AAV Vector Design for MPS IIIA/Sanfilippo A
With the increasing use of adeno-associated virus vectors (AAV) in gene therapy research, Creative Biolabs' AAV vector design platform has also improved in all aspects. The excellent safety and the efficient transduction into a wide range of target tissues make AAV one of the preferred carriers for gene therapy in vivo. However, the development and screening of safe and efficient gene therapy vectors is a long process that requires adjustments to the pathological characteristics of various diseases. Our team of experienced scientists provides AAV vector design services for the treatment of mucopolysaccharidosis type IIIA (MPS IIIA) to meet your professional needs.
Introduction to MPS IIIA
MPS is a type of lysosomal storage disease (LSD) caused by the accumulation of undegraded glycosaminoglycans (GAGs) in various tissues and organs after lysosomal enzyme deficiency. Seven types of MPS are classified according to the lack of specific enzymes, accompanied by various symptoms including central nervous system (CNS) damage, hearing loss, respiratory hazard, valvular heart disease, hepatosplenomegaly, and the like. If there is no effective treatment, the patient usually dies within a few decades. MPS IIIA, also known as Sanfilippo A syndrome, is an autologous recessive neurodegenerative metabolic disease caused by sulfoglucosamine sulfohydrolase (SGSH; sulfamidase) deficiency, resulting in the accumulation of undegraded heparan sulfate (HS) in the organelles, triggering cells disfunction.
Limitations of Current Treatment Strategies
Despite extensive research, current therapeutic approaches remain limited:
| Approach | Limitation |
|---|---|
| Enzyme replacement therapy (ERT) | Poor penetration of the blood-brain barrier (BBB), limited impact on neurological symptoms, requirement for repeated lifelong infusions, and potential immune responses. |
| Hematopoietic stem cell transplantation | Limited neurological benefit, especially after symptom onset; associated risks such as graft-versus-host disease (GVHD), conditioning toxicity, and transplant-related mortality. |
| Symptomatic treatment | Does not address the underlying genetic cause; fails to prevent progressive neurodegeneration and premature mortality. |
