A is required for heparin-induced expression of dual specificity phosphatase-1 (DUSP1) and subsequent downregulation of MAPK signaling. Heparin-binding results in elevated levels of DUSP1 protein and decreased ERK activity. Therefore, TMEM184A is a heparin receptor that is involved in heparin-induced signaling transduction and mediates the anti-inflammatory response of ECs and VSMCs. The response of VSMC and ECs to heparin includes cell proliferation, ERK pathway activity, and reduction in activation of specific transcription factors. In addition, Furthermore, expression of TMEM184A is specifically upregulated during mouse development and in the Sertoli cells of the testis. Thus, it may mediate signaling events between germ cells and Sertoli cells or between other somatic and Sertoli cells, or mediate regulatory pathways that control the compartmentalization of testicular structures.
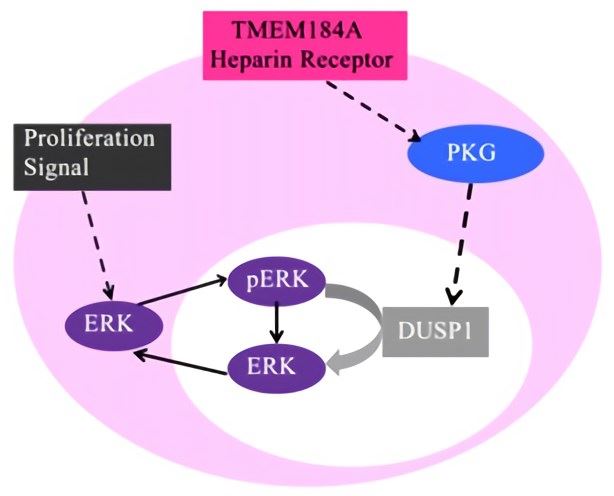 Fig.1 Signaling pathway for heparin receptors. Heparin interacts with TMEM184A and leads to an increase in DUSP1 and a decrease in ERK activation. (Pugh, 2016)
Fig.1 Signaling pathway for heparin receptors. Heparin interacts with TMEM184A and leads to an increase in DUSP1 and a decrease in ERK activation. (Pugh, 2016)
The study demonstrates that Tmem184a is a novel angiogenesis regulator using the adult zebrafish regenerating caudal fin as an in vivo model.
This report presents a series of experimental protocols based on GFP-TMEM184A constructs expressed in vascular cells that can be easily adapted to other new proteins.
The data from this study support the discovery that TMEM184A is a heparin receptor in vascular cells.
The results of this study suggest that Tmem184a is expressed only in the Sertoli cells of the developing testicular cord, which mediates gender-specific signals during Sertoli cell differentiation.
This article demonstrates that TMEM184A acts as a heparin receptor and mediates anti-inflammatory responses in endothelial cells, including reduced JNK and p38 activity.
Based on our versatile Magic™ membrane protein production platform, we could offer a series of membrane protein preparation services for worldwide customers in reconstitution forms and multiple active formats. Aided by our versatile Magic™ anti-membrane protein antibody discovery platform, we also provide customized anti-TMEM184A antibody development services.
In the past years, Creative Biolabs has successfully generated many functional membrane proteins for our global customers. We are happy to accelerate the development of our clients’ programs with our one-stop, custom-oriented service. For more detailed information, please feel free to contact us.
Reference
All listed services and products are For Research Use Only. Do Not use in any diagnostic or therapeutic applications.