CRISPR mediated Interferon-stimulated Gene Knockout Screening Service
Introduction
Creative Biolabs' CRISPR-mediated Interferon-Stimulated Gene (ISG) Knockout Screening Service leverages cutting-edge CRISPR/Cas9 loss-of-function screening paired with quantitative transcriptional and high-content analyses. It pinpoints precise, drug-susceptible host regulators that govern virus-defensive ISG expression—validating well-characterized factors while unearthing previously unknown ones. As a pioneer in functional genomics, we provide mechanistic insights, ISG pathway categorization, and high-confidence candidates to empower host-focused antivirals and mitigate drug development risks.
Discover How We Can Help - Request a Consultation Today
CRISPR mediated Interferon-stimulated Gene Knockout Screening Service
Background of Interferon-Stimulated Genes (ISGs)
ISGs are a family of hundreds of proteins induced by IFN signaling upon viral detection. They represent the primary cellular defense, acting via diverse mechanisms to restrict viral entry, replication, and egress. However, viruses like SARS-CoV-2 and Influenza A have evolved intricate mechanisms to suppress or dysregulate this pathway, leading to either uncontrolled viral spread or harmful immunopathology. Effective therapeutic intervention hinges on a thorough grasp of the host factors that either enhance ISG function or modulate the viral-host transcriptional crosstalk.
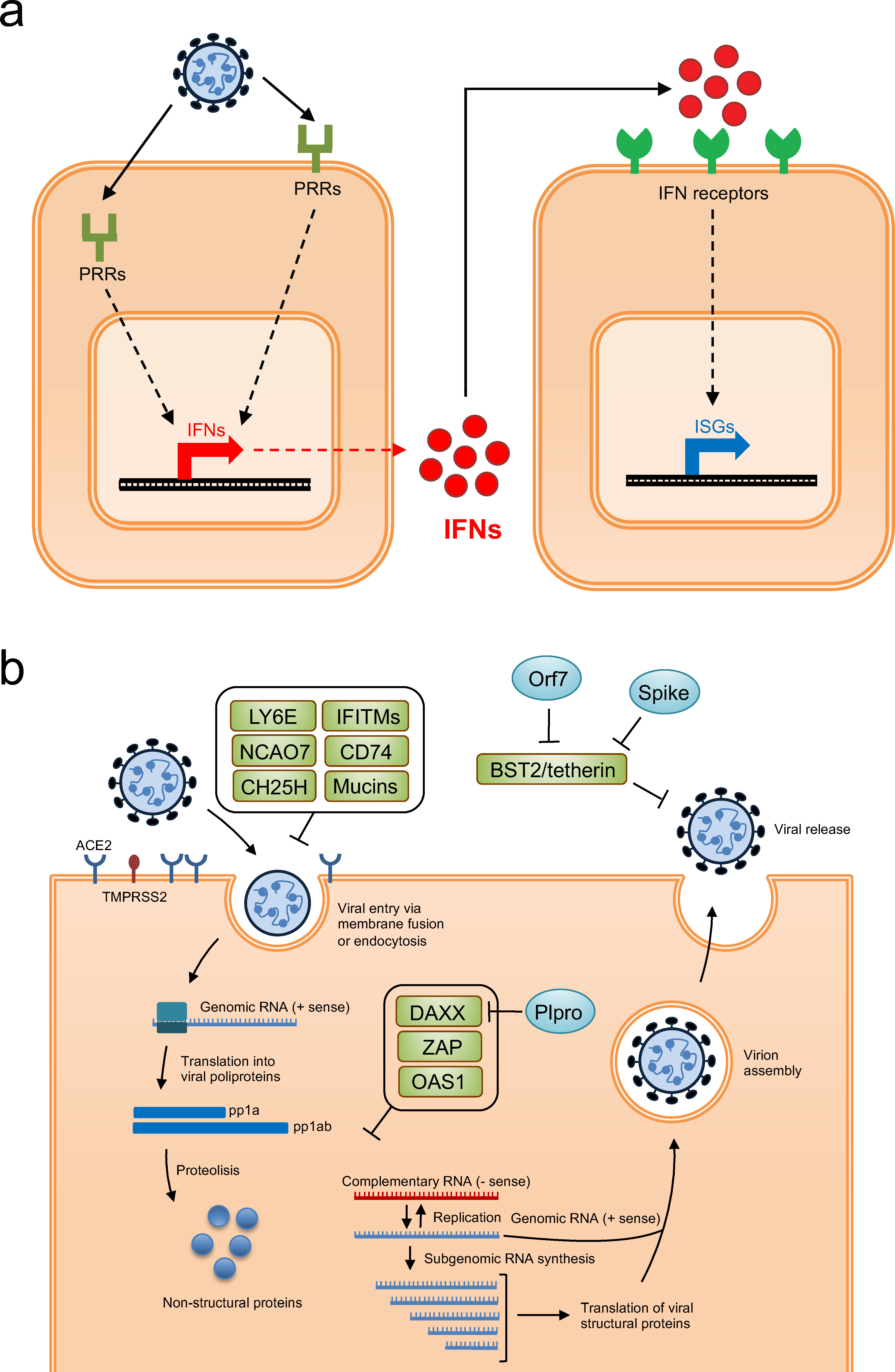 Fig.1 Schematic diagram of interferon response and various ISGs with identified antiviral activity.1,3
Fig.1 Schematic diagram of interferon response and various ISGs with identified antiviral activity.1,3
Screening Purpose: Identification of Regulatory Host Factors
The core purpose of our Knockout Screening Service is to execute a systematic, unbiased search for host factors whose loss-of-function significantly impacts the ISG response. Specifically, we aim to:
- Validate Canonical Components: Confirm the essential role of known factors.
