Formulation Optimization Services
Vaccines are formulated to stimulate a protective immune response, while minimizing the potential for negative side-effects on exposure to the recipient. Vaccine formulation is complex and should be tightly regulated to maximize safety. Creative Biolabs is expert in formulation development. Our formulation optimization strategies can provide you with the best advice and accelerate your vaccine development program.
Stable Liquid Formulation
The stability of vaccine components in solution is a direct function of several environmental conditions that ultimately contribute to the measurable physicochemical properties of the critical biomolecules. Most significant are solution pH, ionic strength, redox potential, and temperature, all of which often contribute directly to the stability of macromolecules. In addition, more pharmaceutical variables such as agitation and freeze/thaw stress, as well as surface activity, may all play key roles in formulation considerations.
- Solution pH contributes to both the chemical and physical stability of biomolecules. pH can affect the rate of enzymatic activity of live and attenuated, bacterial or viral vaccines, such as the intrinsic endonuclease activity that was a serious concern with the oral polio vaccine.
- The ionic strength of solutions can dramatically affect the solubility of biomolecules. Ionic strength can also shift the osmotic balance and promote membrane lysis of enveloped viruses and whole-cell bacterial vaccines. Ionic strength may also contribute favorably to the stability of vaccines by maintaining the overall repulsive forces between molecules, inhibiting aggregation and precipitation.
- The effects of temperature on the stability of vaccines can be disruptive at both extremes. As harmful as increased temperature can be to all but a select few classes of biological molecules that are used in vaccines, freezing may also be destructive as seen in the case of diphtheria, pertussis, tetanus, and polio vaccines.
Lyophilised Formulation
The properties of high stability, easy to storage and transport make lyophilization a suitable alternative to liquid formulations, especially for highly thermolabile products and live virus vaccine products. The lyophilization process involves freezing and then performing primary and secondary drying under vacuum. The drying process itself presents a range of additional challenges. In order to solve these problems, some specific excipients must be added, such as salts and buffer, pH, lyoprotectants and bulking agents.
Adjuvant Optimization
An important component of many vaccines such as those employing recombinant proteins is the adjuvant. Adjuvants are used to increase the effectiveness of vaccines by enhancing the immune response to the antigenic components.
As a leading service provider, Creative Biolabs offers a full range of adjuvants, including:
Table 1. Classification of Adjuvants.
| Type | Adjuvant | Advantages |
| Inorganic compounds | alum, aluminum hydroxide, aluminum phosphate, calcium phosphate hydroxide |
safe well sourced |
| Oil Adjuvant | Mineral oil (paraffin oil), Food Based oil (Adjuvant 65) |
safe potent immune responses for delivery of poorly water-soluble drugs |
| Liposomes | Cationic lipids |
can deliver various antigens can co-deliver various immune potentiators |
| Bacterial products | killed bacteria Bordetella pertussis, Mycobacterium bovis, toxoids | potent immunostimulatory capacity |
| Immunostimulatory Compexes (ISCOMS) |
detergents (Quil A) IL-1 AND-2 |
composed of two veterinary products. Scalable manufacturing process in place |
Creative Biolabs also provides PRR Ligands Scanning and Custom Adjuvant Synthesis services for vaccine adjuvantation to meet different customer's demand.
Delivery System Optimization
To obtain high potency of a vaccine, the adjuvant, and the antigen should be able to present at the same site. However, this is not always the case at the early stage of vaccine development. Several formulations and delivery systems have been developed to solve this problem, including nanoparticle, liposome, immune-stimulating complexes (ISCOMs), emulsion and microspheres. A successful delivery system should have the following properties:
- protect antigen from degradation
- sustain release of antigen
- intracellular delivery of antigen contributing to cytotoxic T-cell stimulation and targeting at APCs
Adjuvants can improve the effectiveness of vaccines by promoting the immune response with a longer duration, increase the low response rate of some individuals and reduce the cost of vaccination programs by reducing the quantity of antigen needed. Delivery systems concentrate the adjuvant and antigens in repetitive patterns, target vaccine antigens to APCs and help co-localize antigens and immunopotentiators. Together, adjuvants and delivery systems can enhance body-specific and active immune reaction.
Other Excipients Optimization
Vaccines are inherently unstable and susceptible to degradation by several physical and chemical degradation mechanisms. Therefore, various excipients are required during processing and storage to stabilize biological agents and vaccines. Excipients are an integral part of vaccine candidates and play an essential role in the formulation development of vaccines. New therapies and powerful pharmaceutical products can be developed by selecting and using the right excipients.
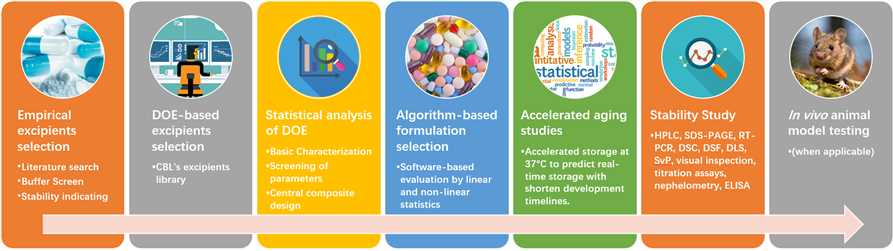
Formulation technology platform of Creative Biolabs is based on a library of up to hundreds of different, regulatory well-known excipients. Our scientists have a comprehensive understanding of vaccine excipients and are sure to find the best solution for your vaccine formulation. We have established a comprehensive empirical and design of experiments (DoE)-based formulation platform which can help you explore the best formulations with high success rate and short timeline. Our platform could minimize time, energy and supplies while maximizing product performance.
In terms of the extensive experience in formulation optimization, Creative Biolabs is proud to offer our clients formulation development services with the most competitive price and best quality.
All of our products can only be used for research purposes. These vaccine ingredients CANNOT be used directly on humans or animals.


