Overcoming Barriers in HSC Gene Therapy
Hematopoietic Stem Cell (HSC) gene therapy holds immense promise for curing monogenic diseases like Sickle Cell Disease (SCD), Beta-Thalassemia, and SCID. However, the path to the clinic is obstructed by technical hurdles: maintaining HSC "stemness" during transduction, achieving therapeutic levels of expression (e.g., hemoglobin), and minimizing the risk of insertional mutagenesis.
Creative Biolabs provides a specialized Lentiviral Vector (LV) development platform tailored for HSCs. We engineer vectors that balance high transduction efficiency in CD34+ cells with precise, lineage-specific expression control, ensuring safety and efficacy from early R&D through to preclinical validation.
Stemness Retention
Optimized transduction protocols and media formulations that prevent premature differentiation of primitive HSCs.
Therapeutic Expression
Incorporation of Locus Control Regions (LCRs) and physiological promoters to achieve curative protein levels.
Safety First
Advanced SIN designs with chromatin insulators (e.g., cHS4) to reduce genotoxicity and positional effects.
HSC Vector Design Services
Hemoglobinopathies (SCD & Beta-Thal)
Vectors designed for high-level, erythroid-specific expression of β-globin or γ-globin.
Incorporation of Locus Control Regions (LCR) and erythroid-specific promoters (e.g., Ankyrin, Spectrin) to mimic physiological regulation.
Optimization of vector backbones to accommodate large globin gene inserts containing introns, critical for mRNA stability.
Assessment of hemoglobin tetramer formation and reversal of sickling phenotype in patient-derived CD34+ cells.
Primary Immunodeficiencies (SCID/WAS)
Restoration of immune function via HSC modification for diseases like X-SCID, ADA-SCID, and Wiskott-Aldrich Syndrome.
Usage of moderate-strength ubiquitous promoters (e.g., EF1α short, PGK) to avoid cytotoxicity associated with overexpression in developing lymphocytes.
Self-Inactivating (SIN) LTRs coupled with insulators to prevent trans-activation of nearby oncogenes (LMO2, etc.).
Metabolic & Lysosomal Disorders
Targeting microglial or systemic enzyme production for conditions like MLD, Hurler Syndrome, or Adrenoleukodystrophy.
Engineering HSCs to act as "protein factories," secreting enzymes for cross-correction of neighboring cells in the CNS and periphery.
Fusion of signal peptides (e.g., ApoE, Iduronate-2-sulfatase signal) to enhance secretion and blood-brain barrier crossing.
HSC Gene Editing (CRISPR/Cas9)
Transient delivery or stable integration of editing machinery for precise correction.
Integration-deficient Lentiviral Vectors (IDLVs) for transient expression of Cas9/gRNA to minimize off-target effects.
Design of AAV6-mimicking lentiviral donors for Homology Directed Repair (HDR) knock-in strategies.
Barcoding & Library Screening
Tools for tracking clonal diversity and studying hematopoiesis dynamics.
High-complexity cellular barcoding libraries to trace lineage output and clonal dominance in repopulation studies.
Pooled shRNA/CRISPR screens tailored for primary HSCs to identify factors regulating self-renewal vs. differentiation.
Preclinical Scale-up
Process development to transition from research-grade to preclinical-grade vector manufacturing.
Suspension cell culture adaptation and scalable purification (chromatography-based) to ensure high yield and purity.
Comprehensive testing for Replication Competent Lentivirus (RCL), host cell proteins/DNA, and endotoxin levels.
Technical Capabilities
Our platform integrates cutting-edge virology with hematopoietic biology to solve the unique challenges of stem cell gene therapy.
Vector Architecture Optimization
Standard lentiviral vectors often suffer from silencing or low titers when dealing with complex, lineage-specific cassettes. We utilize proprietary backbone modifications to enhance stability and nuclear import in quiescent stem cells.
