Overcoming Barriers in Stem Cell Engineering
Genetic modification of stem cells holds immense promise for regenerative medicine and disease modeling. However, researchers face persistent challenges: promoter silencing during differentiation, cytotoxicity affecting pluripotency, and low transduction efficiency in quiescent stem cell populations.
Creative Biolabs provides a specialized Lentiviral Vector (LV) design platform tailored for stem cell applications. We engineer vectors with chromatin-opening elements (UCOEs), tissue-specific promoters, and optimized envelopes to ensure stable, long-term transgene expression in Induced Pluripotent Stem Cells (iPSCs), Hematopoietic Stem Cells (HSCs), and Mesenchymal Stem Cells (MSCs).
Anti-Silencing Design
Incorporation of UCOEs and scaffold attachment regions (SARs) to prevent transgene silencing during the epigenetic remodeling of differentiation.
Lineage Specificity
Promoters driven by lineage-specific transcription factors (e.g., MHCK7 for muscle, GFAP for glia) to restrict expression to differentiated progeny.
Reprogramming Efficiency
Polycistronic vectors (STEMCCA) delivering OKSM factors for efficient generation of iPSCs with excision capabilities (Cre-LoxP).
Stem Cell Vector Services
Cellular Reprogramming & Engineering
Tools for generating and modifying Induced Pluripotent Stem Cells with minimal genomic footprint.
Polycistronic STEMCCA vectors (Oct4, Klf4, Sox2, c-Myc) with excisable Cre-LoxP systems for footprint-free iPSC generation.
Knock-in of fluorescent reporters (GFP/RFP) driven by pluripotency markers (Nanog, Oct4) or lineage-specific promoters to monitor differentiation status in real-time.
Verification of pluripotency maintenance post-transduction via alkaline phosphatase staining and teratoma formation assays.
Hematopoietic Stem Cells (HSCs)
High-efficiency transduction of CD34+ cells for gene therapy research and blood lineage studies.
Usage of VSV-G or specialized envelopes (e.g., BaEV, RD114) to enhance entry into quiescent CD34+ stem cells with reduced toxicity.
Incorporation of Locus Control Regions (LCRs) specifically for globin gene expression or other erythroid/myeloid specific targets to ensure physiological regulation.
MSC & Mesenchymal Vectors
Engineering Mesenchymal Stem Cells for exosome secretion and tissue regeneration.
Vectors designed to fuse therapeutic proteins/RNAs with exosomal markers (CD63, CD9), enabling MSCs to secrete engineered exosomes for cell-free therapy.
Overexpression of angiogenic or neurotrophic factors (VEGF, BDNF) to enhance the regenerative potential of MSC grafts.
Gene Editing Delivery
Transient or stable delivery of CRISPR machinery into difficult-to-transfect stem cells.
All-in-one or dual-vector systems delivering SpCas9 and sgRNAs. We utilize integrase-deficient lentiviral vectors (IDLVs) for transient expression to minimize off-target effects.
Packaging of large base editor constructs (ABE/CBE) for precise correction of point mutations in patient-derived iPSCs.
Differentiation Screening
Discovery of factors that drive lineage commitment using pooled libraries.
High-complexity cellular barcoding libraries to trace clonal expansion and lineage bias during stem cell differentiation and engraftment.
Genome-wide knockout or activation screens to identify key transcription factors regulating pluripotency exit or specific tissue formation.
Technical Capabilities
Our platform overcomes the unique biological hurdles of stem cell gene transfer, focusing on stability and safety.
Silencing Resistance
Stem cells are prone to epigenetically silencing foreign promoters. We engineer vectors to maintain expression through extensive chromatin remodeling.
-
UCOE & cHS4 Elements Integration of Ubiquitous Chromatin Opening Elements (UCOEs) and insulators to protect the transgene from heterochromatin spreading.
-
Promoter Selection Utilization of "safe" promoters like EF1α-short or CAG, as well as testing panels for specific stem cell lines to ensure robust activity.
Safety & Integration
For applications in disease modeling and potential therapy, vector safety profile is paramount.
-
Integration-Deficient (IDLV) Non-integrating vectors for transient expression of editing tools (Cas9, Cre), reducing the risk of insertional mutagenesis.
-
Self-Inactivating (SIN) Deletion of the viral promoter in the 3' LTR ensures the vector cannot mobilize after integration, meeting strict biosafety standards.
Production & Purification
Stem cells are highly sensitive to contaminants which can trigger spontaneous differentiation or apoptosis. We employ rigorous purification protocols.
| Feature | Specification / Capability | Impact on Stem Cells |
|---|---|---|
| Concentration Method | Ultracentrifugation / TFF | Removes cytokines and debris that could induce unwanted differentiation. |
| Buffer Formulation | PBS / Histidine / Sucrose | Optimized pH and osmolarity to maintain cell viability post-transduction. |
| Purity Grade | Stem Cell Grade | Minimizes protein impurities and residual DNA content. |
QC Analysis
Comprehensive quality control ensures lot-to-lot consistency and data reliability for sensitive iPSC and HSC experiments.