| Gene Therapy | Challenges include potential immune responses to AAV, optimization of dosing and delivery routes, long-term safety monitoring, and the need for scalable vector manufacturing, although it represents the most promising disease-modifying approach. |
These challenges highlight the urgent need for gene therapy solutions capable of targeting CNS pathology.
Why Gene Therapy is the Most Promising Approach for MPS IIIA?
Gene therapy directly addresses the root cause of MPS IIIA by delivering a functional copy of the SGSH gene to affected cells, restoring enzyme activity and reducing toxic substrate accumulation.
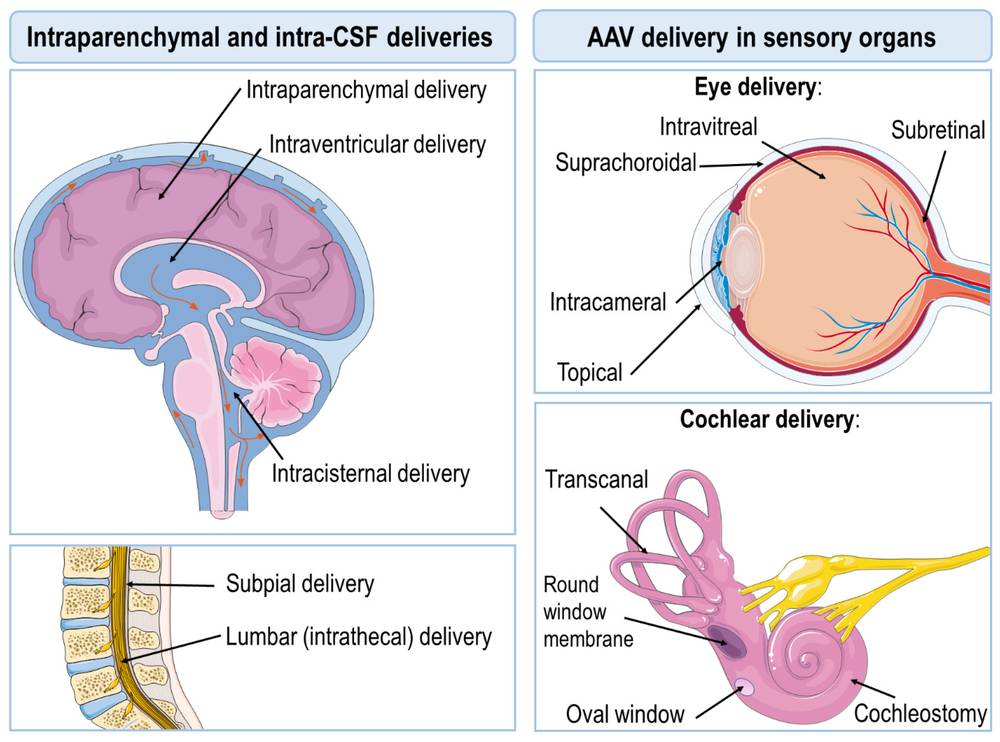 Figure 1. Illustration of most commonly used AAV delivery routes.1,3
Figure 1. Illustration of most commonly used AAV delivery routes.1,3
Mechanism of Action
- Delivery of functional SGSH gene
- Restoration of sulfamidase enzyme activity
- Clearance of accumulated heparan sulfate
- Prevention or slowing of neurodegeneration
Clinical and preclinical studies demonstrate that AAV-mediated gene therapy can significantly improve biochemical and neurological outcomes, particularly when administered early.
MPS IIIA Gene Therapy
Currently, MPS IIIA gene therapy can be divided into two main approaches: one is in vivo gene therapy, which involves injecting the virus carrying the gene intravenously or locally delivering the gene to the patient's body cells; the second is in vitro gene therapy, in which genes are transferred to somatic cells from the patient and then transplanted back into the patient. Recombinant AAV (rAAV) is a common vector for gene therapy for the treatment of MPS. It can effectively infect different cell types, persist as an episome, and has a low risk of insertional mutagenesis and genotoxicity.
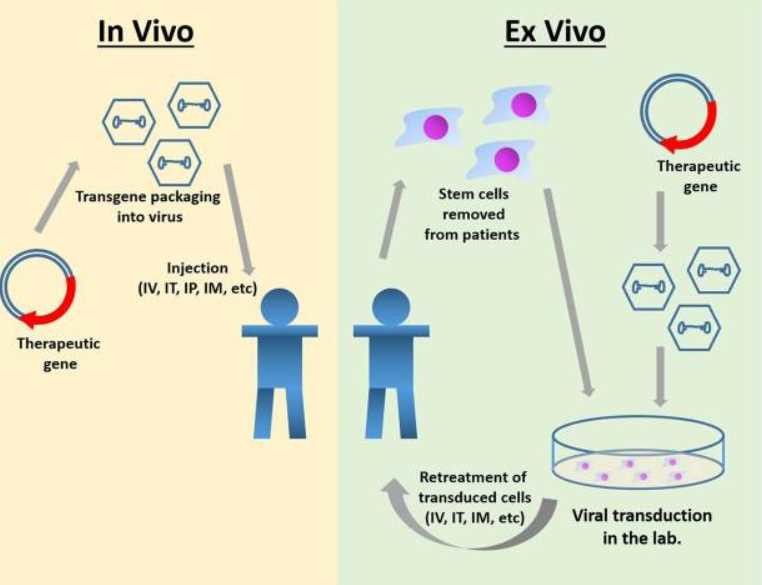 Figure 2. Gene therapy for MPS IIIA.2,3
Figure 2. Gene therapy for MPS IIIA.2,3
Our MPS IIIA Gene Therapy Development Services
At Creative Biolabs, we have built a robust AAV engineering platform tailored for lysosomal storage disorders and CNS-targeted therapies.
Core Capabilities
1. AAV Serotype Selection and Engineering
We offer a comprehensive library of AAV serotypes, including:
- AAV9 (widely used for CNS delivery)
- AAVrh.10 (high CNS tropism and safety profile)
- Engineered capsids for enhanced BBB penetration
These serotypes exhibit broad tissue tropism and efficient neuronal transduction, making them ideal for MPS IIIA applications.
