All products and services are For Research Use Only and CANNOT be used in the treatment or diagnosis of disease.
As cellular immunotherapy continues to revolutionize cancer treatment, the demand for precision, efficiency, and scalability in research is paramount. Creative Biolabs provides an elite portfolio of integrated CAR-T development kits, meticulously engineered to streamline your workflow, from genetic construction and viral packaging to functional validation and quality control.
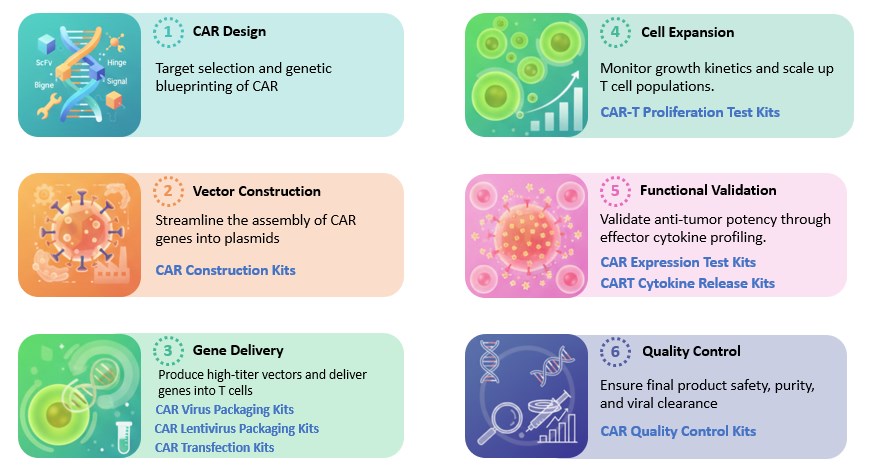
Our CAR development kits are modularly designed and may include the following key components:
| Genetic Engineering & Vector Construction | ||
|---|---|---|

|
|
|
|
CAR Construction Kits For the design and assembly of CAR gene sequences (scFv, hinge, and signaling domains). |
CAR Virus Packaging Kits General systems for producing viral vectors. |
CAR Lentivirus Packaging Kits Specialized high-titer systems specifically for lentiviral vector production. |
| Cell Engineering & Expansion | ||

|

|
|
|
CAR Transfection Kits Optimized reagents for introducing genetic material into T-cells (via viral or non-viral methods). |
CAR-T Proliferation Test Kits Comprehensive assays designed to evaluate the expansion capacity, growth kinetics, and metabolic fitness of genetically engineered T cells following CAR modification. |
|
| Quality Control & Functional Validation | ||

|
|

|
|
CAR Expression Test Kits For quantifying the density and percentage of CAR expression on the cell surface. |
CAR-T Cytokine Release Test Kits To evaluate the functional activation and safety profile. |
CAR Quality Control Kits Comprehensive assays for safety testing, including purity, sterility, and vector copy number. |
Streamline your research from initial concept to final validation with our specialized kits designed for every critical milestone.
How to improve CAR construct transfection efficiency?
Optimize plasmid quality (high purity, endotoxin-free), DNA-to-reagent ratio, and cell density; use immune cell-specific reagents and validated protocols.
Common causes of low CAR lentivirus titer?
Suboptimal plasmid ratios, poor packaging cell health/over-confluency, inefficient transfection. Improve with high-viability cells, optimized plasmid stoichiometry, and standardized packaging kits.
Factors influencing CAR-T in vitro expansion?
Activation strength, cytokines, nutrients, culture environment. Use CAR-T Proliferation Test Kits to monitor growth/metabolism and optimize conditions.
If you are planning or optimizing a CAR-T research project and need guidance on kit selection or experimental design, our scientific support team is ready to assist you. Contact us to discuss your research goals and discover tailored solutions for your study.
For any technical issues or product/service related questions, please leave your information below. Our team will contact you soon.

 NEWSLETTER
NEWSLETTER
The latest newsletter to introduce the latest breaking information, our site updates, field and other scientific news, important events, and insights from industry leaders
LEARN MORE NEWSLETTER NEW SOLUTION
NEW SOLUTION
CellRapeutics™ In Vivo Cell Engineering: One-stop in vivo T/B/NK cell and macrophage engineering services covering vectors construction to function verification.
LEARN MORE SOLUTION NOVEL TECHNOLOGY
NOVEL TECHNOLOGY
Silence™ CAR-T Cell: A novel platform to enhance CAR-T cell immunotherapy by combining RNAi technology to suppress genes that may impede CAR functionality.
LEARN MORE NOVEL TECHNOLOGY NEW SOLUTION
NEW SOLUTION
Canine CAR-T Therapy Development: From early target discovery, CAR design and construction, cell culture, and transfection, to in vitro and in vivo function validation.
LEARN MORE SOLUTION