Pseudotyping Service of Lentiviral Vectors with Hepatitic C Virus
Lentiviral vectors (LV) have been widely used as gene therapy vectors for the treatment of multiple acquired and inherited diseases. Particularly, glycoproteins derived from viruses that cause specific infection could be useful for gene therapy of related diseases. As a leading company in the field of gene therapy, Creative Biolabs has developed advanced platforms for LV construction and optimization. Our experienced scientists are happy to help LV pseudotyping for targeting numerous cells, such as CD81+ cells.
What is Hepatitis C Virus (HCV)?
HCV is a positive-stranded RNA virus classified in the family Flaviviridae. Infection is often associated with chronic disease, sometimes resulting in liver cirrhosis and hepatocellular carcinoma. The principal site of HCV replication is liver. Other than hepatocytes, HCV also infects PBMCs, such as B cells, T cells, monocytes/macrophages, and dendritic cells. The initiation infection of a virus to a target cell is usually determined by an interaction between the viral GPs and specific cell-surface receptor(s). Investigations of such interaction(s) will provide vital information about the host range and cellular or tissue tropism of a virus. However, the lack of in vitro systems for HCV propagation hampered biological and physicochemical studies on the virion and its mechanism(s) of cell entry.
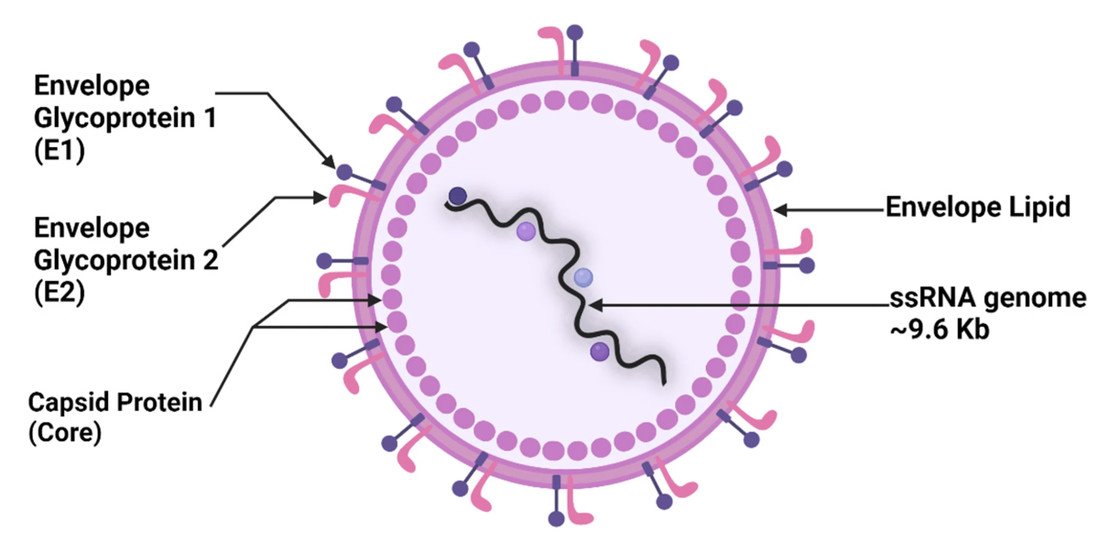 Figure 1. Structure of HCV. E: envelope glycoprotein; ssRNA: single-stranded RNA.1
Figure 1. Structure of HCV. E: envelope glycoprotein; ssRNA: single-stranded RNA.1
LV Pseudotypes Bearing HCV-derived GPs
HCV encodes two putative envelope glycoproteins (GPs), E1 and E2, which are believed to be type I integral transmembrane proteins. Recently, a major technical advance in the field has been the discovery that unmodified HCV envelope glycoproteins can pseudotype retroviral particles and mediate entry into target cells. This model seems to authentically replicate the early steps of the HCV life cycle, enabling detailed studies of HCV tropism and entry into target cells. Especially, vesicular stomatitis virus (VSV) and HIV based pseudotypes have been confirmed infectious for the human hepatoma cell lines Huh-7 and PLC/PR5. These pseudotype viruses are invaluable to investigate the mechanism of HCV entry and evaluate therapeutics targeting the HCV GP-cell interaction.
Services at Creative Biolabs
Our specialized pseudotyping service equips researchers with high-quality, targeted HCV-pseudotyped lentiviral vectors, bridging the gap between sophisticated vector technology and specific research needs in hepatology, virology, and immunology.