2. Therapeutic Gene Cassette Design
We design optimized SGSH expression constructs with:
- Codon optimization for enhanced expression
- Tissue-specific or ubiquitous promoters
- Regulatory elements for sustained expression
- Self-complementary AAV constructs for rapid onset
3. CNS-Targeted Delivery Optimization
We support multiple administration routes:
| Delivery Route | Application |
|---|---|
| Intracerebral (IC) | Direct CNS targeting |
| Intrathecal (IT) | CSF-mediated brain distribution |
| Intravenous (IV) | Systemic delivery with CNS reach |
Comprehensive Quality Control (QC) & Analytical Development
For MPS IIIA gene therapy delivered directly to the brain (ICV/IT), quality is safety. Even minor impurities can trigger neuroinflammation or loss of effect. Our QC platform focuses on the five most critical parameters that regulators and investigators demand.
1. Empty/Full Capsid Ratio – ≤10% for CNS
Empty capsids dilute potency and may cause immune responses. We use AUC (analytical ultracentrifugation) – gold standard – to guarantee ≤10% empty for ICV/IT products. For IV delivery, ≤20% is acceptable.
2. Replication-Competent AAV (rcAAV)
rcAAV poses an insertional risk. Our highly sensitive serial passage + qPCR assay detects as low as 1 rcAAV per 1×10¹⁰ GC. All lots are rcAAV negative.
3. Potency Assay Tailored to MPS IIIA
We don't just measure titer – we measure functional SGSH enzyme activity in transduced patient fibroblasts. Acceptance: ≥10 fold increase over background. Optional HS reduction by LC MS/MS as a secondary potency endpoint.
4. Sterility, Endotoxin & Mycoplasma
- Sterility (USP <71>): No growth
- Endotoxin: < EU/kg (research) / <2 EU/kg (GLP)
- Mycoplasma: Negative by qPCR
- Aggregates (SEC HPLC): <2% high molecular weight species
5. Residual Impurities
| Impurity | Specification | Method |
|---|---|---|
| Host cell protein | <50 ng/mg (research); <10 ng/mg (GLP) | ELISA |
| Host cell DNA | <10 ng/dose; fragment size <200 bp | qPCR/ddPCR |
| Plasmid backbone DNA | <1% of vector genome copies | qPCR |
What You'll Receive
| Deliverable | Description |
|---|---|
| Customized AAV vector design | Tailored to MPS IIIA pathology |
| Optimized SGSH gene construct | High-expression therapeutic cassette |
| CNS-targeting strategy | Delivery route + capsid selection |
| Preclinical validation data | In vitro and in vivo results |
| Regulatory-ready documentation | IND-supporting data packages |
Our End-to-End Development Workflow
-
Step 1: Project Consultation & Feasibility Assessment
We begin with an in-depth consultation to understand your therapeutic objectives, target patient population, and development timeline. Our experts evaluate the feasibility of your MPS IIIA gene therapy strategy, including SGSH gene selection and delivery approach. A customized project plan is then established, outlining technical milestones, regulatory considerations, and expected outcomes. This strategic foundation ensures alignment with both scientific and translational goals.
-
Step 2: Therapeutic Gene & Vector Design
Our scientists design an optimized SGSH gene cassette to achieve robust and sustained expression. This process includes codon optimization, promoter selection (ubiquitous or CNS-specific), and incorporation of regulatory elements such as enhancers and polyadenylation signals. We also evaluate options like self-complementary AAV constructs or co-expression with SUMF1 to enhance enzymatic activity. Each vector is tailored to maximize therapeutic efficacy while maintaining safety.
-
Step 3: AAV Capsid Selection & Engineering
Selecting the appropriate AAV serotype is critical for efficient CNS targeting. We assess clinically validated capsids such as AAV9 and AAVrh.10, as well as engineered variants with enhanced blood-brain barrier penetration. Capsid modifications can be implemented to improve tissue tropism, transduction efficiency, and immune evasion. This step ensures optimal biodistribution and therapeutic gene delivery to neurons and glial cells.
-
Step 4: Vector Construction & Production
Following design finalization, the recombinant AAV vector is constructed and produced using scalable, high-yield manufacturing platforms. Our production capabilities include research-grade processes to support preclinical and clinical development. Rigorous purification methods, such as affinity chromatography, ensure high vector purity and potency. Comprehensive quality control guarantees consistency and regulatory compliance.
