Introduction of ADRB3
ADRB3 is a beta-adrenergic receptor, which belongs to the G protein-coupled receptor (GPCR) superfamily characterized by seven transmembrane domains of 22-28 amino acids. Structurally, the N-terminus of ADRB3 protein is extracellular and glycosylated. The C-terminus is intracellular and has no sites for phosphorylation by protein kinase A (PKA) and β-adrenoceptor kinase (βARK). TM3, TM4, TM5, and TM6 are essential for ligand binding. TM2 and TM7 are involved in Gs activation. The activation of this receptor produces several signaling pathways, including activation of adenylyl cyclase and cAMP-dependent phosphorylation. ADRB3 is insensitive to the commonly used beta-adrenergic receptor antagonists.
| Basic Information of ADRB3 | |
| Protein Name | Beta-3 adrenergic receptor |
| Gene Name | ADRB3 |
| Aliases | Beta-3 adrenoreceptor |
| Organism | Homo sapiens (Human) |
| UniProt ID | P13945 |
| Transmembrane Times | 7 |
| Length (aa) | 408 |
| Sequence |
MAPWPHENSSLAPWPDLPTLAPNTANTSGLPGVPWEAALAGALLALAVLATVGGNLLVIV AIAWTPRLQTMTNVFVTSLAAADLVMGLLVVPPAATLALTGHWPLGATGCELWTSVDVLC VTASIETLCALAVDRYLAVTNPLRYGALVTKRCARTAVVLVWVVSAAVSFAPIMSQWWRV GADAEAQRCHSNPRCCAFASNMPYVLLSSSVSFYLPLLVMLFVYARVFVVATRQLRLLRG ELGRFPPEESPPAPSRSLAPAPVGTCAPPEGVPACGRRPARLLPLREHRALCTLGLIMGT FTLCWLPFFLANVLRALGGPSLVPGPAFLALNWLGYANSAFNPLIYCRSPDFRSAFRRLL CRCGRRLPPEPCAAARPALFPSGVPAARSSPAQPRLCQRLDGASWGVS |
Functions of ADRB3 Membrane Protein
ADRB3 receptors are identified in a variety of tissues and mediate various physiological effects. They inhibit the contractile activity of ileum and colon, induce relaxation of airway smooth muscle, produce peripheral vasodilation, reduce the contractile force in human ventricular muscle, and stimulate L-type calcium current in human atrial myocytes. More importantly, it is discovered to play an important role in energy metabolism through its role in enhancing lipolysis, mobilization of free fatty acids into the circulation in white adipose tissue, and thermogenesis in brown adipose tissue. Polymorphisms of the ADRB3 gene has been reported to be associated with resistance to weight loss. Studies have reported ADRB3 as a potential target for anti-obesity and anti-diabetes therapy.
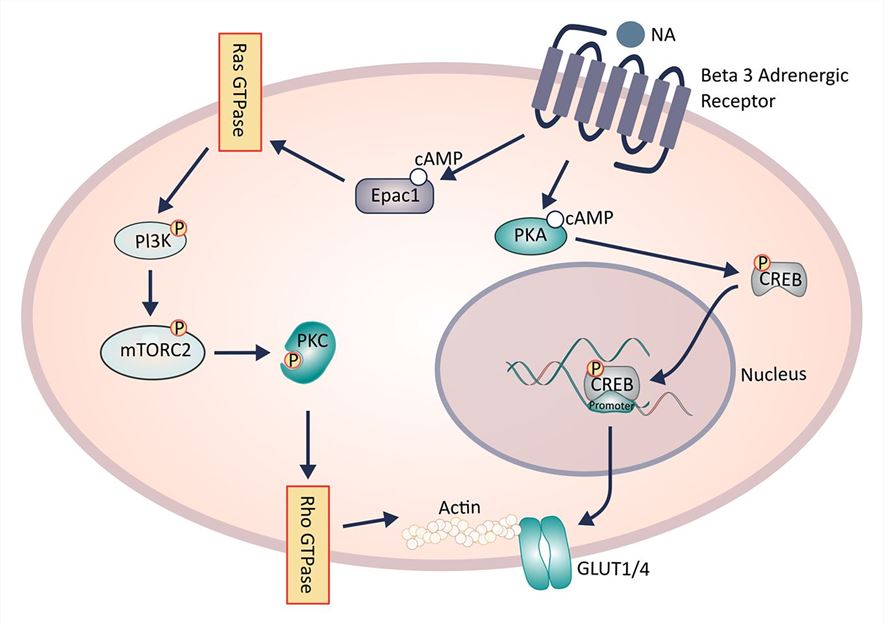 Fig.1 Intracellular signaling cascade governing ADRB3 stimulated glucose uptake by brown adipocytes. (Hankir, 2016)
Fig.1 Intracellular signaling cascade governing ADRB3 stimulated glucose uptake by brown adipocytes. (Hankir, 2016)
Applications of ADRB3 Membrane Protein in Literature
1. Jin J., et al. Design and synthesis of aryloxypropanolamine as β3-adrenergic receptor antagonist in cancer and lipolysis. European Journal of Medicinal Chemistry. 2018, 150: 757-770. PMID:29574204
This article described the design, synthesis, and evaluation of a series of novel L-748,337 derivatives as selective human β3-adrenergic receptor antagonists. The most potent one was found to alleviate weight loss and inhibit tumor growth.