HCV-Pseudotyped Lentiviral Vectors
We offer specialized services for the production of high-titer, research-grade lentiviral vectors pseudotyped with HCV envelope glycoproteins. Our process involves molecular cloning of HCV E1 and E2 genes (from various genotypes as required), co-transfection with lentiviral packaging systems, and concentration/purification of the viral particles. We optimize glycoprotein expression and vector production protocols to maximize viral titer, infectivity, and specificity.
Target Cells for HCV Pseudotyping
HCV-pseudotyped lentiviral vectors (HCVpp) primarily target cells expressing the necessary HCV entry receptors, including:
- Primary human hepatocytes
- Various hepatoma cell lines (e.g., Huh-7, HepG2)
- Other engineered cell lines expressing HCV receptors
- This targeted delivery is invaluable for research on hepatitis C infection, liver-specific gene function studies, the development of gene therapies for liver disorders, and the investigation of mechanisms of hepatotropic pathogen entry.
Production and Purification
Our production pipeline is designed to maintain the structural integrity of the delicate HCV E1/E2 glycoprotein complex. We employ a multi-step purification process to remove cellular debris, inflammatory cytokines, and residual plasmid DNA.
| Phase | Description | Key Outcome |
|---|---|---|
| Transfection | High-efficiency PEI or Lipofectamine-mediated triple/quad-plasmid transfection in HEK293T cells. | Robust viral particle assembly. |
| Harvesting | Supernatant collection at optimized time points (24h, 48h, 72h). | Maximized raw viral yield. |
| Concentration | Ultracentrifugation or Tangential Flow Filtration (TFF). | 100x to 1000x concentration increase. |
| Purification | Sucrose gradient or Ion-exchange chromatography. | Removal of impurities and "empty" particles. |
| Formulation | Buffer exchange into PBS or specialized cryoprotectants. | High stability for long-term storage at -80°C. |
Vector Design & Strategy
A successful pseudotyping project begins with a sophisticated design strategy. We don't just "package" vectors; we engineer them for optimal performance. Our approach focuses on the synergistic interplay between the lentiviral backbone and the HCV envelope.
- Codon Optimization: We use proprietary codon-optimized E1/E2 sequences to achieve high-level expression in packaging cells, resulting in significantly higher viral titers.
- Backbone Flexibility: Choose from 2nd- or 3rd-generation lentiviral systems based on your safety requirements and payload size.
- Promoter Customization: We offer a variety of internal promoters (e.g., CMV, EF1α, PGK) or liver-specific promoters (e.g., Albumin, ApoE) to ensure the transgene is expressed exactly where and when you need it.
Service Features and Capabilities
- Custom Pseudotyping: We can pseudotype HCV E1/E2 with sequences from different genotypes or specific clinical isolates to meet your research needs.
- Flexible Backbone: Your gene of interest (reporter, therapeutic, shRNA, etc.) can be packaged into our optimized lentiviral backbone.
- Quality Control: Every batch undergoes rigorous QC testing, including titer determination (by qPCR or functional assay), sterility tests, and confirmation of HCV glycoprotein incorporation via entry inhibition assays with specific antibodies.
- Biosafety: All work is conducted in certified BSL-2 facilities using replication-incompetent systems.
Collaboration Workflow
- Consultation: Discuss your research goals, target genotypes, and transgene requirements.
- Vector Construction: Our team designs and clones the specific HCV E1/E2 and gene-of-interest plasmids.
- Production & Packaging: Large-scale transfection and vector harvesting in a controlled environment.
- Quality Verification: Rigorous QC testing for titer, purity, and hepatocyte-specific entry.
- Delivery: Secure, temperature-controlled shipping of ready-to-use viral aliquots.
Functional QC Suite – Proof-of-Performance Delivered with Every Lot
Flow-Cytometry-Based MOI Matrix in Your Cells
Choosing the correct Multiplicity of Infection (MOI) is critical for experimental success. Our standard functional titer (IU/mL) is determined on reference cell lines. To ensure optimal results in your unique cellular model, we offer a tailored MOI Matrix Analysis. We provide a detailed report including:
- Flow cytometry plots and graphs.
- The transduction efficiency (%) vs. MOI curve.
- Recommendation of the ideal MOI range to achieve your desired outcome (e.g., >70% transduction for population studies, or lower MOI for single-cell cloning).
