Are you constrained by prolonged timelines, exorbitant production costs, and the persistent challenge of achieving sustained efficacy in solid tumors using conventional cell therapies? Creative Biolabs offers an integrated In vivo CAR-MA Development platform designed to overcome these barriers. By utilizing a non-viral mRNA-LNP delivery system engineered for myeloid-specific targeting, we enable direct in vivo generation of CAR-equipped macrophages, which bypasses complex ex vivo manufacturing, enhances deep tumor infiltration, and actively reprograms the immunosuppressive TME, accelerating the path to durable, off-the-shelf immunotherapies.
CAR-MA exert multi-faceted anti-tumor effects by mediating targeted phagocytosis through chimeric antigen receptors, remodeling the tumor microenvironment, and synergizing with adaptive immunity. The in vivo application of CAR-MA not only holds the potential to overcome immunosuppressive barriers in solid tumors but also represents a promising strategic direction for achieving durable and safe cellular therapies.
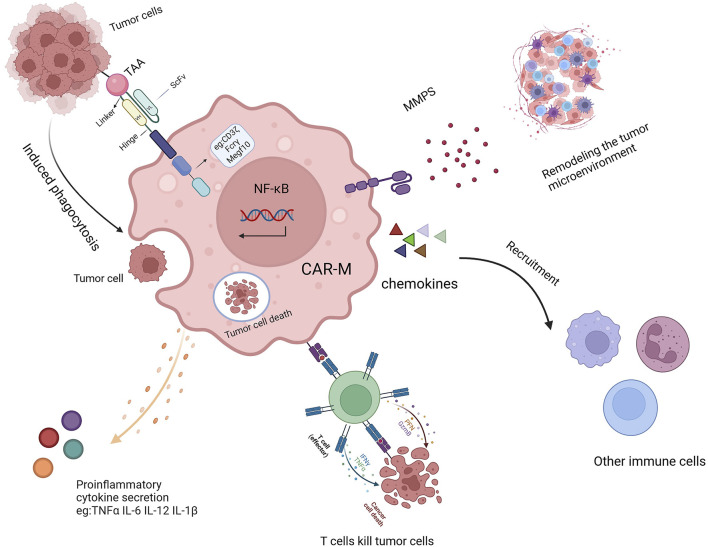 Fig.1 Immunomodulatory Functions of CAR-MA.1
Fig.1 Immunomodulatory Functions of CAR-MA.1
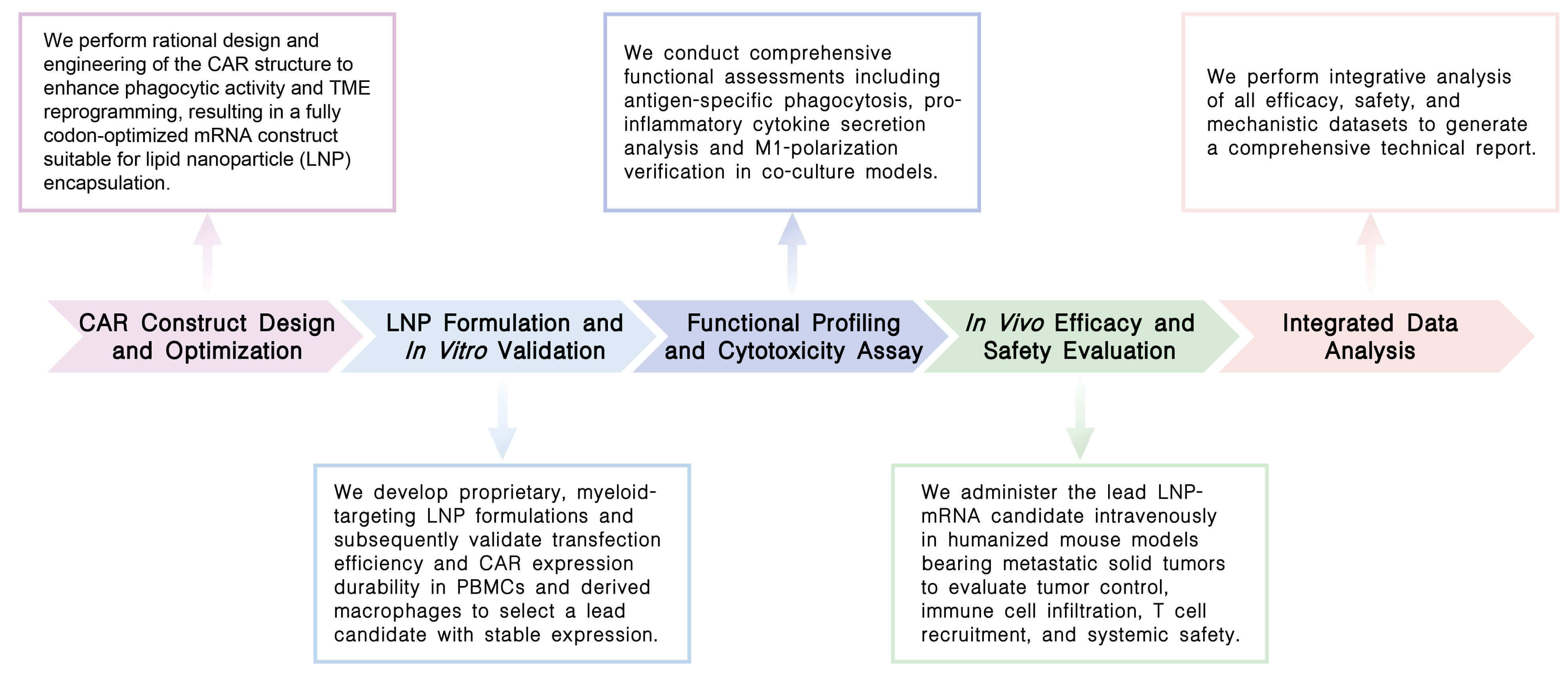
Creative Biolabs' CellRapeutics™ In Vivo CAR-M Cell Engineering Service delivers end-to-end solutions for creating targeted macrophage therapies. We engineer myeloid-specific CAR constructs, optimize proprietary LNP formulations for in vivo mRNA delivery, and validate systemic efficacy in advanced humanized tumor models, bypassing costly ex vivo manufacturing while enabling solid tumor penetration, TME reprogramming, and durable anti-tumor responses.