2. Procino G., et al. β3 adrenergic receptor in the kidney may be a new player in sympathetic regulation of renal function. Kidney International. 2016, 90(3): 555-567. PMID: 27206969
This article investigated the expression and the possible physiologic role of β3-adrenergic receptor (β3-AR) in mouse kidney. The findings uncover the antidiuretic effect of β3-AR stimulation in the kidney and suggest that β3-AR agonism might be useful to bypass AVPR2-inactivating mutations.
3. Hong S., et al. Phosphorylation of Beta-3 adrenergic receptor at serine 247 by ERK MAP kinase drives lipolysis in obese adipocytes. Mol Metab. 2018, 12: 25-38. PMID: 29661693
This article demonstrated that ERK activation in adipocytes and subsequent phosphorylation of the β3 adrenergic receptor on Serine 247 are critical regulatory steps in the enhanced adipocyte lipolysis of obesity.
4.Zhang R., et al. Effects of carvedilol on ventricular remodeling and the expression of β3-adrenergic receptor in a diabetic rat model subjected myocardial infarction. International Journal of Cardiology. 2016, 222: 178-184. PMID: 27497092
This article assessed effects of carvedilol on ventricular remodeling and expression of β3-adrenergic receptor (β3-AR) and Gi protein in a rat model of diabetes subjected to myocardial infarction. The results showed that the expression of this receptor and its coupled Gi protein was increased in this model and Carvedilol treatment prevented many of these harmful effects.
5. Cao X., Li Y. β3-Adrenergic Receptor Regulates Hepatic Apolipoprotein A-I Gene Expression. Journal of Clinical Lipidology. 2017, 11(5): 1168-1176. PMID: 28802864
This article investigated the molecular mechanism of β3-AR-mediated regulation of hepatic apoA-I gene expression. The results showed that β3-AR-mediated upregulation of hepatic apoA-I expression might be attributed to the increased activity of the apoA-I promoter, HNF-4, and HNF-3. Thus, β3-AR might exert an anti-atherosclerotic effect.
ADRB3 Preparation Options
Our technical platform has extensive experience in reconstituting membrane proteins, including GPCRs, ion channels, transporters, into functional and active formats for subsequent structural and functional studies. Available preparation options include detergent micelles, liposomes, nanodiscs, amphipols, and SMALPs. Aided by our versatile Magic™ anti-membrane protein antibody discovery platform, we also provide customized anti-ADRB3 antibody development services.
With the successful accomplishment of various difficult-to-prepare membrane proteins, Creative Biolabs is proud to introduce our all-around, custom-oriented services of Magic™ membrane protein production to our clients across the globe. We are happy to share our advanced technologies and extensive experience to promote your brilliant studies. Please feel free to contact us for more information.
Reference
All listed services and products are For Research Use Only. Do Not use in any diagnostic or therapeutic applications.