SLC32A1 is encoded by SLC32A1 gene which is located on 20q11.23. The molecular mass of SLC32A1 is about 57 KDa. It is a member of the solute carriers, also known as SLCs, which may be the second largest family of membrane proteins. Structurally, SLCs owns a few of hydrophobic transmembrane alpha-helices which take use of hydrophilic intra- and extra-cellular loops to connect to each other. What’ s more, SLC32A1 is only localized intracellularly, it involves in composing cytoplasmic vesicle membrane.
| Basic Information of SLC32A1 | |
| Protein Name | Vesicular inhibitory amino acid transporter |
| Gene Name | SLC32A1 |
| Aliases | GABA and glycine transporter, Solute carrier family 32 member 1, Vesicular GABA transporter, hVIAAT |
| Organism | Homo sapiens (Human) |
| UniProt ID | Q9H598 |
| Transmembrane Times | Multi-pass membrane |
| Length (aa) | 525 |
| Sequence | MATLLRSKLSNVATSVSNKSQAKMSGMFARMGFQAATDEEAVGFAHCDDLDFEHRQGLQMDILKAEGEPCGDEGAEAPVEGDIHYQRGSGAPLPPSGSKDQVGGGGEFGGHDKPKITAWEAGWNVTNAIQGMFVLGLPYAILHGGYLGLFLIIFAAVVCCYTGKILIACLYEENEDGEVVRVRDSYVAIANACCAPRFPTLGGRVVNVAQIIELVMTCILYVVVSGNLMYNSFPGLPVSQKSWSIIATAVLLPCAFLKNLKAVSKFSLLCTLAHFVINILVIAYCLSRARDWAWEKVKFYIDVKKFPISIGIIVFSYTSQIFLPSLEGNMQQPSEFHCMMNWTHIAACVLKGLFALVAYLTWADETKEVITDNLPGSIRAVVNIFLVAKALLSYPLPFFAAVEVLEKSLFQEGSRAFFPACYSGDGRLKSWGLTLRCALVVFTLLMAIYVPHFALLMGLTGSLTGAGLCFLLPSLFHLRLLWRKLLWHQVFFDVAIFVIGGICSVSGFVHSLEGLIEAYRTNAED |
The main function of SLC32A1 is the uptake of GABA and glycine, the major fast inhibitory neurotransmitters released at central synapses, into the synaptic vesicles. However, there is a functional coupling between SLC32A1 and the vesicle-bound biosynthetic enzyme GAD65 which makes the neosynthesized GABA have a kinetic advantage over glycine for vesicular uptake. SLC32A1 also takes part in aging, hippocampus development, amino acid transmembrane transport, ion transport, neurotransmitter secretion, and many other biological processes. It is worth mentioning that SLC32A1 has gamma-aminobutyric acid: proton symporter activity. SLC32A1 is expressed in all parts of the spinal cord and brain, and in the retina or more specifically in the horizontal cells, and in the skeletal muscle at low levels, spleen, thymus, liver, and blood. SLC32A1 is present in all inhibitory neurons in the central nervous system. Beyond that, SLC32A1 together with the transporters GlyT2 cooperate to determine the vesicular Glycinergic phenotype.
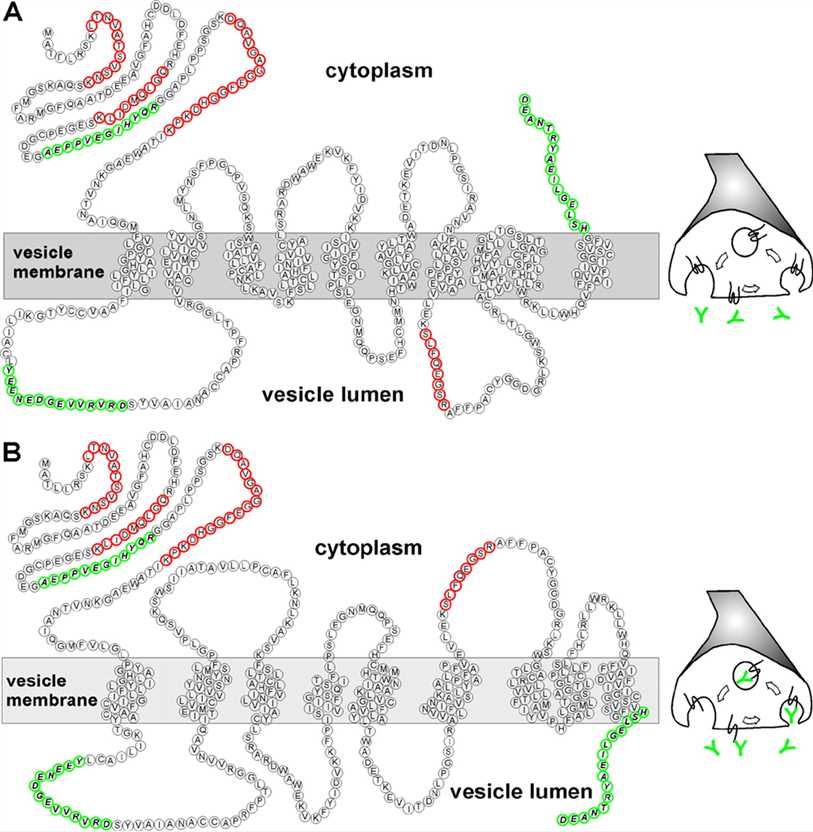 Fig.1 Refined transmembrane topology of VGAT/SLC32A1 (Martens, 2008).
Fig.1 Refined transmembrane topology of VGAT/SLC32A1 (Martens, 2008).
This article mainly talks about the relationship between glycine and GABA accumulation in the presynaptic cytosol and the inhibitory vesicle phenotype. The conclusion reveals that vesicular phenotype is not simply determined by the competition of inhibitory transmitter for VIAAT/SLC32A1, it more likely that the GABA/glycine balance in vesicles is dynamically regulated.
This article reveals that the deletion of VIAAT in GlyT2-Cre expressing neurons has a strong connection with the GABAergic transmission. It also shows a large overlap of the glycinergic and the GABAergic neuron population during early development in the caudal parts of the brain.
Authors in this article use a novel double-sniffer patch-clamp technique to measure the quantal release of glycine and GABA. They suggest that the increased cytosolic availability of glycine in VIAAT/SLC32A1-containing terminals is very important for the emergence of glycinergic transmission.
This article focuses on the role of GABA signaling in palatogenesis. The authors want to see whether Gad1 or SLC32A1/VIAAT function is necessary for the fetal CNS for normal palate development.
Authors of this article reveal that VGLUT2 and VGAT coexist in a sizeable pool of vesicles. SLC32A1/VGAT immunoisolates transport glutamate as well as GABA. The synaptic coexistence of vesicular glutamate and GABA transporters may prevent systemic overexcitability by downregulating synaptic activity.
To obtain the soluble and functional target protein, the versatile Magic™ membrane protein production platform in Creative Biolabs enables many flexible options, from which you can always find a better match for your particular project. Besides, aided by our versatile Magic™ anti-membrane protein antibody discovery platform, we also provide customized anti-SLC32A1 antibody development services.
As a forward-looking research institute as well as a leading custom service provider in the field of membrane protein, Creative Biolabs has won a good reputation among our worldwide customers for successfully accomplishing numerous challenging projects including generation of many functional membrane proteins. Please feel free to contact us for more information.
Reference
All listed services and products are For Research Use Only. Do Not use in any diagnostic or therapeutic applications.