- Discover Novel Transcriptional Regulators: Identify uncharacterized genes that modulate the expression magnitude or specificity of ISGs.
- Identify Vulnerabilities: Pinpoint host factors that, when targeted, can either enhance a protective ISG response or selectively suppress a pathogenic, constitutive IFN state (interferonopathies).
Subsequent Application: Drug Development Leads
The high-confidence host factors identified by our screening service are immediately translated into therapeutic lead programs, focusing on two key application areas:
- Pan-Viral Antivirals: Targeting host factors (like specific ISGs themselves) offers a defense strategy less susceptible to rapid viral evolution or mutation, providing a path to broad-spectrum antivirals.
- Immunomodulatory Therapeutics: Targeting regulatory factors allows for the development of small molecules that offer a nuanced therapeutic control over the IFN system, avoiding the severe immunosuppression associated with broad inhibitors (e.g., JAK inhibitors).
Workflow
Required Starting Materials
- Specific Viral Strain/Stimulus: E.g., SARS-CoV-2 spike pseudovirus, Influenza A variant, or innate immune agonist (poly (I:C)).
- Target Cell Line: E.g., A549, Vero E6, or Primary PBMCs, expressing stable Cas9.
- Specific Genes/Pathways: Existing preliminary data or a focus list for customized sub-library design.


Library Generation & Transduction
Utilize high-complexity GeCKOv2 or custom CRISPR-KO libraries targeting over 19,000 genes, ensuring low multiplicity of infection (MOI) for single-guide RNA (sgRNA) integration.
Phenotypic Selection & Quantitative Readout
Challenge cells with the target virus or stimulate (e.g., with IFN). Employ specialized assays, including the gold-standard CRITR-seq for quantitative transcriptional measurement and eGFP-reporter-based FACS sorting for complex phenotypes.


Genomic DNA Isolation & NGS Sequencing
Extract genomic DNA from selected and control cell populations, followed by targeted Next-Generation Sequencing (NGS) of the integrated sgRNA barcodes.
Bioinformatic Deconvolution & Analysis
Use proprietary algorithms (e.g., αRRA (Adjusted Robust-Rank Aggregation)) to analyze sgRNA enrichment/depletion, providing statistically significant host factor hit lists.


Hit Validation & Mechanistic Profiling
Individually knock out top candidates (e.g., BRD9, NELF) for detailed secondary confirmation using Western Blot, qPCR of key ISGs, and viral titer reduction assays to confirm the role and dissect the mechanism (e.g., chromatin state, transcription factor association).
Final Deliverables
- Prioritized Gene Hit List: A comprehensive list of statistically significant host factors with associated p-values and screening scores.
- Comprehensive Mechanistic Report: Detailed validation data (e.g., qPCR of ISGs, viral titer reduction curves) for all top-tier candidates.
- Customized Data Set: Raw and processed NGS sequencing data suitable for advanced in-house bioinformatic analysis.


Estimated Timeframe
The typical timeframe for this comprehensive service ranges from 14 to 20 weeks, depending on the complexity of the target virus and the number of secondary validation assays required.
