Mammalian Cell Expression System Evaluation Services for Vaccine Production
Creative Biolabs is an industry leader in vaccine development with a dedicated research team and more than a decade of experience. We are familiar with and proficient in all aspects of vaccine development, especially in optimizing the expression system and improving the process. We are convinced that our expertise can help you solve the problems you face in your research and remove obstacles to your smooth research.
Mammalian Expression System
The use of mammalian cells to produce vaccines is a long-standing method. The advent of recombinant DNA technology has made researchers aware that these technologies can be applied to mammalian cells and turned into reliable and safe vaccine production tools. The advantage of mammalian cell lines for the production of recombinant subunit vaccines is their ability to produce secreted proteins as protective antigens in serum-free media at high levels and robustly, and the ability to introduce correct protein folding, post-translational modifications and product assembly into the produced proteins. The complete biological activity of the protein makes it an effective vaccine candidate. A successful example of the production of vaccines using mammalian cell lines is recombinant hepatitis B vaccine.
Design of Basic Expression Unit
Initiating the transcription process requires that the nucleic acid sequence encoding the protein of interest be linked to a promoter that controls expression, typically from a highly expressed viral or cellular gene. A promoter is a DNA fragment located upstream of the transcription start site that binds to other transcription factors and factors that recruit coactivators to remove core histones, allowing RNA polymerase to bind to the DNA template to start the transcription process. Commonly used virus-derived promoters are SV40, RSV and human cytomegalovirus (CMV) immediate early promoters. Appropriate polyadenylation signals are equally important for efficient expression of genes. Efficient translation also requires that the initiation codon of the gene of interest be embedded in the sequence that conforms to the Kozak rules. Most of the genes have a start codon of AUG, so the design of the expression vector should be considered to avoid the upstream sequence of AUGs and adjacent hairpin structures, their presence may lead to a decline in protein production.
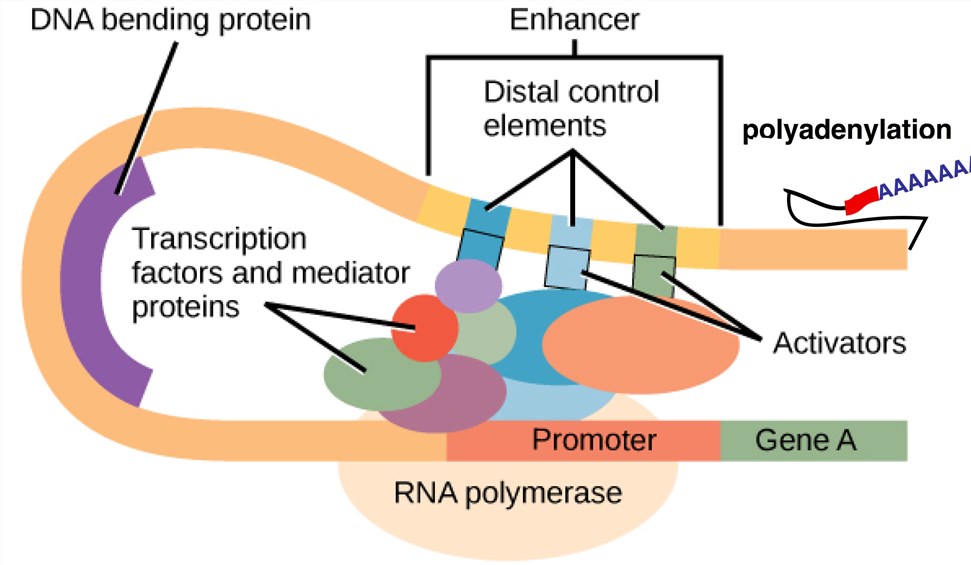 Fig. 1 Expression unit. Distributed under CC BY 4.0, from Wikimedia, only use the top half of the image.
Fig. 1 Expression unit. Distributed under CC BY 4.0, from Wikimedia, only use the top half of the image.
Services
- Protective antigen prediction and screening
- Design of expressing vector
- Condon optimization and gene synthesis
- Construction of expressing vector
Transient Expression
Establishing a recombinant mammalian cell line typically takes months or even a year, and the steps are cumbersome and labor intensive. In the early stage of vaccine development, when conducting animal experiments and preparing detection antigens for evaluating the immunogenicity of the candidates, it is often required to produce a certain amount of protein in a short period of time. For rapid production of a small amount of recombinant protein, transient transfection of mammalian cell lines can meet this requirement. Using appropriate transfection techniques, it is possible to induce receptor cells of almost all cell lines to take up foreign DNA to express the corresponding protein. Commonly used transfection techniques include the traditional calcium phosphate co-precipitation technique, polyethylenimine, dendrimers and the like. When a foreign gene enters a recipient cell, it is transported to the nucleus and maintained in the nucleus for several days. Usually, it does not have a replication and nuclear retention signal, so it is rapidly lost and causes a decrease in expression. In this case, a virus-based carrier (SV40, BPV or EBV) is often used to extend the life of such systems.
