AAV Vector Design Services for Canavan's Disease
Recombinant adeno-associated virus (rAAV) is one of the most promising therapeutic delivery systems for gene therapy of the central nervous system (CNS). Creative Biolabs has rich experience and advanced platform in AAV vector design, which enable us to provide a wide range of AAV vector design services to meet our customers.
Introduction of Canavan's Disease
Canavan's disease (CD) is a rare lethal pediatric leukodystrophy characterized by extensive demyelination, edema, and progressive spongy degeneration of the CNS. This disease is caused by autosomal recessive genetic mutations in aspartoacylase (ASPA), a metabolic enzyme that deacetylates N-acetyl aspartic acid (NAA) to produce aspartic acid and acetate, and it was the first disorder approved for AAV gene therapy in the CNS. ASPA deficiency results in the accumulation of the substrate NAA in oligodendrocytes and subsequent hypomyelination/dysmyelination. Currently, traditional treatments for CD include the application of lithium, glyceryl triacetate, triheptanoin and oral N-acetyl-aspartate administration. Recently, it is evident that efficient gene therapy for CD would be an ideal treatment if begun at an early age.
Understanding Canavan Disease Mechanism
| Key Aspect | Description |
|---|---|
| Genetic Cause | Mutations in ASPA gene |
| Enzyme Deficiency | Loss of aspartoacylase activity |
| Pathological Effect | Accumulation of N-acetylaspartate (NAA) |
| Biological Outcome | Impaired myelin formation |
| Clinical Impact | Progressive neurodegeneration |
ASPA plays a critical role in breaking down NAA into acetate and aspartate, both essential for normal brain metabolism and myelin synthesis. When ASPA is dysfunctional, NAA accumulates in the central nervous system, leading to white matter degeneration and neuronal dysfunction.
AAV-based Gene Therapy for CD
Gene therapy is a promising clinical intervention for inborn errors like CD. Novel rAAV vectors represent a promising gene delivery platform because of their broad tropism, low immunogenicity, highly efficient and sustained gene transduction, and clinically proven track record in safety. It delivers the therapeutic gene to the CNS directly and provides long-term and functional correction of missing or mutated genes. Delivery of ASPA by rAAV is an effective system to achieve significant ASPA enzyme delivery in the CNS. The increase of ASPA activity indicates that the rAAV-mediated expression of the enzyme hydrolyzes NAA to aspartate and acetate, reducing NAA levels. Additionally, early intervention is also very important for the treatment of CD, because improvements in younger patients seem to be more obvious than those older patients. Currently, phase I clinical trials of delivering ASPA by rAAV2 to CD patients have been completed and the results showed that the treatment was well tolerated and no serious side effects occurred. Therapeutic benefits include reduced brain NAA accumulation, delayed progressive brain atrophy and less frequent seizures. Neurological examination revealed a significant improvement in motor function in the younger patients treated for CD, indicating the probable advantage of early-onset therapeutic intervention.
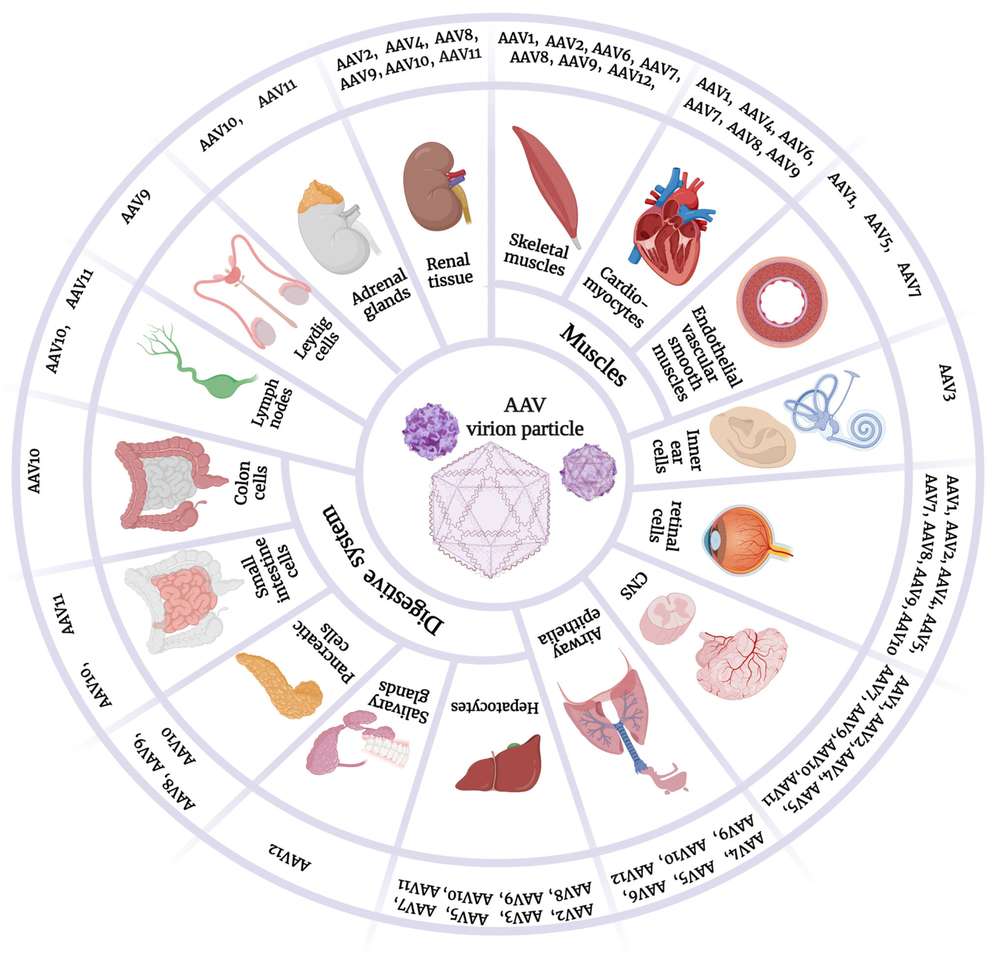 Figure 1. Variant tropisms of AAV serotypes.1
Figure 1. Variant tropisms of AAV serotypes.1
Our Services