- Assessment of potential cytotoxicity at higher MOIs.
ddPCR Integration Profiling ± Clonal Expansion
For applications requiring stable genomic integration and long-term expression—such as generating engineered cell lines or functional genomics studies—verifying vector copy number (VCN) is essential. We utilize digital Droplet PCR (ddPCR) for absolute, sensitive quantification of integrated vector genomes.
In Vivo Biodistribution Pilot (On Request)
For research paving the way toward preclinical or therapeutic applications, understanding vector biodistribution is paramount. While standard HCVpp targets hepatocytes in vitro, it’s in vivo tropism can be influenced by the route of administration and physiological factors. We offer a collaborative, small-scale pilot study to assess the preliminary biodistribution of your HCVpp vector in a relevant animal model (e.g., mouse).
Frequently Asked Questions
Q: What are the primary advantages of using lentiviral vectors for liver-targeted gene therapy?
A: Lentiviral vectors offer a significant edge due to their ability to integrate directly into the host genome, providing a permanent genetic fix that is passed down to daughter cells during hepatocyte division. Unlike AAVs, which primarily remain episomal and can be "diluted" as liver cells regenerate, LVs ensure life-long transgene expression. Furthermore, their large packaging capacity enables the delivery of complex genetic circuits or significant therapeutic genes that other viral systems cannot accommodate.
Q: How does the HCV pseudotyping process specifically enhance targeting and research safety?
A: By replacing the standard VSV-G envelope with HCV E1/E2 glycoproteins, the vector is "reprogrammed" to enter cells only through specific receptors like CD81 and SR-BI, which are highly expressed on hepatocytes. This mimics the natural entry pathway of the Hepatitis C virus without using the actual infectious viral genome. Consequently, researchers can study viral entry and neutralization in a BSL-2 environment, significantly reducing the biosafety risks and stringent regulatory requirements associated with handling live HCV.
Q: What is the typical lead time and process for submitting a custom HCV-pseudotyped project?
A: The process begins with a technical consultation to finalize your vector map and desired HCV genotype, followed by a formal quotation. Once the design is approved, the typical production cycle—including plasmid construction, HEK293T packaging, and multi-step purification—takes approximately 4 to 6 weeks, depending on the scale. We provide regular milestone updates, and the project concludes with the delivery of high-titer, purified viral aliquots, along with a comprehensive Certificate of Analysis (CoA) detailing all functional QC metrics.
Q: Can these vectors be used for high-throughput screening of antiviral compounds or antibodies?
A: Absolutely; our HCV-pseudotyped lentiviral vectors are optimized for high-throughput applications, especially when engineered with reporter genes such as Luciferase or GFP. They serve as an ideal platform for screening large libraries of neutralizing antibodies or small-molecule entry inhibitors, providing a clear, quantifiable readout of inhibitory activity. Because our production process ensures high batch-to-batch consistency and minimal background noise, the resulting data is highly reproducible and suitable for statistical validation in drug discovery pipelines.
Q: How do you ensure the purity and safety of the vectors, especially for in vivo applications?
A: Every lot undergoes a rigorous purification sequence involving Tangential Flow Filtration (TFF) and chromatography to eliminate host cell proteins, residual DNA, and endotoxins. For in vivo grade vectors, we guarantee endotoxin levels below 10 EU/mL and perform sterile filtration to ensure the highest safety profile for animal studies. Additionally, our use of 3rd-generation self-inactivating (SIN) transfer plasmids ensures the vectors are replication-deficient, thereby preventing the risk of unintended viral spread within the target organism.
Connect with Us Anytime!
To meet every client's specific requirement, Creative Biolabs has successfully established a top technology platform for lentiviral vector construction. Our professional scientists are able to design and optimize a full package of lentiviral vectors bearing numerous heterologous GPs, which including but not limited to HCV glycoprotein. Particularly, we have established stable cell lines, such as HEK293T and HEK293 cells, for LV vectors production. We are dedicated to optimizing lentivirus vectors to promote the development of global customers' projects. For more detailed information, please feel free to contact us or directly send us an inquiry.
Reference
- Le D H H, Kanokudom S, Nguyen H M, et al. Hepatitis C Virus—Core Antigen: Implications in Diagnostic, Treatment Monitoring and Clinical Outcomes. Viruses, 2024, 16(12): 1863. https://doi.org/10.3390/v16121863 Distributed under Open Access license CC BY 4.0, without modification.
