Cancer Vaccine related Services
As an excellent supplier in vaccine field, Creative Biolabs provides cancer vaccine related services and products according to customer's detailed requirements.
Vaccines were originally developed as a prophylactic agent, administered to healthy individuals to induce long-term immunity against a pathogen and to prevent the outbreak of viral diseases. Therapeutic vaccines aim at inducing strong antigen-specific T cell responses. In contrast to prophylactic vaccines, they are used in patients who already have a growing, established tumor. In the field of cancer immunotherapy, prophylactic vaccination only applies to the few virally induced malignancies, such as the vaccine against hepatitis B virus that can cause liver cancer, or the nowadays widespread human papilloma virus (HPV) vaccination of teenagers, which aims to prevent genital cancers induced by high-risk HPV strains. Unfortunately, however, most cancers are of nonvital origin, instead resulting from a succession of inherited and somatic DNA mutations leading to malignant cell transformation. Most of the time when the cancer is diagnosed, it is already well established and has largely evaded the control of the immune system. In this context, therapeutic cancer vaccines have been developed to stimulate or boost the immune system of the patient against established tumors.
The main components of cancer vaccines are (a) a tumor antigen, (b) an immunological adjuvant, and (c) a vehicle or carrier. Cancer vaccine success depends on a number of factors, including choosing the appropriate target antigen, formulation optimization, adjuvant, and delivery route. Each of these components must be selected from a variety of choices, which makes the process of vaccine optimization a complex problem.
The Antigen Formulation
Most cancer vaccines include well-defined tumor antigens, which can be administered in various forms, such as DNA or RNA, full protein or protein derived synthetic peptides of various lengths. Although numerous clinical trials have reported the generation of detectable and sometimes large T cell responses, very few of them could demonstrate objective clinical responses, and the immune responses observed were often short-lived. Whole tumor cell lysates are also very promising, as they overcome important limitations of a single antigen vaccine. Indeed, they contain multiple undefined antigens expressed by a patient’s tumor tissue, which may simultaneously induce a cytotoxic CD8+ T cell response as well as CD4+ T helper response, which should help maintain long-term tumor immunity. The multiple antigens present in the lysate would, in principle, reduce the risk of tumor-immune escape resulting from the downregulation or mutation of a single targeted antigen, which may occur during cancer progression or through the selective pressure of immunotherapy. The immunogenicity of a tumor lysate may also be higher than that of a synthetic antigen, as a result of posttranslational modification of proteins occurring within the tumors (oxidation, acetylation) which might be further increased by specific chemical treatments of the tumor lysates.
More recently, there has been a resurgence in interest regarding defined antigen vaccines, as bioinformatic tools and modern in vitro screening technologies now allow their validation as good binders to HLA molecules. A very promising approach has involved the elution, purification, and sequencing by mass spectrometry of the peptides from the tumor cell surface, which are by definition processed and presented by major histocompatibility complex (MHC) molecules of a patient’s tumor. They represent the effective immunopeptidome of a given tumor and provide a personalized vaccine containing a mixture of peptides, some of which might be potent tumor neoepitopes.
The Choice of the Adjuvant
The initial aim of formulating vaccines in adjuvants was to deliver the antigen in a poorly metabolizing and slowly degrading substance. The intention was to favor the slow and sustained release of the antigen to be captured by antigen-presenting cells (APCs) and be subsequently presented to T cells. Aluminum salts are widely used to favor T helper cell 2 (Th2)-mediated humoral immunity, but they are less efficient for promoting Th1-dependent immunity. To this aim, water-in-oil adjuvants have been developed to create a depot of the antigen at the site of the injection.
The next generation of vaccine delivery agents includes nanoparticles such as silica or liposomes or synthetic polymers, which are ideal vehicles to be taken up by dendritic cells (DCs) patrolling within the subcutaneous tissues. However, the challenge with such supports is to selectively promote DC uptake while eluding the systemic reticuloendothelial network of macrophages, which routinely clear circulating particles. In addition to these substances designed to favor delivery of the antigen to APCs, today’s therapeutic vaccines also contain another class of adjuvants aimed to deliver danger signals to activate the immune system, as antigen alone may fail to prime effective T cell responses or even induce tolerance.