-
Step 5: In Vitro Functional Validation
We perform a series of in vitro assays to confirm transgene expression and biological activity. These studies include evaluation of SGSH protein expression, enzymatic activity, and cellular uptake in relevant cell models. Transduction efficiency and vector potency are also assessed to ensure functional performance. The resulting data provide critical evidence supporting progression to in vivo studies.
-
Step 6: In Vivo Preclinical Evaluation
Preclinical studies are conducted in appropriate animal models to assess therapeutic efficacy and safety. Key evaluations include biodistribution, CNS transduction efficiency, and reduction of heparan sulfate accumulation. Behavioral and neurological assessments may also be performed to demonstrate functional improvement. These studies generate essential data for advancing the therapy toward clinical translation.
-
Step 7 Ongoing Collaboration & Translational Support
Beyond initial development, we offer continued scientific and technical support to accelerate your program's success. This includes data interpretation, process optimization, and technology transfer for large-scale manufacturing. We maintain close communication throughout the project to ensure flexibility and responsiveness to evolving needs. Our collaborative approach enables a seamless pathway from discovery to clinical application.
Why Choose Creative Biolabs for Your MPS IIIA Program?
Proven Track Record in CNS Gene Therapy
We have successfully supported >50 CNS gene therapy programs, including several for lysosomal storage disorders (MPS I, MPS II, MPS IIIB, CLN2, Gaucher). Our team has authored multiple peer-reviewed publications on AAV-mediated SGSH delivery.
Integrated Platform – One Vendor, Seamless Transition
From molecular cloning to IND filing, you work with a single project manager. No need to outsource vector production, animal studies, and toxicology separately.
Flexible Engagement Models
- Fee-for-service – pay per study
- Milestone-based collaboration – shared risk, royalty on back-end
- Licensing of our proprietary AAV capsids
Frequently Asked Questions (FAQ)
Q: What is the optimal route of administration for MPS IIIA gene therapy?
A: In our experience, ICV delivery in neonates provides the highest CNS transduction but requires a surgical procedure. For older patients, IT-Lumbar injection (lumbar puncture) or high-dose IV AAV9 (1e14 GC/kg) can achieve widespread brain distribution. We can help you compare routes in mouse and large animal models.
Q: Can you develop a gene therapy targeting both CNS and somatic organs?
A: Yes. We design dual-route studies: IV AAV9 for peripheral correction (liver, spleen, heart) plus IT or ICV for CNS. Alternatively, we can engineer a single AAV9 with enhanced BBB crossing to treat both compartments with one injection.
Q: Do you offer CRISPR-based approaches for MPS IIIA?
A: Absolutely. We have a separate genome editing platform that includes homology-directed repair (HDR) and base editing (ABE/CBE) to correct the common p.R74H mutation in patient-derived iPSCs. In vivo editing using AAV-CRISPR (e.g., SpCas9 with sgRNA) is also available.
Q: How do you address pre-existing immunity to AAV?
A: We screen NAb titers before enrollment. For high-titer subjects, we can: (i) use a different serotype (e.g., AAVrh10 or AAV-DJ), (ii) perform plasmapheresis, or (iii) co-administer immunomodulators (e.g., rapamycin). We also offer empty capsid competition strategies.
Q: Can you help with orphan drug designation (ODD) applications?
A: Yes. Our regulatory team has successfully filed ODDs for 6 rare disease gene therapies. We compile the necessary epidemiology data, mechanism of action rationale, and preclinical evidence.
Contact Us
If you have any questions about our vector design service, you can contact us by email or send us an inquiry to find a complete solution.
Reference
- Fajardo-Serrano A, Rico A J, Roda E, et al. Adeno-associated viral vectors as versatile tools for neurological disorders: focus on delivery routes and therapeutic perspectives. Biomedicines, 2022, 10(4): 746. https://doi.org/10.3390/biomedicines10040746.
- Nair K, Bhat A R. Applications of Gene Therapy in Dentistry: A Review Article. Journal of Health and Allied Sciences NU, 2023, 13(04): 445-452. 10.1055/s-0042-1759711.
- Distributed under Open Access license CC BY 4.0, without modification
