Pseudotyping Service of Lentiviral Vectors with Sendai Virus
Targeting vectors pseudotyped with distinct viral envelope proteins that influence cell tropism and transfection efficiency has been treated as a useful tool in the field of gene therapy. Up to now, numerous lentivirus vectors pseudotyped with different glycoproteins have been constructed for the treatment of multiple diseases. As a pioneer company in this field, Creative Biolabs is capable of offering comprehensive lentiviral vectors construction and optimization services for worldwide customers. Here, we are happy to introduce our lentiviral vector pseudotype derived from Sendai virus (SeV) glycoprotein.
Sendai Virus (SeV)
SeV, also known as mouse parainfluenza virus type 1 or hemagglutinating virus of Japan (HVJ), is a nonsegmented negative-strand RNA virus belonging to the Paramyxovirus family with a large spherical shape and an average diameter of 260 nm. SeV has two glycoproteins, HN and F proteins. HN is for attachment of the virus to cellular receptors and F is for cell fusion. Early studies have demonstrated that SeV could efficiently overcome a series of extra- and intracellular barriers in the respiratory tract, such as the glycocalyx, mucus layer, mucociliary clearance, and cell membranes. In addition, the Sendai virus F envelop protein (SV-F) is capable of binding specifically to the hepatic asialoglycoprotein receptor (ASGP-R), mediating the fusion of the viral envelope with the cell membrane.
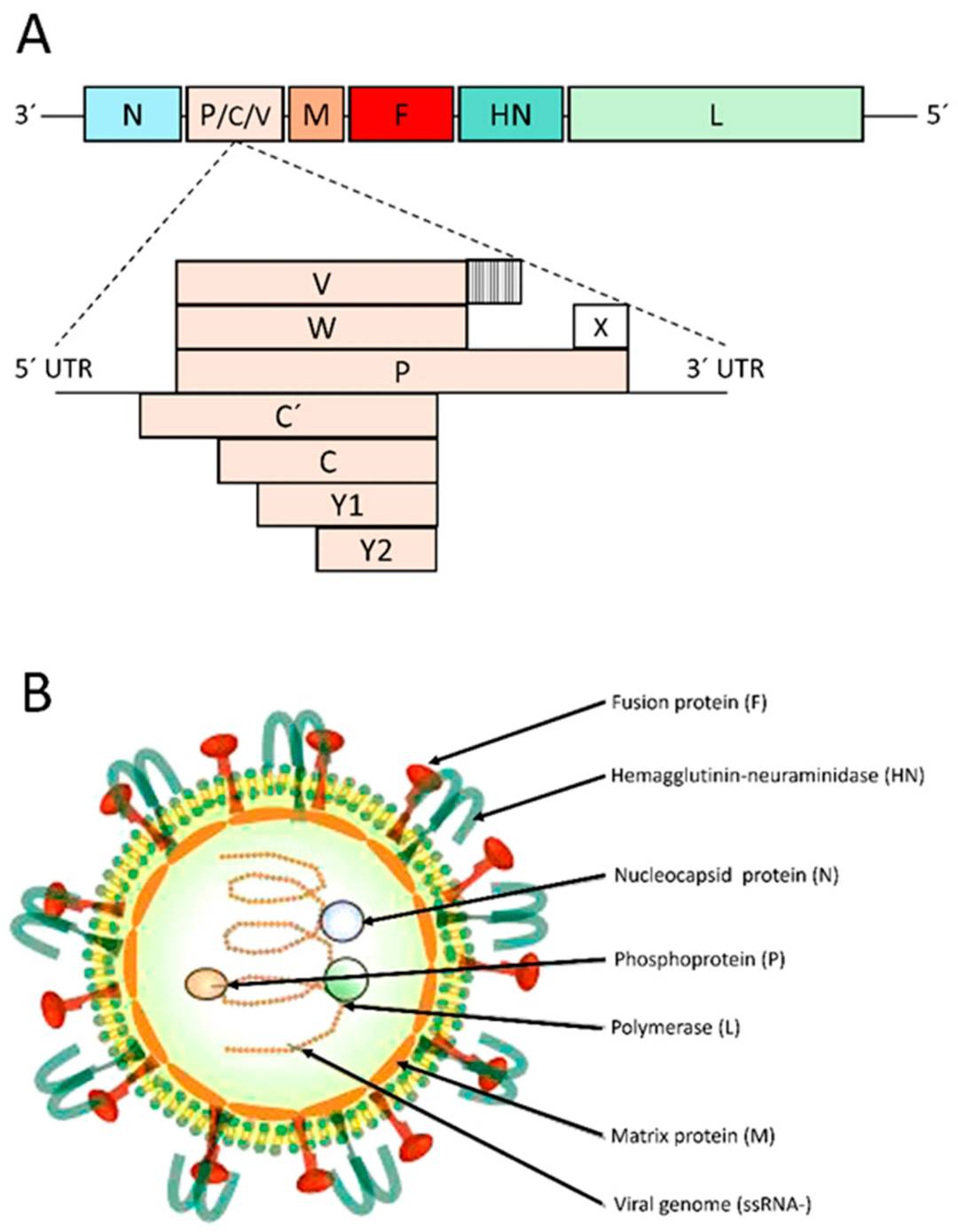 Figure 1. Genome and virion structure of Sendai virus (SeV).1
Figure 1. Genome and virion structure of Sendai virus (SeV).1
LVs Pseudotypes Bearing SeV-derived GPs
SeV is enveloped with fusion (F) and hemagglutinin-neuraminidase (HN) proteins. Since isolation in 1950s, recombinant SeV has been applied in multiple areas for basic and applied biology, such as preparation of hybrid cells and large-scale production of interferon (IFN). Recently, incorporation of heterologous envelope proteins into the virus particles becomes an important factor for the production of functional pseudotyped lentivirus vectors. In view organ and tissue tropism of SeV, SeV glycoprotein- pseudotyped vectors are expected to show expanded cell tropism, making them ideal tools for gene therapy, including therapy for respiratory diseases and hepatoma. Particularly, pseudotyped lentiviral vector derived from simian immunodeficiency virus SIVagm with SeV envelop glycoprotein has been confirmed to transduce various types of animal and human cell lines, which provides a new tool in gene therapy for various therapies.
Mechanism of SeV-Pseudotyped Lentiviral Vectors
The superior performance of Sendai virus-pseudotyped lentiviral vectors stems from a finely tuned entry mechanism that combines the robust delivery framework of lentivirus with the unique fusion and entry properties of Sendai virus. This hybrid system is engineered for maximum efficiency and reliability. During the viral production process, the two key envelope glycoproteins of Sendai virus—the Fusion (F) protein and the Hemagglutinin-Neuraminidase (HN) protein—are incorporated into the lipid bilayer of the lentiviral vector, replacing its native envelope.