-
Insulators & Boundary Elements We routinely incorporate cHS4 or other synthetic insulators to block enhancer-promoter interactions between the vector and the host genome, mitigating silencing and genotoxicity risks.
-
Codon Optimization Crucial for cryptic splice site removal and enhancing mRNA stability, especially for large genes like Factor VIII or globin variants.
Transduction Enhancement
Achieving high Vector Copy Number (VCN) without compromising viability is key. We offer optimized pseudotyping and adjuvant protocols specifically for CD34+ cells.
-
Pseudotyping Options Beyond VSV-G, we offer Gibbon Ape Leukemia Virus (GALV) or RD114-TR pseudotypes which interact with different receptors (PiT-1/ASCT2), often resulting in higher efficiency in HSCs with lower toxicity.
-
Transduction Enhancers Protocols compatible with Poloxamer 338, PGE2, or Cyclosporine A to boost entry and overcome intracellular restriction factors.
Production & Purity
HSCs are extremely sensitive to impurities. Our downstream processing minimizes cellular debris and plasmid carryover that can trigger immune responses or toxicity.
| Grade | Titer (Functional) | Target Application |
|---|---|---|
| Research Grade | > 10^8 TU/mL | Cell lines, preliminary CD34+ screening. |
| Preclinical Grade | > 10^9 TU/mL | Animal models (NSG mice xenografts), colony-forming unit (CFU) assays. |
| GMP-Like / Pilot | Custom High Concentration | Toxicology studies, large animal validation, process lock-in. |
Application Scenarios
Hemoglobin Disorders
- • Beta-Thalassemia correction
- • Sickle Cell Disease (Anti-sickling globin)
- • Fetal Hemoglobin reactivation
Immunodeficiencies
- • SCID-X1 (IL2RG)
- • ADA-SCID restoration
- • Wiskott-Aldrich Syndrome (WAS)
Metabolic Diseases
- • Metachromatic Leukodystrophy (ARSA)
- • Adrenoleukodystrophy (ABCD1)
- • Fabry Disease
Drug Discovery
- • Hematotoxicity screening models
- • Leukemic stem cell targeting
- • HSC expansion factor screening
Genome Editing
- • IDLV delivery of Cas9/ZFNs
- • HDR donor template delivery
- • Base editor delivery
Lineage Tracing
- • Cellular barcoding libraries
- • Clonal tracking in vivo
- • Hematopoiesis dynamics study
Why Partner with Creative Biolabs?
Deep Design Expertise
We understand the intricacies of HSC gene regulation. We don't just clone; we advise on LCR inclusion, intron positioning, and insulator selection to ensure your vector works in vivo.
Scalable Manufacturing
Our platform spans from small-scale screening (mL) to large-scale bioreactor production (L), enabling a seamless transition from concept to pre-clinical validation.
Robust Quality Control
Rigorous safety testing including RCL, sterility, and specific titer assays on relevant cell lines (e.g., K562 for globin vectors) ensures data reliability.
Collaborative Support
Dedicated project managers with gene therapy backgrounds keep you updated weekly. We act as an extension of your own laboratory.
Development Workflow
A rigorous, stage-gated process designed to ensure high-titer, safety-optimized vectors suitable for sensitive hematopoietic stem cell applications.
Consultation & Strategy Design
Our scientific team collaborates with you to define the Target Product Profile (TPP). We analyze the specific gene therapy requirements—whether for gene addition (e.g., Globin) or editing (CRISPR/Cas9)—and select the optimal promoter (lineage-specific LCRs vs. ubiquitous EF1α), regulatory elements (WPRE, cPPT), and safety features (chromatin insulators, SIN configuration) to maximize efficacy in CD34+ cells while minimizing genotoxicity risks.