-
Multi-Method Titration We provide both physical (p24 ELISA) and functional titers (FACS on 293T or iPSC lines) to ensure accurate MOI calculations (≥ 10^8 - 10^9 TU/mL).
-
Sterility & Safety Rigorous testing for Bacteria, Fungi, and Mycoplasma. Endotoxin levels are strictly controlled (< 10 EU/mL) to prevent TLR activation.
-
RCL Testing Replication Competent Lentivirus (RCL) testing ensures the vectors are safe for long-term culture and potential clinical translation studies.
Which Vector Fits My Study?
Select the optimal vector architecture based on your specific stem cell application and downstream goals.
| Research Goal | Recommended Vector | Key Features | Why Choose This? |
|---|---|---|---|
|
iPSC Reprogramming
|
Polycistronic STEMCCA (Cre-LoxP) | Delivers Oct4, Klf4, Sox2, c-Myc on a single cassette; excisable. | High efficiency reprogramming with the ability to remove viral sequences later for "footprint-free" iPSCs. |
|
Long-term Differentiation
|
UCOE-EF1α / UCOE-CAG | Contains Ubiquitous Chromatin Opening Elements (UCOE). | Prevents promoter methylation and silencing, ensuring transgene expression persists from iPSC stage to mature neuron/cardiomyocyte. |
|
Transient Gene Editing
|
IDLV (Integration-Deficient) | Non-integrating mutated integrase packaging. | Delivers CRISPR/Cas9 or Cre transiently to modify the genome without permanent viral integration or insertional mutagenesis risk. |
|
HSC Gene Therapy
|
LCR-Driven Vectors | Includes Locus Control Regions (e.g., β-globin LCR). | Essential for physiological, high-level expression restricted to the erythroid lineage in Hematopoietic Stem Cells. |
|
Lineage Tracing
|
Barcode Library Vector | High-complexity DNA barcodes + Constitutive GFP/RFP. | Allows tracking of clonal expansion and differentiation bias of individual stem cells via NGS analysis. |
Not sure which vector is right for your cells?
Application Scenarios
From basic developmental biology to translational gene therapy, our vectors are designed to meet diverse research needs.
Disease Modeling
Generate isogenic disease models by transducing healthy iPSCs with vectors carrying patient-specific mutations or dominant-negative alleles. Our tightly regulated inducible vectors allow for the temporal study of disease onset during differentiation.
Development of therapeutic vectors for HSC gene correction (e.g., Sickle Cell Disease, Beta-Thalassemia). We optimize globin-specific promoters and LCRs to ensure high-level, erythroid-specific expression in potential clinical candidates.
Lineage Tracing
Utilize our high-complexity barcode libraries to label individual stem cells. This allows researchers to track clonal dynamics and lineage bias during hematopoiesis or organoid development via Next-Generation Sequencing (NGS).
Organoid Engineering
Efficiently label or modify cells prior to organoid formation. Our vectors enable the creation of reporter organoids to visualize developmental processes, such as neural tube closure or gut epithelial differentiation, in 3D culture systems.
Gene Editing Support
Deliver CRISPR/Cas9, Cas12a, or Base Editors to iPSCs and MSCs. We offer selectable markers (Puromycin, Blasticidin) to enrich for edited cells, facilitating the generation of knockout or knock-in cell lines.
Direct Reprogramming
Trans-differentiation of somatic cells directly into therapeutic cell types (e.g., fibroblasts to neurons or cardiomyocytes) using vectors expressing lineage-determining transcription factors (e.g., Ascl1, Brn2, Myt1l).
Why Partner with Creative Biolabs?
We combine expertise in virology and stem cell biology to deliver vectors that maintain the delicate balance of pluripotency and differentiation.
Pluripotency Safe
We understand that standard viral preparations can induce differentiation. Our "stem-cell grade" purification removes impurities that trigger signaling pathways, ensuring your iPSCs remain undifferentiated until you decide otherwise.
Complex Payload Design
From polycistronic reprogramming cassettes (4-in-1 vectors) to large homology repair templates for gene editing, we have the molecular biology expertise to construct stable, high-capacity vectors.
Custom Tropism
While VSV-G is standard, we offer alternative envelopes (e.g., Mokola, Rabies, Measles) for specific targeting needs or to avoid toxicity associated with constitutive VSV-G expression in sensitive stem cell populations.
Rapid Turnaround
Our optimized production pipeline allows for the delivery of high-titer, purified virus in as little as 2-3 weeks, accelerating your research timeline from vector design to functional data.
Development Workflow
From in silico design to transduced stem cells, our workflow is rigorous and transparent.
Vector Strategy
We analyze your gene of interest and target cell (iPSC, HSC, MSC). We select the optimal promoter (EF1a, CAG, or tissue-specific) and include chromatin opening elements (UCOEs) if silencing is a concern.
Cloning & QC
The construct is built using scar-free cloning techniques. We perform full sequencing of the insert and restriction analysis to ensure plasmid integrity before packaging.
