CAR-T cell therapy has made great achievements in the past decade, while conventional CAR-T cell therapy also has some obstacles to overcome, such as the CRS and ICANS toxicity, and the expensive and complex manufacturing process. The in-vivo CAR-T cell engineering by nanoparticles carrying CAR genes and gene-editing tools has shown potential breakthroughs in overcoming the above obstacles of traditional CAR-T cell therapies. Therefore, there is more urgent to develop safe and simple in-vivo CAR-T cell engineering solutions.
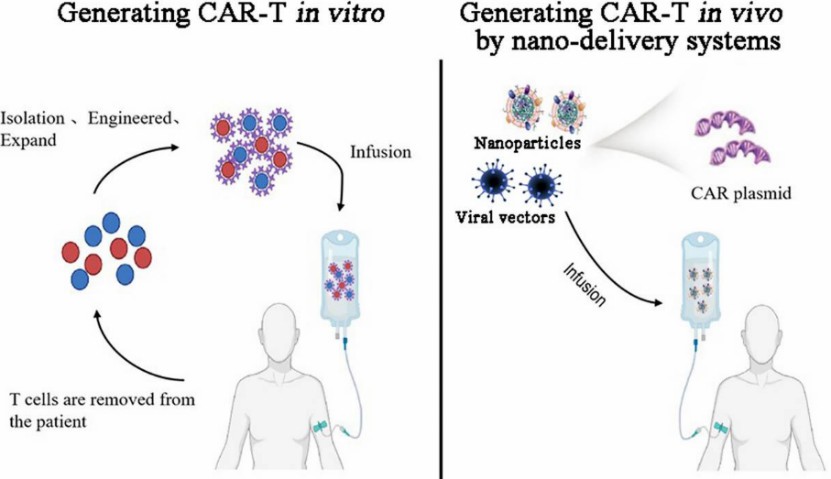 Fig.1 Comparison of generating CAR-T in-vitro and CAR-T in-vivo. (Xin, et al., 2022)
Fig.1 Comparison of generating CAR-T in-vitro and CAR-T in-vivo. (Xin, et al., 2022)
To overcome the limitations of current CAR-T therapy, Creative Biolabs has developed one-stop in vivo CAR-T cell engineering services to support global customers. Our in vivo CAR-T cell engineering carries engineered CAR genes through delivery systems (such as non-viral or viral systems) characterized by multiple functions to overcome the obstacles of current CAR-T, such as CRS & ICANS toxicities, immunosuppressive microenvironments, and complex manufacturing processes.
Our in vivo CAR-T cell engineering services have the following characteristics.
Our CAR structure incorporates special cytokine genes that enable the CAR-T cells to secrete cytokines and flush out the immunosuppression microenvironment, which makes it suitable for T-cell survival and proliferation. There are some cytokine types to design for, including but not limited to: IL-7, IL-12, IL-15, IL-18, IL-19, IL-23;
The delivery system plays an important role in achieving specific and efficient in vivo T cell transduction, which can greatly enhance the in vivo generation and anti-tumor activity of CAR-T cells. Creative Biolabs offers the following two modified delivery systems to support our customers' projects.
We run comprehensive in vitro tests (expression, viability, activation) and in vivo evaluations (biodistribution, persistence, safety) to validate performance.
Expert data interpretation, detailed reporting, and guidance to help you make decisions and refine your strategy for subsequent development phases.
Whether you need just the CAR design, delivery development, or full in vivo programming + validation, our services are modular and customizable.
Our team has deep expertise in both CAR engineering and in vivo delivery systems, with a track record across multiple platforms and cell types.