- HN Protein (Attachment): This protein initiates the process by binding with high affinity to sialic acid receptors, which are ubiquitously present on the surface of most mammalian cells. This provides the vector with its broad cellular tropism.
- F Protein (Fusion): Upon receptor binding by HN, a conformational change is triggered in the adjacent F protein. This activated F protein directly mediates the fusion of the viral envelope with the host cell's plasma membrane.
Services at Creative Biolabs
We provide a comprehensive and fully customizable suite of services to generate high-performance SeV-pseudotyped lentiviral vectors, supporting your project from initial design through to ready-to-use viral stocks.
Custom Vector Design & Cloning
We begin by collaborating with you to design the optimal lentiviral construct for your specific application. Our service includes:
- Consultation & Strategy
- Molecular Cloning
- Reporter & Custom Constructs
Envelope Protein Optimization
- Tailored Pseudotyping Strategy: Based on your target cell type, we recommend and employ the most effective F/HN combination to achieve superior gene delivery efficiency.
- Validation & Screening: For novel applications, we can perform small-scale screening of different pseudotypes to identify the optimal envelope pairing before scaling up production.
Scalable Virus Production
From initial feasibility studies to pre-clinical scale, our production platform ensures consistency and quality at every step.
- Small-Scale Pilot Production: Ideal for initial proof-of-concept experiments, we produce pilot batches (typically 1-10 mL) to allow you to test functionality in your specific assay system.
- Large-Scale & High-Volume Production: For animal studies or large-scale cell engineering, we offer scaled-up production in multi-layered cell factories or bioreactors, delivering volumes up to several litres.
Comparison with Other Pseudotyping Systems
Choosing the right envelope is critical for the success of your in vitro or in vivo study.
| Feature | SeV (Sendai) | VSV-G | Rabies Virus | GaLV / LCMV |
|---|---|---|---|---|
| Primary Receptor | Sialic acid | LDL Receptor | NCAM / p75NTR | PiT-1 / alpha-dystroglycan |
| Entry Pathway | pH-Independent (Surface) | pH-Dependent (Endosomal) | Endosomal | Surface Fusion |
| Tissue Preference | Airway/Lung, Stem Cells | Pantropic (Most cells) | Neurons (Retrograde) | Hematopoietic cells |
| Cytotoxicity | Low | High (at high titers) | Moderate | Low |
| Main Advantage | High lung/immune efficiency | Extremely broad tropism | Targeted CNS delivery | Stable for blood lineages |
Quality Control & Characterization
Every batch undergoes rigorous testing to ensure safety, potency, and consistency:
- Titer Analysis: Determination of both physical (e.g., p24 ELISA) and functional (TU/mL) titers.
