The utilization of liposomes has been widely acknowledged as a revolutionary strategy in the drug delivery system. Among various modes of administration, the intraperitoneal administration of liposomes has gained popularity due to its benefits and applications in several scientific fields, particularly medical and pharmaceutical research.
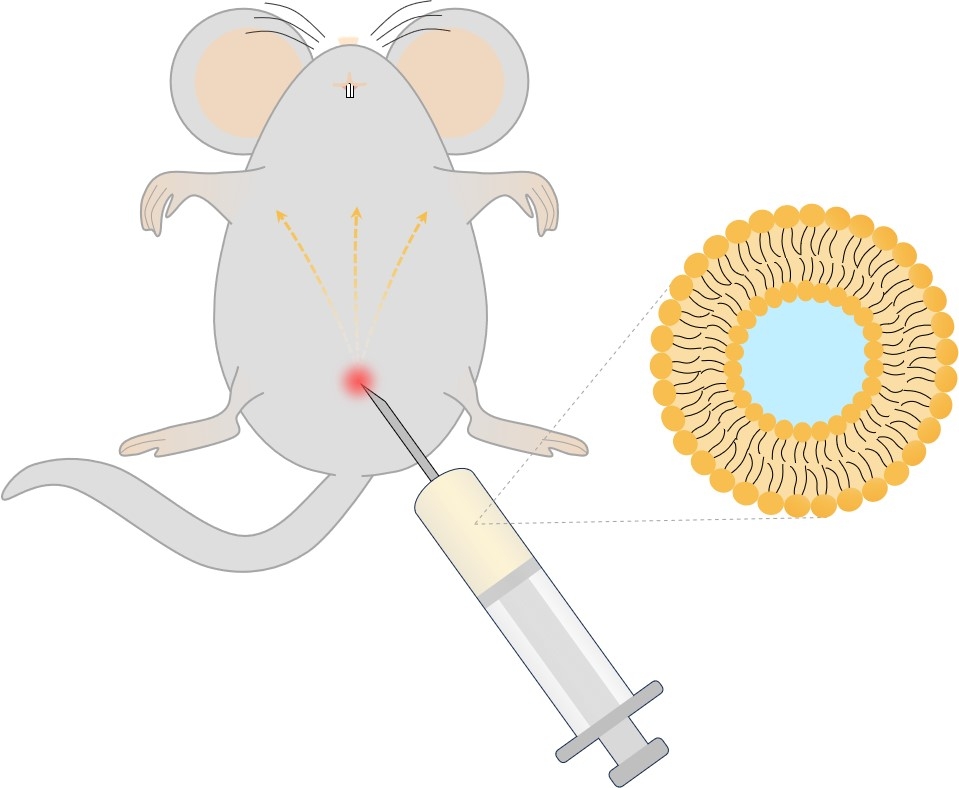 Fig.1 Intraperitoneal administration.
Fig.1 Intraperitoneal administration.
Intraperitoneal administration of liposomes employs a direct delivery technique where liposomes carrying therapeutic drugs are injected into the peritoneal cavity. The liposomes have an amalgamation of lipids that help segregate the drug from the body fluids, allowing for extended retention within the cavity. Liposomes as drug carriers can encapsulate both hydrophobic and hydrophilic drugs and deliver them to the specific site in a controlled, targeted manner, reducing the systemic side-effects.
Designed to mimic the natural biological membranes, liposomes passively diffuse across the peritoneal lining. Additionally, the small size of the liposomes allows for better penetration of therapeutic drugs. The fusion of liposomes and other peritoneal cells results in the direct delivery of the encapsulated drug, initiating the process of exerting its therapeutic effects in a local and systematic context.
The various types of liposomes used in intraperitoneal administration are primarily dictated by the nature of the therapeutic drug encapsulated and the target site for drug delivery. The key types include multilamellar vesicles (MLV), unilamellar vesicles (ULV), and stealth liposomes.
Multilamellar vesicles, with their multiple concentric bilayers, encapsulate a larger quantity of drugs, making them suitable for the delivery of large doses. Unilamellar vesicles, with a single lipid layer, are known for their ability to release drugs quickly at the targeted site. Stealth liposomes, embedded with PEG (PEGylated Lipids) , prolong drug circulation time and delay clearance from the body tissues, enhancing the drug's therapeutic efficacy.
The intraperitoneal administration of liposomes plays a crucial role in a broad array of applications in biomedical research. Notably, it holds great promise in the treatment of diseases localized within the peritoneal cavity, such as peritoneal carcinomatosis, a condition characterized by the spread of malignant cells in the peritoneal cavity. By employing intraperitoneal administration of liposomes, the encapsulated chemotherapeutic drugs can be administered directly to the abdominal cavity, enhancing its therapeutic efficacy and minimizing systemic toxicity.
Additionally, this approach has also proven effective in pain management, immunotherapy, and combating severe abdominal infections. The targeted, localized drug delivery system allows for the optimal use of therapeutic drugs, thus enhancing the treatment outcomes.
Intraperitoneal administration of liposomes offers several advantages over traditional drug delivery methods. By allowing direct drug delivery to the peritoneal cavity, it enhances the absorption and bioavailability of the encapsulated drug, improving the drug's therapeutic efficacy and lowering the frequency of administrations.
This method also exhibits a favorable distribution profile. High concentrations of the therapeutic drug are maintained at the target site for prolonged periods, while systemic exposure is limited, reducing the risk of adverse effects. Furthermore, the ability to deliver high doses of the drugs directly to the affected area results in a significant reduction in systemic toxicity, improving patient outcomes.
The intraperitoneal administration of liposomes represents an insightful breakthrough in the medical field, stepping up traditional approaches to drug delivery and therapy.
At Creative Biolabs, we offer comprehensive liposome products and services. Please contact us for more information.
 For Research Use Only. Not For Clinical Use
For Research Use Only. Not For Clinical UseSupports
Online Inquiry