What We Can Offer
Our CRISPR mediated Interferon-stimulated Gene Knockout Screening Service is purpose-built for experienced biology experts seeking to identify and validate novel, druggable host targets with unparalleled precision. At Creative Biolabs, we don't just provide a data dump; we deliver high-confidence, mechanistically defined therapeutic leads.
End-to-End Functional Genomics Solution
Comprehensive, end-to-end functional genomics solution from customized library design to validated therapeutic lead identification.
Pioneering CRITR-seq Technology
Pioneering application of CRISPR with Transcriptional Readout (CRITR-seq), allowing for superior quantitative analysis of complex, heterogeneous IFN induction phenotypes.
Druggable Host Regulator Identification
Focus on identifying non-obvious, druggable host regulators (e.g., chromatin remodelers like BRD9, transcriptional elongation factors like NELF) that are ideal starting points for small-molecule programs.
Full Customization & Adaptability
Full customization and adaptability of the screening platform, supporting virtually any viral strain, unique cell line (including primary human cells), and targeted ISG sub-libraries to meet your exact project needs.
Five-Step Hit Validation
Rigorous five-step hit validation and mechanistic profiling that includes qPCR, Western Blot, and viral titer reduction assays to de-risk targets before they enter the drug pipeline.
Dual Therapeutic Strategy Support
Translational data that empowers the development of two distinct therapeutic strategies: broad-spectrum pan-viral antivirals and nuanced immunomodulatory therapeutics.
Experience the Creative Biolabs Advantage – Get a Quote Today
Case Study
The type I interferon signaling pathway induces ISG expression, which contains multiple classical gene components, including IFNAR1, JAK1, STAT1, STAT2, IRF9, etc. To identify other previously uncharacterized genes that are crucial for interferon signaling and the induction of ISG expression, the CRISPR-Cas9 screening technology was used to screen the A549-based reporter cell line. It was found that BRD9 was significantly enriched, and subsequent research was conducted.
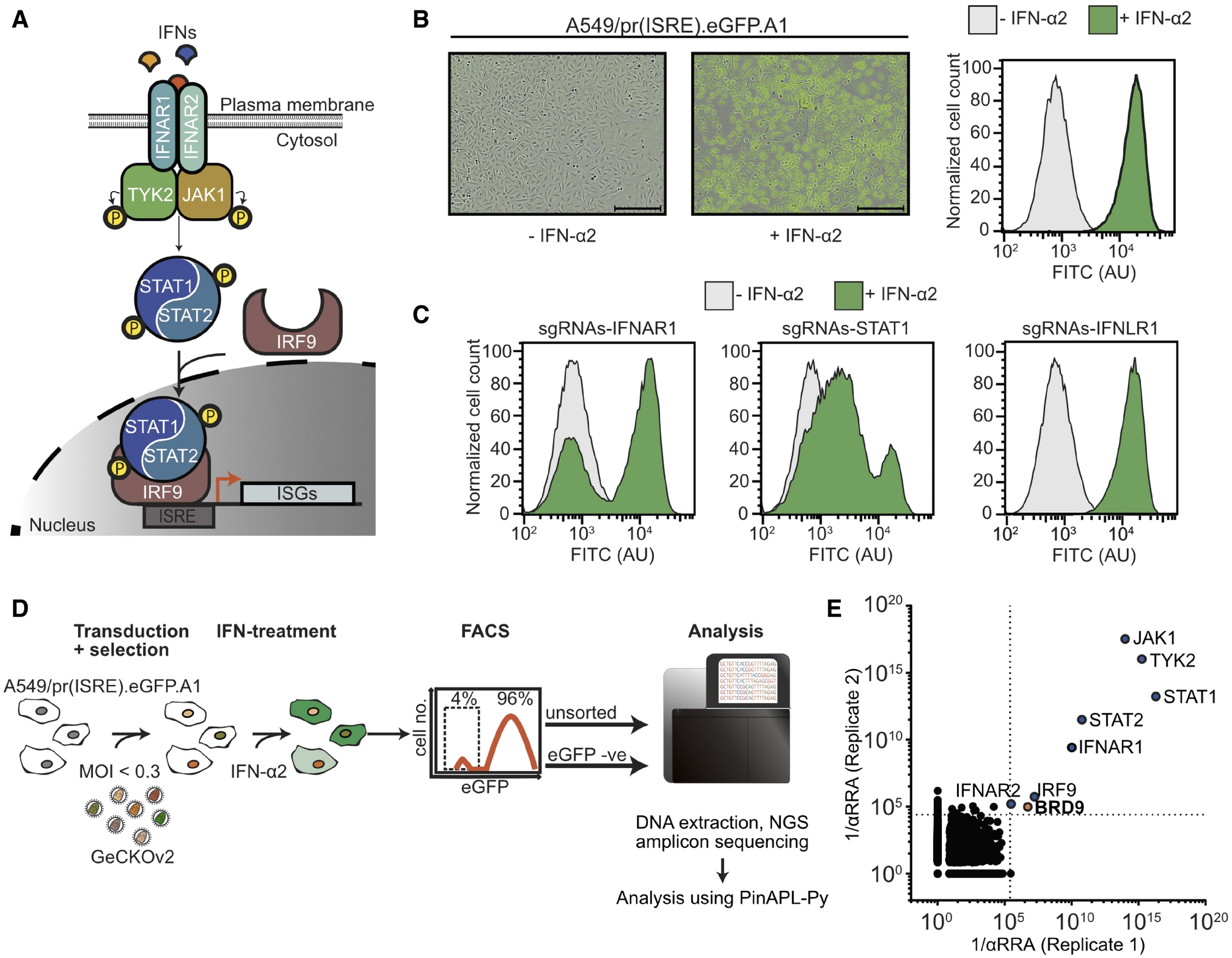 Fig.2 Using CRISPR KO whole-genome screening technology, human genes that are crucial for interferon-stimulated gene expression were identified.2,3
Fig.2 Using CRISPR KO whole-genome screening technology, human genes that are crucial for interferon-stimulated gene expression were identified.2,3
Customer Reviews

FAQs
How does your CRISPR-KO screen address the heterogeneity of the IFN response, which can vary widely from cell to cell?
We overcome this challenge by employing CRITR-seq (CRISPR with Transcriptional Readout using sequencing), a next-generation approach that provides a quantitative, sequencing-based measurement of transcription at the IFN promoter. This method captures the true distribution and magnitude of the ISG phenotype, delivering more precise and reliable data than binary selection methods. We encourage you to contact us for a detailed demonstration of our CRITR-seq capabilities.