- Efficiency Validation: Transduction efficiency testing on relevant cell lines or primary cells.
- Safety Testing: Screening for Replication-Competent Lentiviruses (RCL).
- Purity Assessment: Sterility (bacterial/fungal) and endotoxin testing.
Service Features
Focusing on gene therapy for years, we have constructed and optimized numerous glycoprotein-based lentiviral vectors for global customers. With abundant experience, our professional scientists will help you select optimal virus type and glycoprotein for targeting specific cells or tissues to promote the development of your gene therapy programs. The vectors we are providing are characterized by:
- Broad host range: high-efficiency infection of dividing and nondividing cells
- Long-term stable expression of a transgene
- Low immunogenicity
- Low cytotoxicity
Why Choose Our Service?
We combine deep virological expertise with cutting-edge lentiviral production platforms. Our team is dedicated to providing optimized, reliable, and fully characterized SeV-pseudotyped lentiviral vectors, backed by comprehensive data and expert support to accelerate your project from concept to results.
Turnaround Time & Requirements
| Service Stage | Estimated Turnaround Time (TAT) | Requirements from Customer |
|---|---|---|
| Consultation & Strategy Design | 1-2 Weeks (Includes pseudotype selection and vector design) | 1. Target Information: Specific cell type (e.g., airway epithelial, HSPCs) or tissue to determine the optimal F/HN combination.2. Application Context: In vitro vs. In vivo requirements. |
| Vector Construction & Cloning | 2-3 Weeks (Includes molecular cloning and plasmid preparation) | 1. Gene of Interest: Sequence data (GenBank ID) or plasmid map for the custom payload.2. Payload Size: Confirmation that the insert is within the ~8-9 kb standard capacity. |
| Viral Production & QC | 4-6 Weeks (Varies by scale: Pilot vs. Large-Scale) | 1. Scale Preference: Selection of small-scale pilot (1-10 mL) for testing or large-scale (litres) for animal studies.2. QC Specifications: Specific requirements for titer analysis or purity assessment beyond standard RCL and endotoxin testing. |
Customer Reviews

Frequently Asked Questions
Q: Why choose Sendai Virus for lentiviral pseudotyping?
A: SeV pseudotyping offers a unique blend of high transduction efficiency in challenging primary cells, low cytotoxicity, and a distinct, pH-independent entry mechanism, often outperforming VSV-G where cell health and primary cell transduction are paramount.
Q: What cell types benefit most from SeV pseudotyping?
A: Respiratory epithelial cells, various immune cells (T cells, dendritic cells), hematopoietic and mesenchymal stem cells, neurons, and other primary cell types show significantly improved transduction with SeV-pseudotyped vectors.
Q: Can SeV-pseudotyped lentivirus be used in vivo?
A: Yes, they are highly effective for local in vivo delivery, particularly to the respiratory tracts via intranasal or intratracheal administration. Their compatibility with systemic delivery depends on the specific model and should be evaluated.
Q: How does SeV pseudotyping compare to VSV-G in safety?
A: SeV-pseudotyped vectors generally demonstrate lower cellular toxicity and are not inactivated by human serum complement, which can be an advantage over VSV-G for certain ex vivo and in vivo applications.
Q: What vector payload sizes are supported?
A: Like other lentiviral vectors, the standard capacity is up to ~8-9 kb. While the SeV envelope does not impose additional size restrictions, larger payloads may see a reduction in overall viral titer.
Connect with Us Anytime!
Pseudotyping lentiviral vectors with the Sendai virus envelope represents a superior strategy for efficient and gentle gene delivery to a wide array of sensitive and therapeutically relevant cell types. Our specialized service provides you with the tools and quality to push the boundaries of your research in gene therapy, cell engineering, and beyond. Contact us to discuss how our SeV pseudotyping service can advance your next project. For more detailed information, please feel free to contact us or directly send us an inquiry.
Reference
- Gómez Á, Reina R. Recombinant Sendai Virus Vectors as Novel Vaccine Candidates Against Animal Viruses. Viruses, 2025, 17(5): 737. https://doi.org/10.3390/v17050737 Distributed under Open Access license CC BY 4.0, without modification.