Vector Construction & Verification
We perform in silico optimization of the transgene to enhance mRNA stability and remove cryptic splice sites. The transfer plasmid is constructed using seamless cloning technologies to ensure sequence fidelity. Before packaging, every construct undergoes comprehensive validation, including Sanger sequencing of the full insert and restriction enzyme digestion, to confirm structural integrity and alignment with the design strategy.
Packaging & Optimization
Vectors are produced in HEK293T cells using a 3rd generation, helper-independent packaging system to ensure safety. We optimize the ratio of transfer, envelope (VSV-G or alternative pseudotypes like RD114-TR for higher HSC tropism), and packaging plasmids. Transfection conditions are fine-tuned to maximize viral particle production while maintaining cell viability, crucial for generating high-quality supernatants.
Purification & Concentration
Crude supernatants undergo multi-step downstream processing tailored to the required grade. For preclinical applications, we utilize anion-exchange chromatography and Tangential Flow Filtration (TFF) to effectively remove cellular debris, host cell proteins, and plasmid DNA. This process concentrates the vector to high titers (>10^9 TU/mL) while preserving particle infectivity, ensuring the material is non-toxic to sensitive primary stem cells.
Comprehensive Quality Control
Our release testing goes beyond basic titration. We perform physical titer assays (p24 ELISA) and functional titer assays (qPCR/FACS on relevant cell lines). Safety testing includes screening for Replication Competent Lentivirus (RCL), sterility (bacteria/fungi), mycoplasma, and endotoxin levels (< 10 EU/mL). For HSC applications, we can also assess transduction efficiency in CD34+ cells upon request.
Delivery & Technical Support
The final product is aliquoted and shipped on dry ice with a complete Certificate of Analysis (CoA) and full vector maps. Our support doesn't end at delivery; we provide detailed protocols for thawing, handling, and transduction, including recommendations for Multiplicity of Infection (MOI) and the use of transduction enhancers (e.g., Poloxamer 338, PGE2) to ensure optimal experimental outcomes.
What You Receive
We deliver more than just vectors; we provide a comprehensive research package designed to ensure seamless integration into your HSC workflow, complete with rigorous documentation and quality assurance.
Vector Data & Reference Materials
You will receive fully annotated electronic vector maps compatible with standard molecular biology software, alongside raw sequencing data verifying the integrity of your specific insert. We also typically include a sample of the purified plasmid DNA used for packaging, allowing you to propagate the construct in your own laboratory for future needs or verification.
High-Quality Viral Preparations
The lentiviral vectors are delivered as high-titer, cryopreserved aliquots optimized to minimize freeze-thaw cycles. The total volume and concentration are tailored to the scale of your project request, ensuring you have sufficient material for your planned transduction experiments, whether for in vitro stem cell assays or in vivo animal models.
Comprehensive Quality Documentation
Every batch release is accompanied by a detailed Certificate of Analysis (CoA). This document summarizes critical quality attributes—such as functional titer results, sterility confirmation, and relevant safety assessments—providing you with the necessary data to validate the reagent before use in sensitive hematopoietic applications.
Published Data
Development Case: High-titer LV production via host-factor discovery
Background
The rapid expansion of LV-based therapies (e.g., CAR-T, HSCs) requires scalable, high-titer manufacturing. However, traditional production is often limited by yield bottlenecks. To overcome this, we moved beyond rational engineering to a systematic, unbiased CRISPR screening approach to identify novel host factors that fundamentally drive viral productivity.
Solution
Researchers established a genome-wide host factor discovery platform using CRISPR Knockout and Activation libraries in HEK293T cells.
- Dual Screening: Parallel interrogation of gene knockouts and activations.
- Iterative Enrichment: A "Transduction → Rescue → Re-transduction" cycle was used to selectively enrich gRNAs that boost viral output.
- Target Identification: NGS analysis pinpointed functional drivers promoting high-titer production.
Result
The screen successfully identified potent candidates for yield enhancement. Validation confirmed that modulating these targets leads to measurable titer increases. These findings enable the engineering of next-generation packaging cell lines with combinatorial modifications for superior industrial-scale LV manufacturing.