rAAV-mediated gene therapy is the most promising therapeutic modality for CD and the absence of any major adverse event further supports the use of rAAV as clinical vectors for CD gene therapy. Eleven AAV serotypes have been identified to date. These serotypes differ in their tropism making AAV a very useful tool for preferentially transducing specific cell types, such as AAV1, AAV2, AAV4, AAV5, AAV8, AAV9 targeting CNS and AAV7, AAV8, AAV9 targeting liver. Differential transduction and tissue distribution of AAV can be achieved through the use of different AAV serotype capsids. In addition, the rational design of AAV expression vectors, vector titers and purity are also critical for gene therapy of CD. In terms of gene therapy for CD, Creative Biolabs provides tissue/cell-specific targeting AAV service, AAV vector design for gene expression, titration service of AAV vector, AAV vector purification service to improve the efficacy of gene therapy for CD and meet customer's project needs.
AAV Vector Engineering & Design Optimized for the CNS
A successful CD gene therapy begins with flawless vector design. We provide comprehensive de novo plasmid construction and optimization:
- Optimal Serotype Screening: In addition to traditional AAV2, we highly recommend and provide serotypes with superior BBB-penetrating capabilities, including AAV9, AAV-PHP.B, AAV-PHP.eB, and our proprietary next-generation neurotropic variants for whole-brain transduction.
- ASPA Sequence & Codon Optimization: Deep codon optimization tailored for human host cells to significantly enhance the translation efficiency and stability of the ASPA protein.
- Tissue-Specific Promoter Customization: Depending on your therapeutic strategy, we can configure ubiquitous promoters (e.g., CAG, CBh) or highly specific promoters (e.g., GFAP for astrocytes, MBP for oligodendrocytes) to minimize off-target effects and maximize expression precision.
Gene Delivery & Route of Administration Optimization
Crossing the BBB is a major hurdle in CNS therapeutics. Our in vivo pharmacology team possesses extensive neurosurgical expertise, supporting the testing and optimization of multiple delivery routes to determine the best clinical translation strategy:
- Intravenous (IV) Injection: Evaluating BBB penetration efficacy via systemic administration, suitable for projects utilizing novel, highly penetrant capsids.
- Intracerebroventricular (ICV) & Intrathecal (IT) Injection: Direct delivery of AAV into the cerebrospinal fluid to maximize drug concentration in the brain while minimizing peripheral (e.g., hepatic) immunotoxicity.
- Intraparenchymal Stereotaxic Injection: Precision delivery targeted to specific brain regions.
Comprehensive In Vitro & In Vivo Efficacy Evaluation
We maintain a robust repository of CD disease models (including Aspa knockout mouse models) to provide regulatory-compliant, exhaustive pharmacodynamic data for your candidates:
- Biomarker Quantification: Utilizing high-resolution LC-MS/MS to accurately detect and quantify the reduction of NAA levels in brain tissue, CSF, and blood—the most direct indicator of efficacy.
- Myelination Analysis: Visual evaluation of myelin thickness and the reversal of spongy degeneration using Immunohistochemistry (IHC, e.g., MBP staining), Transmission Electron Microscopy (TEM), and high-resolution small-animal MRI.
- Neurobehavioral Testing: Comprehensive assessment of motor function, coordination, and survival improvement using Rotarod, Open Field tests, and gait analysis.