Stable Expression
Unlike transient transfection, stable cell lines are constructed for the industrial production of recombinant protein vaccine. Stable cell lines require the ability to produce the same quality products at different times, at different sites, and between different batches. After selecting the cell line for production, it is necessary to establish a mater cell bank. After resuscitation from the main cell bank, the working cell bank is expanded and the working cell bank is used for production. Cell banks are usually kept in liquid nitrogen and need to maintain the life cycle of the entire product. Host cells and expression vectors are critical in the construction of cell lines for production. CHO and myeloma cells are the two most commonly used host cells for the construction of stable cell lines. The vaccines produced by essentially both types of cells are secreted extracellularly so that the product can be harvested from the cell culture medium.
| Stable expression | Transient expression |
|---|---|
| Foreign DNA is integrated into the genome of host and expression could maintain throughout several generations | Expression of extraneous DNA is limited to 16-96 h, and traits could not be transported to the progeny |
| Extensive period to develop cell line | Quantity of plasmid DNA for transfection is large |
| High expression level | Relatively low expression level |
| Used for large scale manufacturing | Used for pre-clinical evaluation |
| High cost | Low cost |
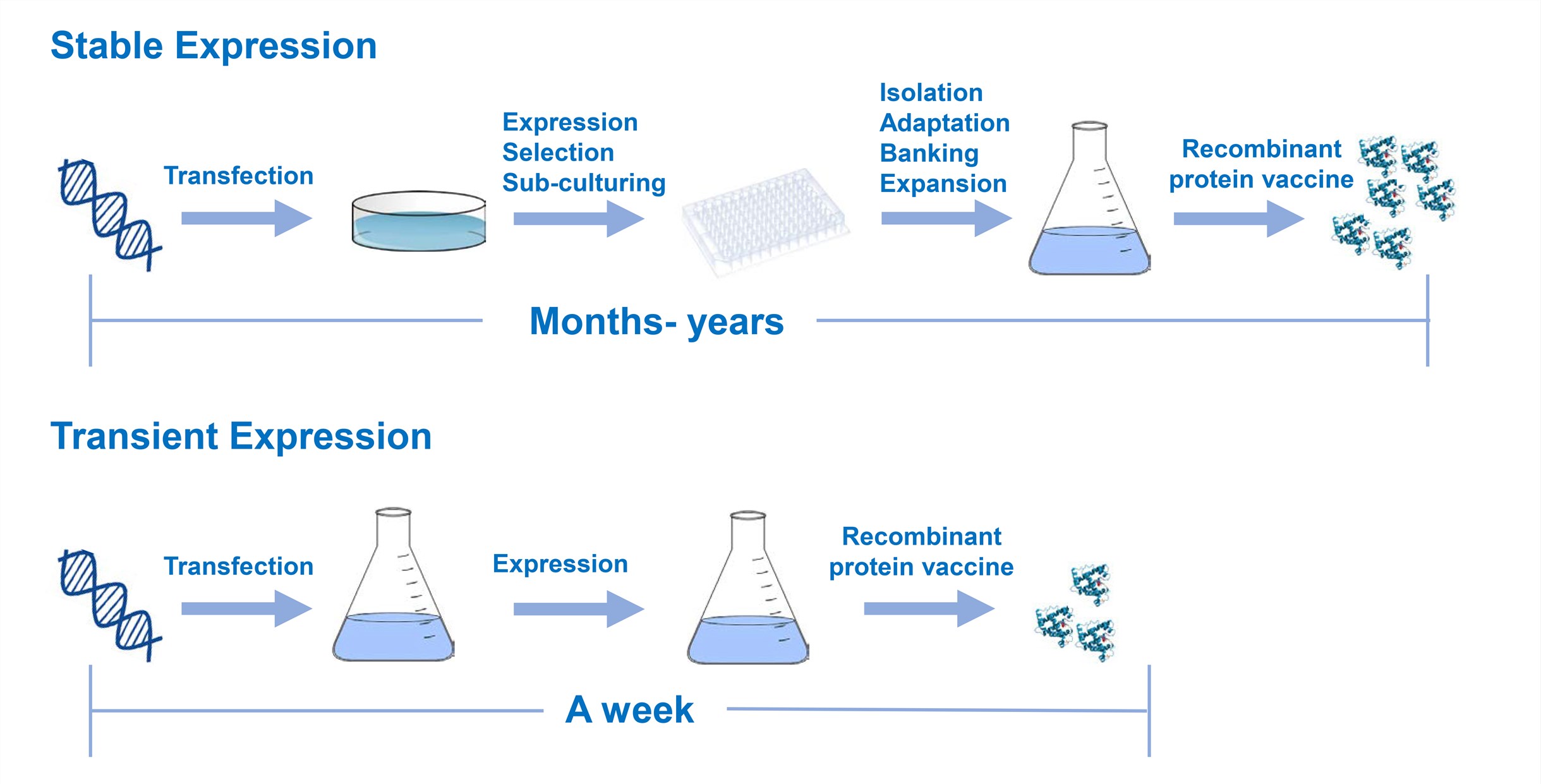
Fig.3 Comparison of transient and stable expression.
CHOL-NSO Expression System
The CHOL-NSO is a unique mammalian cell expression system developed by Creative Biolabs, the featured system is based on NS0 (non-secreting null) murine myeloma line. Utilizing cholesterol synthesis defects, a stable cell line is constructed and thus makes the production of eukaryotic proteins simpler and more efficient along with ensured protein quality and reduced production cycle and cost.
Our Platforms of Mammalian Expression System
- Mature transient and stable expression system
- Flexible and efficient transfect strategies
- Multiple mammalian cell lines for vaccine expressing
- Well-established mammalian cell-based fermentation process
- Specialized improvement for product quality and process control
Creative Biolabs is a professional vaccine development expert with extensive experience in vaccine design, immunogenicity evaluation, safety evaluation, and production process optimization. With a variety of mature and comprehensive platforms and technologies for the evaluation and improvement of vaccine expression systems especially in mammalian expression system, we can meet the various requirements of our customers and make your vaccine development path unimpeded.
All of our products can only be used for research purposes. These vaccine ingredients CANNOT be used directly on humans or animals.