Discover How We Can Help - Request a Consultation
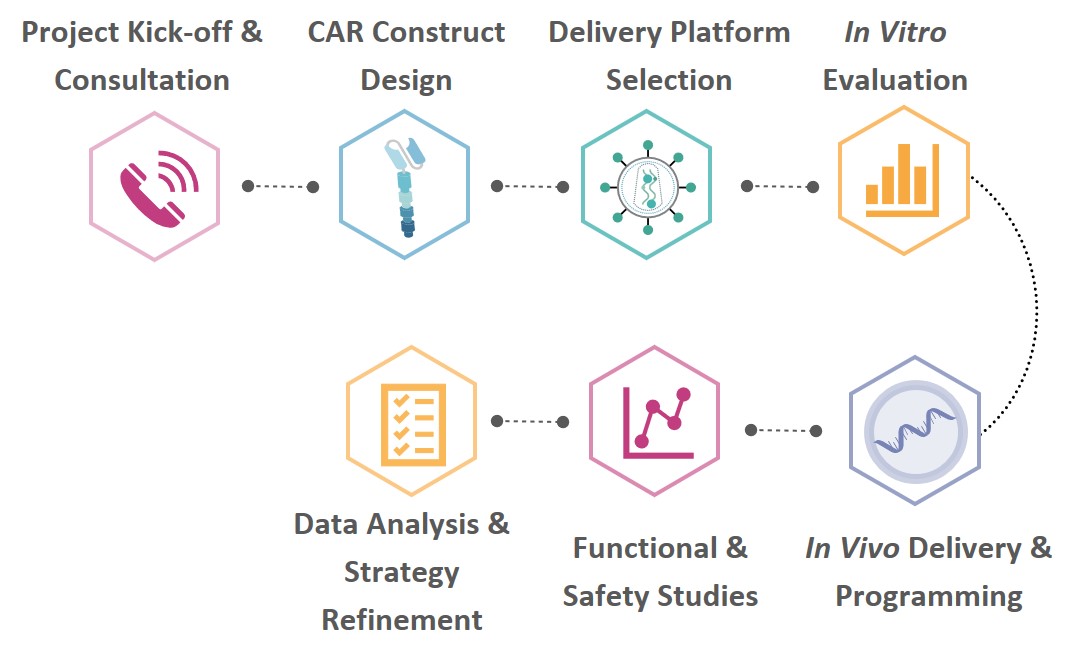
We follow a structured and comprehensive workflow to guide your in vivo CAR‑T project from initial concept to functional validation, ensuring clarity, efficiency, and reliable results at every step.
Paper title: Preclinical proof of concept for VivoVec, a lentiviral-based platform for in vivo CAR T-cell engineering
Summary: Researchers established a lentiviral vector-based platform for in vivo CAR-T cell engineering: UB-VV100. Which encodes a 2nd CD19-targeted CAR displayed an anti-CD3 scFv on the surface with a rapamycin-activated cytokine receptor (RACR) system additionally designed for the treatment of B-cell malignancies. The study found that this strategy produced functional CAR-T cells with favorable biodistribution characteristics in vivo.
Technology: lentiviral-based CD19 CAR vector for in vivo CAR T-cell engineering;
Tumor Type: B-cell malignancy;
Whether your project is preclinical research or translational development, our modular services can adapt to your needs.
We support in-depth functional and safety testing in physiologically relevant models, giving you confidence in the performance of your in vivo engineered CAR-T cells.
We work closely with clients, providing strategic input, data-driven insights, and continuous communication throughout the project lifecycle.
For in vivo CAR-T engineering development, which stage is more challenging and important in the development?
In the development, CAR design & construction, the selection of a modified delivery system seems to be critical to the overall development, optimized CAR construction and the modified delivery system could achieve specific and efficient in vivo T cell transduction, greatly enhancing the in vivo CAR-T generation and anti-tumor activity. Based on these factors, we offer several choices for our customers, such as surface-engineered vectors, and modified mRNA delivery systems.
If you are interested in our CellRapeutics™ in vivo CAR-T cell engineering services, please feel free to contact us to learn more.
With extensive experience in in vivo CAR‑T engineering, we provide modular, flexible solutions covering CAR design, delivery optimization, and functional validation. Our platform ensures efficiency, safety, and robust performance evaluation, supporting both preclinical and translational research. Partner with us to accelerate your project. Contact our team today to discuss your in vivo CAR‑T needs and explore tailored solutions.
References
For any technical issues or product/service related questions, please leave your information below. Our team will contact you soon.
All products and services are For Research Use Only and CANNOT be used in the treatment or diagnosis of disease.
 NEWSLETTER
NEWSLETTER
The latest newsletter to introduce the latest breaking information, our site updates, field and other scientific news, important events, and insights from industry leaders
LEARN MORE NEWSLETTER NEW SOLUTION
NEW SOLUTION
CellRapeutics™ In Vivo Cell Engineering: One-stop in vivo T/B/NK cell and macrophage engineering services covering vectors construction to function verification.
LEARN MORE SOLUTION NOVEL TECHNOLOGY
NOVEL TECHNOLOGY
Silence™ CAR-T Cell: A novel platform to enhance CAR-T cell immunotherapy by combining RNAi technology to suppress genes that may impede CAR functionality.
LEARN MORE NOVEL TECHNOLOGY NEW SOLUTION
NEW SOLUTION
Canine CAR-T Therapy Development: From early target discovery, CAR design and construction, cell culture, and transfection, to in vitro and in vivo function validation.
LEARN MORE SOLUTION