The Choice of Delivery Systems
The choice of delivery systems and route of immunization depends on the end use of the vaccine. For practical reasons and minimal side effects, most prophylactic vaccines are administered via the skin, usually by subcutaneous injections in the epidermis or the dermis. These two locations are ideal, as they are enriched respectively in Langerhans DCs and dermal DCs, both cell populations being very efficient in capturing and processing antigens. The oral route is also very convenient and is used by vaccines against polio, typhoid fever, cholera, and rotavirus. The oral route is, however, more challenging in view of the extreme conditions in the gastrointestinal tract, including the low pH in the stomach and the presence of microbiota, which may degrade the antigen before it reaches the lymphoid organs. Moreover, the usually tolerogenic gut environment may not be ideal to generate a strong systemic immune response.
With regard to therapeutic vaccines used to treat chronic noncontagious diseases such as cancer, atopy, or diabetes, both immediate cellular effector responses and long-term immunity are desired to guarantee the continuous immunosurveillance of the disease. Although prophylactic vaccines for global immunization programs must be simple, inexpensive, and given via a noninvasive route, therapeutic cancer vaccines can benefit from more complicated technologies and use more invasive routes of delivery if beneficial for the patient. There is a very large array of cancer vaccines under development which use various delivery systems, and which are being tested in clinical trials. Other delivery routes tested in therapeutic cancer vaccines range from subcutaneous and intradermal to more invasive intraperitoneal and intranodal injections, to optimize antigen uptake by APCs and favor a local potent immune response. For instance, particulate therapeutic vaccines such as virosomes or nanoparticles can be injected in LNs using an ultrasound-guided imaging procedure. Although most of these strategies are still in the development stage, the potential to achieve strong and long-lasting antitumor responses is high, owing to new delivery systems and better understanding of T cell memory development.
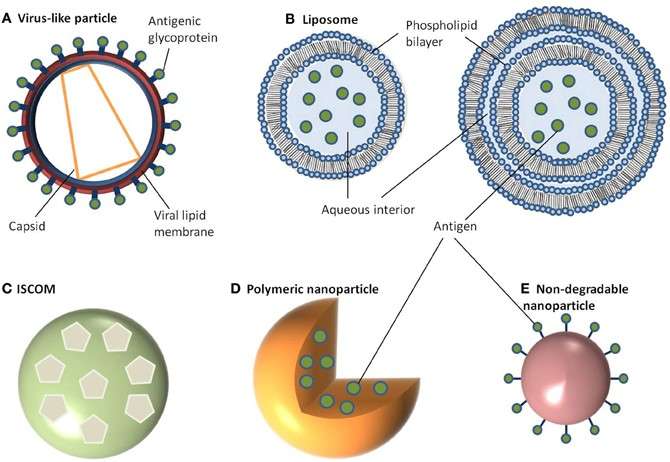
Fig. 3 Vaccine delivery systems. (Gregory, 2013)
Preclinical and clinical studies have clearly demonstrated that tumors are immunogenic. Vaccination could be used as a stand-alone therapy or in combination with other forms of traditional chemotherapy or radiotherapy, or even other immunotherapy strategies.
Cancer Vaccines
Creative Biolabs is a leader in the field of cancer vaccines. We provide development services for different types of cancer vaccines, including:
Cancer Vaccine Development Technology Platforms
Cancer Cell-Surface Targets Development for Vaccine Construction
- PSMA Vaccine
- CA125 Vaccines
- Other cancer cell-surface targets
Reference
- Gregory, Anthony E., Richard Titball, and Diane Williamson. "Vaccine delivery using nanoparticles." Frontiers in cellular and infection microbiology 3 (2013): 13. Distributed under Creative Commons Attribution License (CC BY 3.0), without modification.
All of our products can only be used for research purposes. These vaccine ingredients CANNOT be used directly on humans or animals.


