Intravaginal administration is a route of drug delivery that involves introducing medications directly into the vagina. It offers several advantages, including increased local bioavailability and reduced systemic side effects. Clodronate liposomes, a form of liposomal drug delivery system, have shown promising results after intravaginal administration.
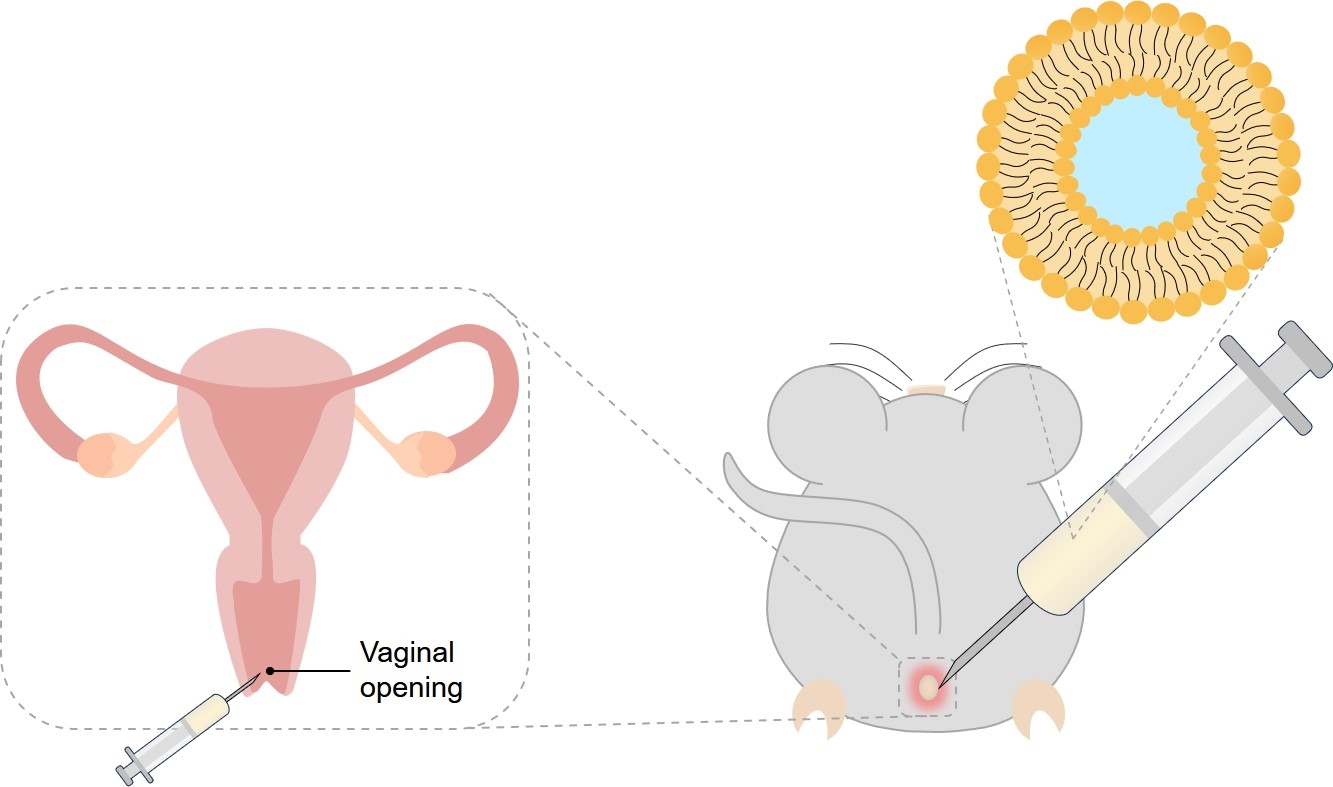 Fig.1 Intravaginal administration.
Fig.1 Intravaginal administration.
The mechanism of action underlying the intravaginal administration of clodronate liposomes involves the utilization of liposomal vesicles to encapsulate and deliver clodronate to the vaginal tissue. By encapsulating clodronate within liposomes, the drug can be effectively targeted and delivered to the vaginal epithelium and underlying tissues.
Upon intravaginal administration, clodronate liposomes interact with the vaginal mucosa, promoting the uptake of liposomal contents by local immune cells, predominantly macrophages. Once taken up, clodronate is released from the liposomes, thus inhibiting the activity of macrophages through the induction of apoptosis. By selectively targeting and depleting macrophages in the vaginal tissue, intravaginal administration of clodronate liposomes may help modulate local immune responses and inflammation.
Management of vaginal infections: Intravaginal administration of clodronate liposomes has shown efficacy in the management of various vaginal infections. By targeting and depleting macrophages, clodronate liposomes can help reduce the inflammatory response associated with infections, potentially improving treatment outcomes.
Treatment of vaginal atrophy: Vaginal atrophy is a common condition characterized by the thinning, drying, and inflammation of the vaginal walls. Intravaginal administration of clodronate liposomes has been investigated as a potential treatment for vaginal atrophy. By modulating local immune responses and reducing inflammation, clodronate liposomes may help alleviate the symptoms associated with this condition.
Improved drug delivery: Intravaginal administration of clodronate liposomes allows for targeted drug delivery to the vaginal tissue. This route of administration offers increased local bioavailability, ensuring that the drug reaches the intended site of action in higher concentrations. Consequently, lower doses of clodronate may be needed, minimizing the risk of systemic side effects.
Reduced systemic side effects: By delivering clodronate directly to the vaginal tissue, intravaginal administration of clodronate liposomes minimizes systemic exposure to the drug. This reduces the likelihood of systemic side effects commonly associated with oral or intravenous administration of bisphosphonates, such as gastrointestinal disturbances or renal toxicity.
Convenience and patient compliance: Intravaginal administration of clodronate liposomes offers a convenient and patient-friendly method of drug delivery. It can be self-administered by the patient, eliminating the need for healthcare professionals, and reducing the overall cost of treatment. This may improve patient compliance and lead to better treatment outcomes.
With further research and development, intravaginal administration of clodronate liposomes may emerge as a valuable therapeutic option in women's healthcare.
Creative Biolabs provide a series of high-quality liposome services and products. If you are interested, please contact us for more details.
 For Research Use Only. Not For Clinical Use
For Research Use Only. Not For Clinical UseSupports
Online Inquiry