Viral Production
Production in HEK293T cells using a 3rd generation SIN system. We optimize transfection ratios to maximize physical particle yield while minimizing empty capsids.
Purification & Titration
Vectors are purified via ultracentrifugation or chromatography to remove serum and cellular debris. Titers are determined via p24 ELISA and qPCR (genomic copies), with functional titration available on request.
Stem Cell Validation (Optional)
We can transduce control iPSC or MSC lines to verify expression, check for cytotoxicity, and ensure markers of pluripotency (e.g., Tra-1-60, SSEA-4) remain unaffected.
Delivery
Final viral stocks are shipped on dry ice with a detailed Certificate of Analysis. We provide support for calculating Multiplicity of Infection (MOI) for your specific cell lines.
What You Receive
Comprehensive data and reagents ready for immediate use in your stem cell culture workflow.
Plasmid DNA
Full sequence data and map of the lentiviral transfer plasmid, plus a glycerol stock or purified DNA aliquot for your internal records or future expansion.
Viral Aliquots
Purified lentiviral particles are suspended in a stem-cell compatible buffer, carefully stored at -80°C to maintain their stability and efficacy for future experimental use.
CoA & Data
Certificate of Analysis including titer (p24/qPCR), sterility, and endotoxin levels. Functional validation data (FACS/Microscopy) included for custom pilot studies.
Published Data
Case Study: Validation of 293T-S Cell Line for Large-Scale Lentiviral Vector Production
Challenge
Generating high-titer lentiviral vectors at scale is a primary bottleneck in stem cell research and clinical translation. Traditional adherent 293T cell cultures are limited by surface area and often require bovine serum, which complicates downstream purification and increases the risk of variability. To support advanced engineering of iPSCs and HSCs, a high-density, serum-free suspension system is required.
Optimization
A specialized suspension 293T cell line (293T-S) was established and adapted to grow in chemically defined, serum-free media. The cells were validated for their packaging capability using a multi-plasmid transfection system. Researchers utilized a lentiviral transfer plasmid carrying a Green Fluorescent Protein (GFP) reporter to monitor transfection efficiency and functional viral titer produced in the suspension culture.
Outcome
The 293T-S cells demonstrated robust growth reaching high densities and maintained high transfection efficiency. Flow cytometry and fluorescence microscopy confirmed successful viral packaging, as evidenced by high-level GFP expression in target cells transduced with the suspension-derived virus. This system provides a scalable and consistent platform for producing high-quality lentiviral vectors necessary for stem cell modification.
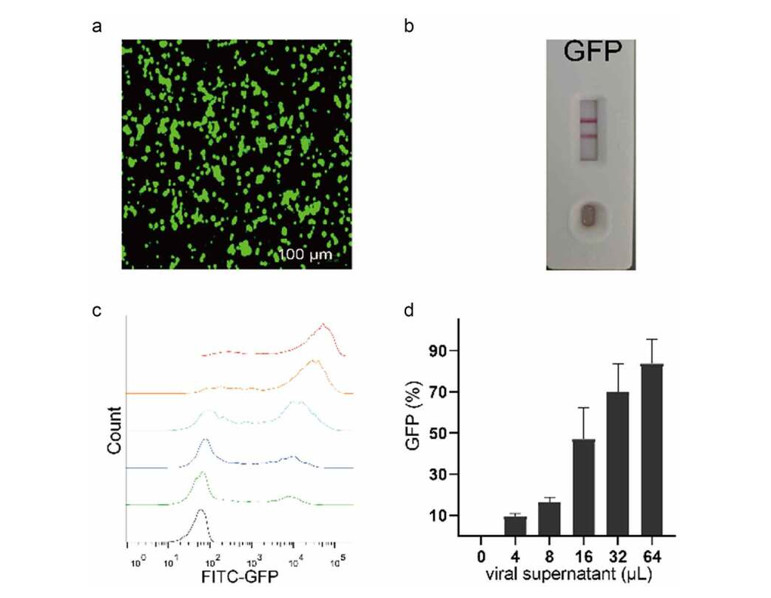
Figure 1: Verification of virus packaging capability of 293 T suspension cells.
Establishment of a bank of suspension 293T cells for the production of recombinant proteins and lentiviral vectors. Commun Biol, 2022;5:102. https://doi.org/10.1080/21655979.2021.1931644 Distributed under Open Access license CC BY 4.0, without modification.
Frequently Asked Questions
Start Your Stem Cell Project
Provide the following details to help us design the perfect vector:
- Cell Type: iPSC, HSC, MSC, or ESC?
- Goal: Stable integration, transient editing, or reprogramming?
- Promoter: Ubiquitous (CAG/EF1a) or Lineage Specific?
- Volume/Titer: Pilot (mL) or High Concentration (>10^9)?
- Safety: Do you require IDLVs or excisable systems?
Get a Specialized Quote
Our stem cell engineering experts will review your requirements and propose a strategy.
Start Your Project Today
Tell us about your project, and our experts will get back to you with a customized quote and proposal.
