The systemic administration of drugs is a widely employed strategy in the treatment of diseases that affect the entire body. Intravenous injection provides a direct pathway for delivering therapeutic agents, like clodronate liposomes, into the bloodstream. We will delve into the concept of systemic clodronate liposome administration, exploring its mechanism of action, diverse applications, and potential advantages.
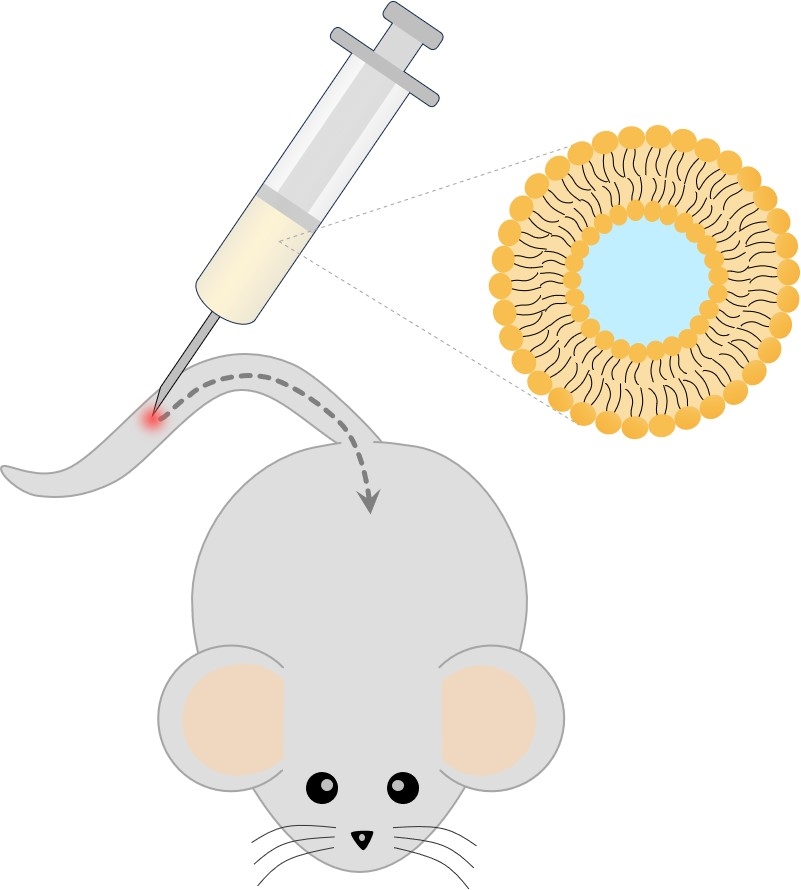 Fig.1 Systemic administration. (Creative Biolabs Original)
Fig.1 Systemic administration. (Creative Biolabs Original)
Liposomes encapsulate clodronate, a bisphosphonate, within their structure. Upon intravenous injection, they are engulfed by monocytes and macrophages through a process called phagocytosis. The gradual release of clodronate inside these cells leads to their depletion, either by triggering apoptosis or disrupting intracellular activities. By specifically targeting monocytes and macrophages, which play pivotal roles in inflammatory responses and immune regulation, clodronate liposomes provide a promising therapeutic approach for various pathological conditions.
In the context of inflammatory diseases, such as rheumatoid arthritis and multiple sclerosis, the use of clodronate liposomes has exhibited encouraging outcomes. By depleting inflammatory macrophages, these liposomes assist in mitigating inflammation and improving disease prognosis.
Moving on to the realm of cancer treatment, tumor-associated macrophages (TAMs) exert a detrimental influence on tumor growth and immunosuppression. To combat this, researchers have investigated the systemic delivery of clodronate liposomes as an adjuvant therapy. By selectively eliminating TAMs, clodronate liposomes possess the potential to enhance the efficacy of conventional cancer treatments and augment anti-tumor immune responses.
Additionally, clodronate liposomes have been explored in the field of organ transplantation to reduce rejection rates. This is particularly important considering the crucial role played by macrophages and other immune cells in mediating transplant rejection. By modulating the immune response through the depletion of these immune cells, clodronate liposomes may aid in prolonging graft survival and improving overall transplantation outcomes.
One notable advantage of systemic clodronate liposome administration is its targeted approach. By specifically targeting monocytes and macrophages, the off-target effects on other cell types can be minimized. This enhances the overall specificity and efficacy of the treatment approach.
Furthermore, clodronate liposomes possess a favorable safety profile and are generally well-tolerated. This is thought to be due to their encapsulation within liposomes, which safeguards the drug from degradation and reduces its systemic toxicity. This attribute significantly contributes to the therapeutic feasibility of clodronate liposomes.
Moreover, the versatility of clodronate liposomes must be emphasized. These liposomes can be easily modified to enhance drug delivery and can also be combined with other therapeutic agents, making them instrumental tools in the realm of personalized medicine.
Systemic administration of clodronate liposomes via intravenous injection represents a powerful therapeutic approach for selectively depleting macrophages. This strategy holds immense potential for the treatment of various diseases characterized by dysfunctional macrophage activation. Nonetheless, further research is warranted to optimize the administration protocols and explore novel applications of this approach.
Creative Biolabs is confident in providing liposomal services and products, please feel free to contact us.
 For Research Use Only. Not For Clinical Use
For Research Use Only. Not For Clinical UseSupports
Online Inquiry