It has been a long journey from Pliny the Elder (23-79 AD) to the first injectable nanomedicine approved in 1997. This is a journey out of intellectual curiosity, which finally promoted the birth of liposome technology. Liposome technology has been studied since the 1960s. As it is looking for a way to promote the delivery of active substances to specific organs/tissues, it has attracted the attention and time of many scientists.
Liposomes are unilamellar or multilamellar spherical vesicles, mainly containing phospholipids from plant or animal sources. They were first discovered by A.D. Bangham in 1965. When phospholipids were dispersed in aqueous medium, they observed spontaneous self-assembled closed vesicles with concentric lipid bilayers and hydrophilic cores.
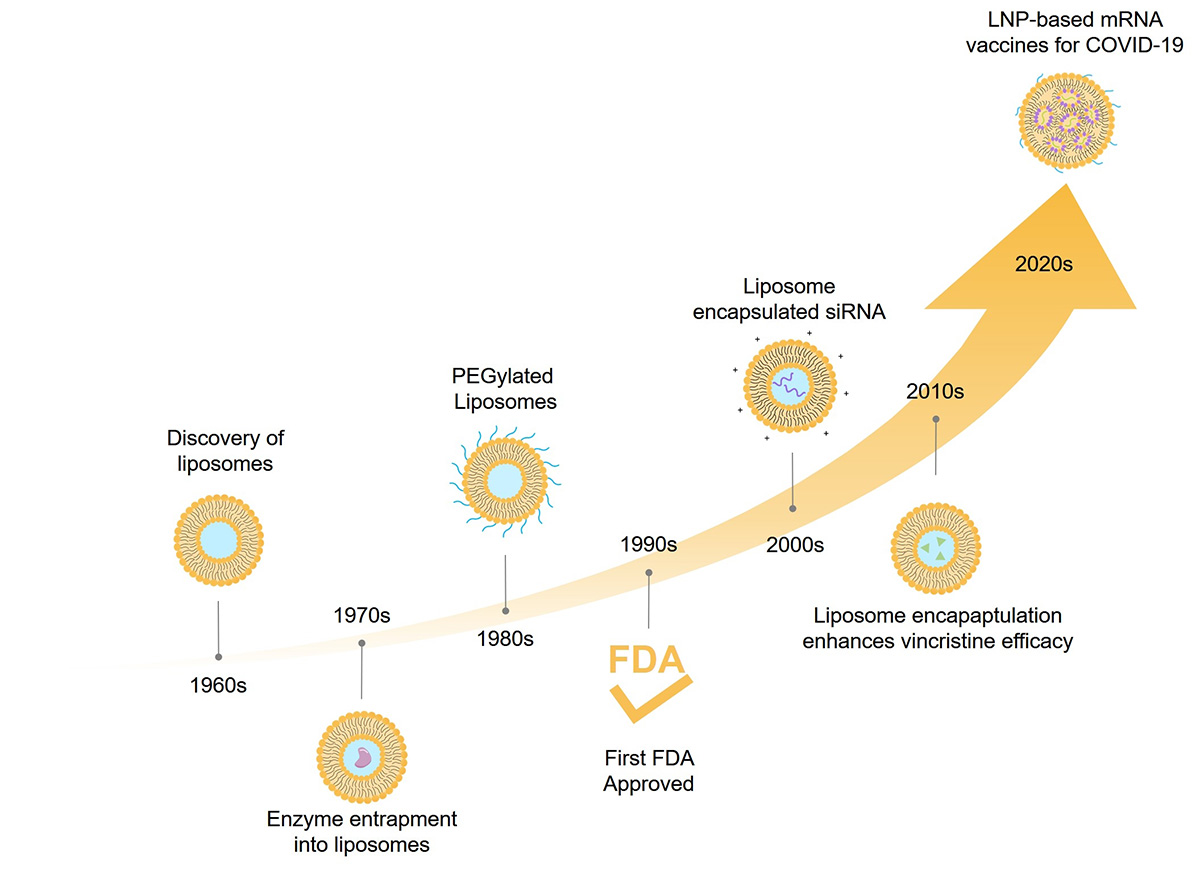 Fig.1 History of liposome.
