The advent of liposomes in the field of pharmaceuticals and drug delivery has been a veritable revolution. These versatile tools are employed in a wide range of medical applications due to their biocompatibility and ability to facilitate the delivery of a variety of therapeutic agents. The importance of liposomes extends beyond their physical structure, delving into the territory of intricate in vitro experiments that determine their behavior, usefulness, and implications.
Central to the utility of liposomes is the in vitro experiment mechanism that elucidates their action. During these experiments, liposomes are often subjected to drug loading processes, including active and passive methods. These encapsulate the therapeutic agents within the liposomal membrane or its aqueous interior, depending on the solubility and preferential localization of the drug. A critical step in these experiments is the use of various methodologies to determine the percentage of drug entrapment and the drug to lipid ratio.
Essential to understanding the functioning of liposomes is the incorporation of dialysis, ultracentrifugation, and chromatography techniques to measure encapsulated drug concentration and removal of free drug molecules. These in vitro experimental methodologies allow for insight into the kinetics of drug release, stability of liposomal formulation, behavior in biological fluids, and cellular uptake. Several factors are analyzed, including temperature, pH level, and agitation rate, which significantly influence drug diffusivity and liposomal integrity.
In vitro experiments of liposomes play a fundamental role in developing drug delivery systems. The appropriate in vitro drug release test from liposomal products is vital to predict their in vivo performance. Techniques such as dialysis sac, sample-and-separate, reverse dialysis, and continuous flow methods provide predictions of drug stability, bioavailability, and potential toxicity.
These tests permit the degree of control required to observe the variables involved, predominantly in new drug development. They are fundamental in determining drug release behavior, which informs dose design and delivery mechanisms. A successful in vitro drug release test is paramount in drug formulation, especially in the advancement of therapeutics that are insoluble or demonstrate poor bioavailability. Additionally, these tests offer a powerful tool in the evaluation of liposomal stability and degradation over predetermined periods.
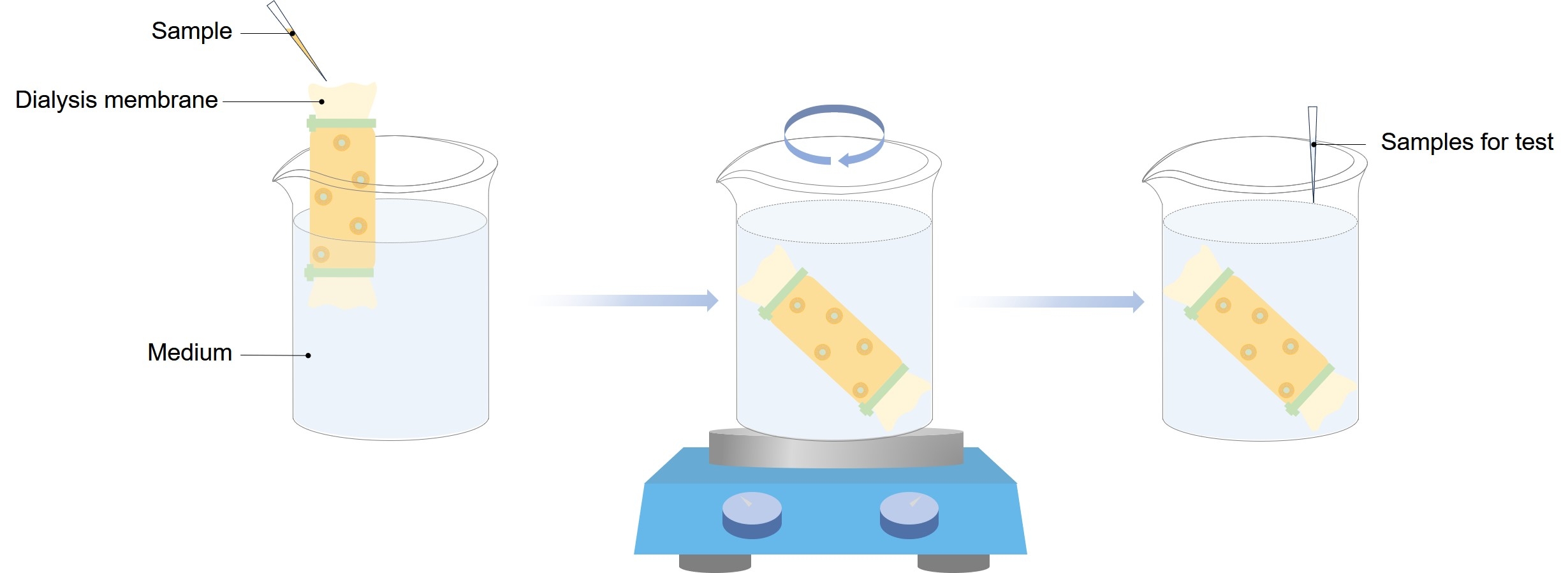 Fig.1 Graphical illustration of the process of dialysis.
Fig.1 Graphical illustration of the process of dialysis.
In vitro experiments with liposomes offer numerous advantages to the field of drug delivery and pharmaceuticals. One of the significant advantages of these experiments is that they provide key data without requiring animal or human subjects. Not only does this greatly reduce ethical complications, but it also allows for more controlled and consistent experimental conditions.
These experiments contribute immensely to understanding the stability and drug release rates of liposomal formulations, thereby reducing the risk and uncertainty in subsequent in vivo experiments. More importantly, they allow the testing of multiple variables and conditions that would be unfeasible or highly complex in an in vivo setting. These range from temperature and pH factors to the impact of specific enzymes or chemicals.
In vitro experiments of liposomes elucidate the mechanism of action of these drug-delivery vehicles, providing critical information on drug encapsulation, release, and stability. These experiments are irreplaceable in translating theoretical benefits into real-world applications, ensuring the effectiveness and safety of liposomal formulations in drug delivery.
Creative Biolabs proudly offers a series of custom liposomal services and products. Please feel free to contact us for more details.
 For Research Use Only. Not For Clinical Use
For Research Use Only. Not For Clinical UseSupports
Online Inquiry