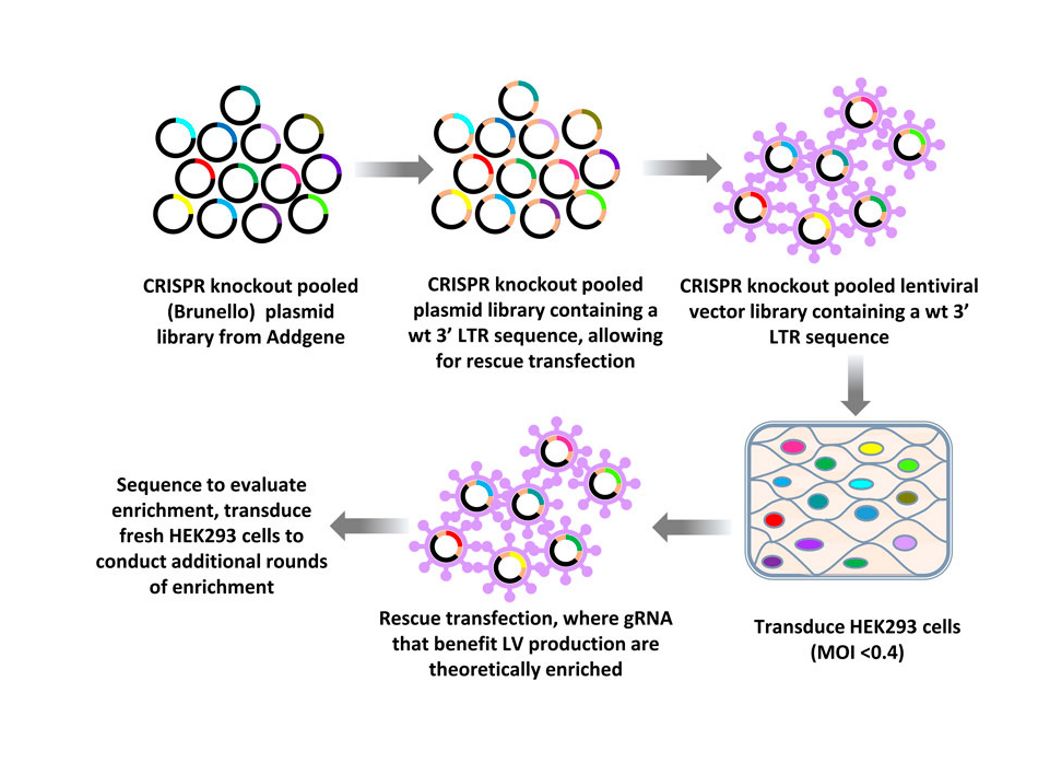
Figure 1. Workflow of CRISPR knockout library screening to identify genomic targets affecting LV vector production.
Iaffaldano B J, Marino M P, Reiser J. CRISPR library screening to develop HEK293-derived cell lines with improved lentiviral vector titers. Frontiers in Genome Editing, 2023, 5: 1218328. https://doi.org/10.3389/fgeed.2023.1218328 Distributed under Open Access license CC BY 4.0, without modification.
Frequently Asked Questions
Start Your HSC Vector Design
Prepare this information to expedite your project proposal:
- Transgene: Gene size and sequence (e.g., Globin, Enzyme, GFP).
- Regulation: Ubiquitous (EF1a) or Lineage Specific (LCR/Ankyrin)?
- Target Cell: Human CD34+, Mouse LSK, or Cell Lines (K562)?
- Scale: Research prep (10^8 TU) or Preclinical (10^9 TU)?
- Safety: Requirement for insulators or specific purity levels?
Get a Specialized Quote
Our experts will review your requirements and propose a vector strategy.
Start Your Project Today
Tell us about your project, and our experts will get back to you with a customized quote and proposal.