Fig.1 History of liposome.
Since then, the history of liposomes can be divided into three periods: genesis, middle age, and modern era.
Genesis (1968-75): The physical and chemical characteristics of liposomes have been studied. During this period, liposomes were used to study the properties of biological membranes, and a thin lipid membrane hydration method was developed to prepare liposomes.
Middle age (1975-85): During this period, the advantages, stability and interaction characteristics, physical and chemical properties of liposomes, as well as their interaction with cells and their behavior in vivo were studied. In addition, various methods of preparing liposomes have been discovered.
Modern era (after 1985): Today, liposomes are used in different scientific fields, such as biophysics (characteristics of cell membranes and channels), mathematics, biochemistry (functions of membrane proteins), theoretical physics (topology of two-dimensional surfaces floating in a three-dimensional continuum) and biology (excretion, cell function, signal transduction, gene delivery, and function).
The name of liposome is derived from two Greek words: "Lipos" means fat, and "Soma" means body. Liposomes are self-assembled closed spherical structures composed of one or more concentric curved lipid bilayers and cholesterol. They range in size from 20 nm to many microns. Because of the amphiphilicity of the lipids used in liposomes, they are candidates for drug delivery. When the amphiphilic molecules come into contact with the aqueous environment, they first dissolve and then aggregate to a certain concentration by increasing the entropy of the system.
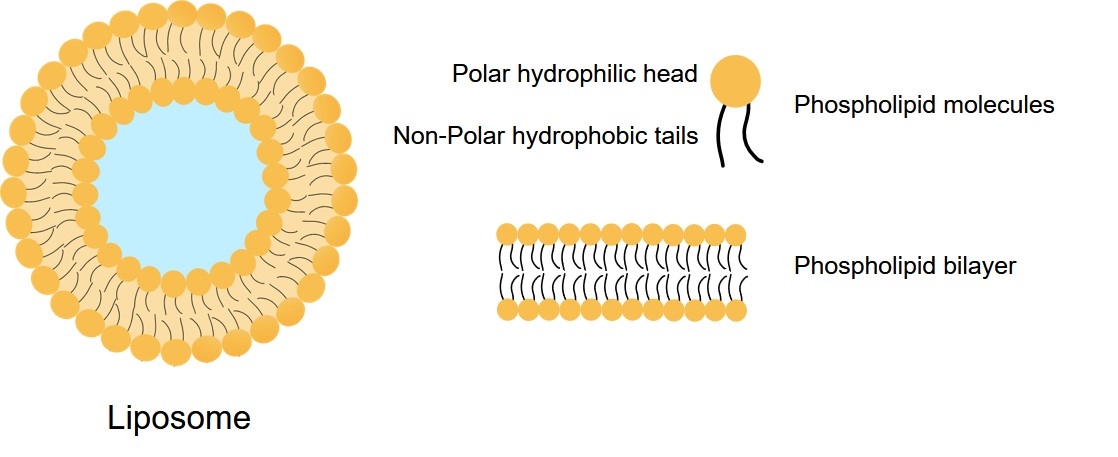 Fig.2 Structure of liposome.
Fig.2 Structure of liposome.
Because of its biodegradable, biocompatible, non-toxic, and non-immunogenic components, liposomes account for a large proportion of nanotherapy in clinical stage. The first liposomal formulation of the cytotoxic anticancer drug doxorubicin (DOX) was launched in the early 1990s. Currently, liposomes have been successfully used in all imaginable drug delivery methods, and their applications in solving various biomedical problems are also steadily increasing.
The amphiphilic phospholipid bilayer of liposome is very similar to mammalian cell membrane, which realizes the effective interaction between liposome and cell membrane and subsequent effective cell uptake. In addition, ligands can be added to liposomes to improve the efficiency and specifically target damaged cells, so as to improve the pharmacokinetics of liposomes and their ability to pass through the target membrane, achieve high concentration in cells, reduce toxicity and improve the therapeutic effect.
Creative Biolabs has strong and extensive expertise and years of experience in the field of drug delivery and liposome development services. For more details about our services, please directly contact us.
 For Research Use Only. Not For Clinical Use
For Research Use Only. Not For Clinical UseSupports
Online Inquiry