Why target host factors like BRD9 instead of directly targeting the virus? Isn't that riskier?
Targeting host factors, especially critical regulators like BRD9 (involved in chromatin remodeling) or NELF (involved in transcription), offers a significant advantage: it makes the resulting drug less susceptible to viral mutation. By focusing on host mechanisms that are essential for the virus's life cycle or the host's response, we unlock pathways for pan-variant therapeutics. Our validation assays rigorously test the specificity to ensure minimal impact on basal cell viability.
What is the typical lead quality derived from your service, and how does it compare to general high-throughput screening (HTS) hits?
Our service delivers genetically validated targets with confirmed roles in ISG regulation. Unlike general HTS hits that often fail downstream validation, our leads are already confirmed by genetic loss-of-function to modulate the desired phenotype. This dramatically improves the success rate of subsequent small-molecule campaigns. The average reduction in false-positive rates leads to high cost and time savings.
Can this service be customized for a specific viral strain or a rare cell type (e.g., primary human cells)?
Absolutely. Our platform is highly flexible. We can adapt the service for virtually any viral strain, pseudovirus, or innate immune agonist. We routinely perform screens in various cell lines, including primary cell-derived models. The initial consultation is critical for defining the perfect model system and customizing the sgRNA library to meet your specific project requirements.
Creative Biolabs' CRISPR mediated Interferon-stimulated Gene Knockout Screening Service represents the most advanced solution for identifying and validating novel host-targeted antiviral and immunomodulatory drug leads. By combining the precision of CRISPR-KO and CRISPRa with quantitative assays like CRITR-seq, we move beyond symptom management to target the core mechanisms of host-virus interaction (e.g., BRD9, NELF). Leverage our expertise to de-risk your pipeline and discover precise, druggable targets with superior therapeutic potential.
Contact Our Team for More Information and to Discuss Your Project
References
- Ortega-Prieto, Ana Maria, and Jose M. Jimenez-Guardeno. "Interferon-stimulated genes and their antiviral activity against SARS-CoV-2." Mbio 15.9 (2024): e02100-24. https://doi.org/10.1128/mbio.02100-24.
- Börold, Jacob, et al. "BRD9 is a druggable component of interferon‐stimulated gene expression and antiviral activity." EMBO reports 22.10 (2021): e52823. https://doi.org/10.15252/embr.202152823.
- Distributed under Open Access license CC BY 4.0, without modification.