Our platform offers an integrated suite of advanced technologies for in vivo CAR-macrophage development, encompassing targeted lipid nanoparticle delivery, engineered viral vectors, synergistic co-delivery systems, and intelligent dual-signaling CAR architectures to achieve precise cell reprogramming and potent anti-tumor efficacy.

Our platform extends the reach of in vivo immune cell engineering beyond T cells, offering two targeted services designed to harness the unique therapeutic potential of B cells and NK cells.
We reprogram B cells in vivo to secrete therapeutic antibodies or cytokines, enabling long-term protein replacement and immunomodulation with minimal manufacturing complexity.
We engineer NK cells in vivo with chimeric antigen receptors, delivering potent, targeted antitumor immunity with reduced risk of cytokine release syndrome and graft-versus-host disease.
Required starting materials:
Key Steps:
Final Deliverables:
What is the biggest challenge in developing an in vivo CAR-MA, and how does Creative Biolabs address it?
The primary technical challenge is ensuring the LNP formulation selectively targets myeloid cells while minimizing off-target transfection in other cell types. We address this using proprietary, optimized LNP chemistries specifically engineered for myeloid tropism, which ensures robust CAR expression only in the desired immune cell population, maximizing therapeutic index.
Can the in vivo CAR-MA platform be used for non-solid tumors, such as hematological malignancies?
While the unique advantage of macrophages is their ability to penetrate solid tumors, the platform is broadly applicable. The systemic delivery is highly effective, and CAR-MA function in the circulation (monocytes) and lymphoid tissues could be leveraged against hematological cancers. We encourage you to contact our team to discuss your specific tumor indication.
As experts in cellular immunotherapy and metabolism, we are committed to delivering cutting-edge solutions. By selecting our In vivo CAR-MA Development Service, you gain access to CAR-macrophages engineered via a non-viral mRNA/LNP delivery platform. Our advanced approach overcomes the penetration barriers of solid tumors and actively reprograms the immunosuppressive TME.
"The implementation of Creative Biolabs' In vivo CAR-MA platform in our study has markedly enhanced the targeting precision of CAR delivery to myeloid-lineage cells. This approach enables the reliable in situ generation of CAR-macrophages, effectively eliminating concerns regarding off-target transduction."— Dr. La Hn.
"Employing Creative Biolabs' In vivo CAR-MA Development system has accelerated the advancement of therapeutic candidates targeting bulky, treatment-resistant solid tumors. This strategy exhibits robust antitumor activity and efficient T-cell recruitment, outcomes that were not achievable with previous ex vivo-engineered T-cell methodologies."— Dr. An Fx.
"Utilization of the In vivo CAR-MA platform from Creative Biolabs has substantially expedited the iterative optimization of CAR architectures and subsequent dosing regimens. This has shortened our preclinical development schedule by several months while allowing more precise management of the therapeutic index."— Dr. Pl Sh.
Ready to advance your next therapeutic breakthrough? Contact our expert scientific team for comprehensive technical consultations, tailored partnership models, and to initiate a collaborative project scoping discussion.
Reference
For any technical issues or product/service related questions, please leave your information below. Our team will contact you soon.
All products and services are For Research Use Only and CANNOT be used in the treatment or diagnosis of disease.
 NEWSLETTER
NEWSLETTER
The latest newsletter to introduce the latest breaking information, our site updates, field and other scientific news, important events, and insights from industry leaders
LEARN MORE NEWSLETTER NEW SOLUTION
NEW SOLUTION
CellRapeutics™ In Vivo Cell Engineering: One-stop in vivo T/B/NK cell and macrophage engineering services covering vectors construction to function verification.
LEARN MORE SOLUTION NOVEL TECHNOLOGY
NOVEL TECHNOLOGY
Silence™ CAR-T Cell: A novel platform to enhance CAR-T cell immunotherapy by combining RNAi technology to suppress genes that may impede CAR functionality.
LEARN MORE NOVEL TECHNOLOGY NEW SOLUTION
NEW SOLUTION
Canine CAR-T Therapy Development: From early target discovery, CAR design and construction, cell culture, and transfection, to in vitro and in vivo function validation.
LEARN MORE SOLUTION