Scientists at Creative Biolabs are expert in targeted delivery system research. We have a series of innovative and advanced platforms to provide fast and convenient liposome development services for our worldwide customers. Currently, we can provide affordable, high-quality ufasomes-based drug delivery services for diseases diagnosis and therapeutic with unbeatable rapid turnaround times.
Discomes are non-ionic surface active agents based discoidal vesicles, which are niosomes solubilized with non ionic surfactant solutions (polyoxyethylene cetyl ether class) and are used for ligand mediated drug targeting. Niosomes possess many advantages over other ocular drug delivery systems. Composed of biodegradable and non-immunogenic materials, they are more chemically stable than liposomes, with lower production cost. They improve drug performance via better physical stability and controlled delivery at the site of action. Niosomes harbor certain attributes related to their potential use as vehicles for ocular drug delivery, including a large enough size to resist drainage by reflex tearing and an eye blinking, an irregular shape to properly fit into the cul-de-sac and lodge on the eye surface. Ocular niosomes are thermoresponsive ideally in order to release drug content in a controlled, yet timely, manner before being flushed by blinking and nasolacrimal drainage.
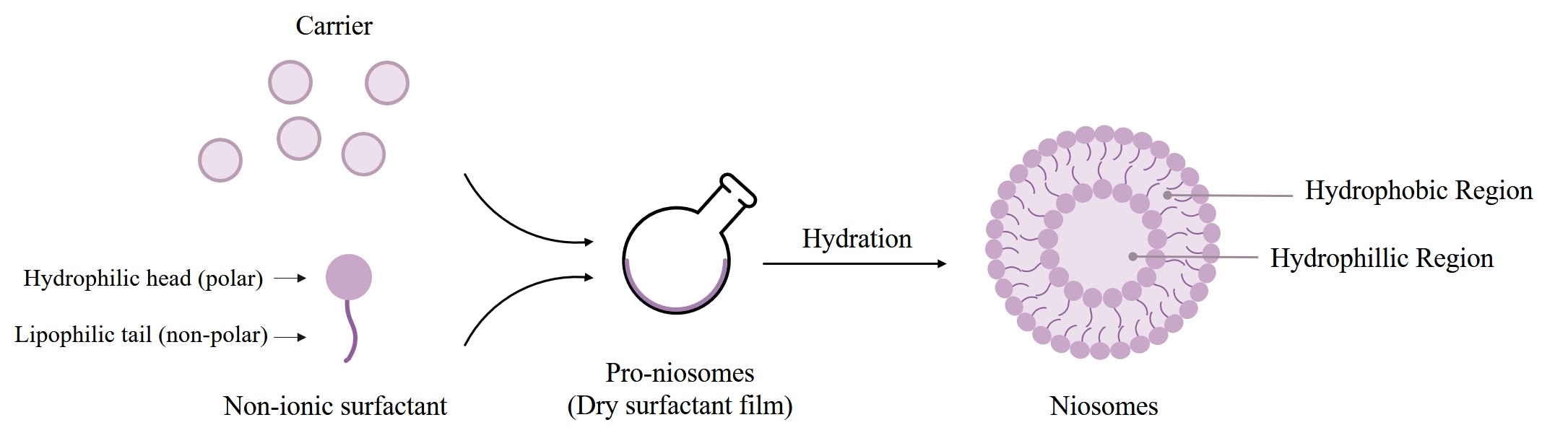 Fig.1 Niosomes formation from provesicular forms by hydration.
Fig.1 Niosomes formation from provesicular forms by hydration.
Discomes are a type of niosomes with advantages similar to polyhedral niosomes. By contrast, discomes consisting of low cholesterol concentration less than 30% mol/mol are prepared by incubating the preformed spherical niosomes in cholesteryl poly-24-oxyethylene ether at 75◦C for 1h, achieving the formation of large (11-60μm) and multifaceted vesicular systems. The inherent large size is considered to prevent discomes from being rapidly washed out by tear dynamics. Also, their nonuniform spherical structure can render a better fit on the ocular surface. For instance, discomes have achieved higher encapsulation efficiency (EE%) and ocular bioavailability. These discomes are reported as enhanced and controlled ocular delivery systems. There have been reports on discomes acting as an ocular delivery system for timolol maleate. The prepared discomes have a relatively large quantity of timolol and enhance the ocular bioavailability compared to timolol maleate solution. One possible reason for such scarcity is the necessity for a relatively high temperature during discome preparation that might influence the chemical stability of some thermolabile therapeutic agents. On the contrary, spherical niosomes have been found to have more stable membranes than polyhedral ones due to the presence of cholesterol.
Along with years of rich experience in the field of drug delivery system discovery and design, Creative Biolabs has successfully finished a number of projects for our customers all over the world. Currently, we provide high-quality discomes-based drug delivery formulation and related services. We are now confident in offering the customized proposal to meet your specific needs. If you are interested in our service, please do not hesitate to contact us for more details.
 For Research Use Only. Not For Clinical Use
For Research Use Only. Not For Clinical UseSupports
Online Inquiry