Pre-clinical Safety, Toxicology & Biodistribution Studies
Ensuring candidate safety is critical for advancing to the IND stage. We provide GLP-aligned toxicological evaluations:
- Biodistribution & Viral Shedding: Utilizing qPCR/ddPCR to precisely map AAV vector abundance in target organs (brain sub-regions) versus non-target organs.
- Immunogenicity Assessment: Detecting neutralizing antibodies (NAb) and binding antibodies (TAb) against the AAV capsid, as well as cellular immune responses against the exogenous ASPA protein.
- Comprehensive Tox Screening: Hepatic enzyme evaluation, histopathology, and neurotoxicity observations.
Key Technical Advantages of Our Platform
01 Precision CNS Targeting
- Advanced capsid engineering
- Cell-type specific targeting (oligodendrocytes)
- Enhanced BBB crossing strategies
02 Disease-Focused Expertise
- Deep understanding of leukodystrophies
- Proven experience with CNS gene therapy
- Integrated biology + vector engineering
03 Flexible Development Models
- Standalone services
- Full-service partnerships
- Custom project design
Our Benefits
- Expert consultation at all stages of the project
- Strong tissue targeting with unique serotype and promoter portfolios
- AAV delivery in less than 2 months
- Highest quality standards and many options to fit your research needs
Our Collaboration Process
-
1. Scientific Consultation
We begin with a deep technical discussion to understand your program goals, timelines, and challenges. Our experts propose a customized development strategy tailored to your needs.
-
2. Project Planning
A detailed project roadmap is established, including milestones, deliverables, and timelines. We ensure full transparency and alignment from the start.
-
3. Execution & Iteration
Our multidisciplinary team executes the project with continuous optimization. Data-driven iterations ensure the highest probability of success.
-
4. Data Delivery & Reporting
You receive comprehensive reports with actionable insights, enabling informed decision-making at every stage.
-
5. Ongoing Support
We provide continued support for downstream development, including regulatory strategy and clinical transition.
Frequently Asked Questions (FAQ)
Q: Which delivery route do you recommend for CD gene therapy?
A: The optimal route depends on your chosen AAV serotype. For variants with strong BBB-crossing capabilities (like AAV9 or novel engineered capsids), Intravenous (IV) injection is a minimally invasive and highly translatable option. However, to achieve maximum parenchymal transduction while lowering the total required dose (thereby reducing hepatotoxicity), cerebrospinal fluid routes (ICV or IT) have shown superior, direct metabolic correction in pre-clinical and early clinical studies for CD. We can conduct parallel comparative studies for both routes.
Q: The packaging capacity of AAV is around 4.7 kb. Is the ASPA gene suitable for packaging?
A: Absolutely. The coding sequence (CDS) of the human wild-type ASPA gene is less than 1 kb. This leaves ample room for genetic engineering. We can utilize this space to incorporate more complex, highly specific promoters (such as longer endogenous promoters) or regulatory elements (like WPRE or optimized polyA tails) to maximize transgene expression efficiency.
Q: Can you provide specific animal models for CD efficacy evaluation?
A: Yes. We have deep partnerships with top-tier global animal model providers and possess robust in-house model generation capabilities. Beyond wild-type mice, we can facilitate the acquisition or custom generation (via CRISPR-Cas9) of Aspa knockout/mutant engineered mouse models. These models accurately replicate human CD pathology—specifically NAA accumulation and spongy leukodystrophy—serving as the gold standard for pharmacodynamic evaluation.
Q: How is Intellectual Property (IP) handled during our collaboration?
A: We operate strictly on a "Client-First" principle. As a pure-play R&D service provider, 100% of the ownership and IP for all customized sequences, design strategies, experimental data, and final products generated during the project belong exclusively to the client. Executing a strict Confidential Disclosure Agreement (CDA) is our prerequisite before initiating any scientific discussions.
Ready to Accelerate Your Canavan Disease Gene Therapy Pipeline?
Every day saved in early development is a step closer to changing the lives of pediatric patients. Do not let manufacturing bottlenecks or a lack of high-quality disease models delay your scientific breakthroughs. Entrust the complexities of AAV vector engineering and in vivo efficacy evaluation to us, empowering your team to focus on core scientific innovation. Whether you seek help customizing a vector for academic use or support for your industrial R&D, the technical experts at Creative Biolabs will help you from square one to discuss and plan your project in detail. For more details, please feel free to contact us.
Reference
- Issa S S, Shaimardanova A A, Solovyeva V V, et al. Various AAV serotypes and their applications in gene therapy: an overview. Cells, 2023, 12(5): 785. https://doi.org/10.3390/cells12050785 Distributed under Open Access license CC BY 4.0, without modification.
