Are you navigating the challenges of exorbitant manufacturing expenses, intricate supply chain logistics, and the mandatory yet toxic lymphodepletion regimen in your cell therapy program? Creative Biolabs' CellRapeutics™ In Vivo CAR-NK Cell Engineering Service platform offers a transformative alternative. We directly administer genetic payloads systemically to generate functional CAR-NK cells within the patient's own immune system, utilizing advanced targeted delivery technologies, which fundamentally bypasses costly ex vivo expansion and eliminates preconditioning chemotherapy. We develop a truly scalable, readily available therapeutic modality.
CAR-NK cell therapy is an innovative immunotherapeutic strategy that involves genetically engineering NK cells to express a CAR targeting specific tumor antigens, thereby enabling them to selectively recognize and eliminate malignant cells. Compared to CAR-T cell therapy, CAR-NK cells offer distinct advantages, including broader cell source availability, no requirement for strict human leukocyte antigen (HLA) matching, feasibility as an "off-the-shelf" therapeutic product, and a lower risk of severe cytokine release syndrome (CRS) or graft-versus-host disease (GVHD). These attributes underscore their favorable safety profile and considerable therapeutic potential in the treatment of both hematological malignancies and solid tumors.
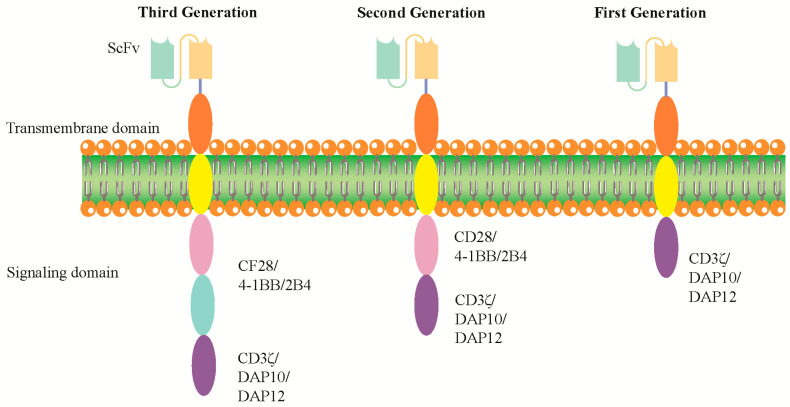 Fig.1 Design and optimization of CAR constructs in CAR-NK cell therapy.1
Fig.1 Design and optimization of CAR constructs in CAR-NK cell therapy.1
Creative Biolabs' CellRapeutics™ In Vivo CAR-NK Cell Engineering Service leverages cell specific delivery of non-viral or engineered viral vectors, bypassing the requirement for preconditioning chemotherapy and resource-intensive ex vivo production. Our platform enables the concurrent induction of multiple cytotoxic immune subsets, extends functional durability via integrated cytokine signaling, and provides a highly efficacious, safer, and operationally simplified therapeutic modality.
We offer a comprehensive suite of technologies for In vivo CAR-NK Development, including targeted cellular engraftment via engineered chemokine receptors, CAR expression control with tissue-specific promoters, enhanced persistence through cytokine co-expression, and immune checkpoint blockade to overcome the suppressive tumor microenvironment.

Our in vivo engineering portfolio extends immune cell reprogramming beyond T cells.
We reprogram B cells in vivo to secrete therapeutic proteins, enabling targeted protein replacement and immunomodulation with sustained, site-specific expression.
We generate CAR macrophages directly in vivo to enhance tumor phagocytosis, remodel the TME, and drive durable anti-tumor immunity with a favorable safety profile.
Required starting materials:
Key Steps:
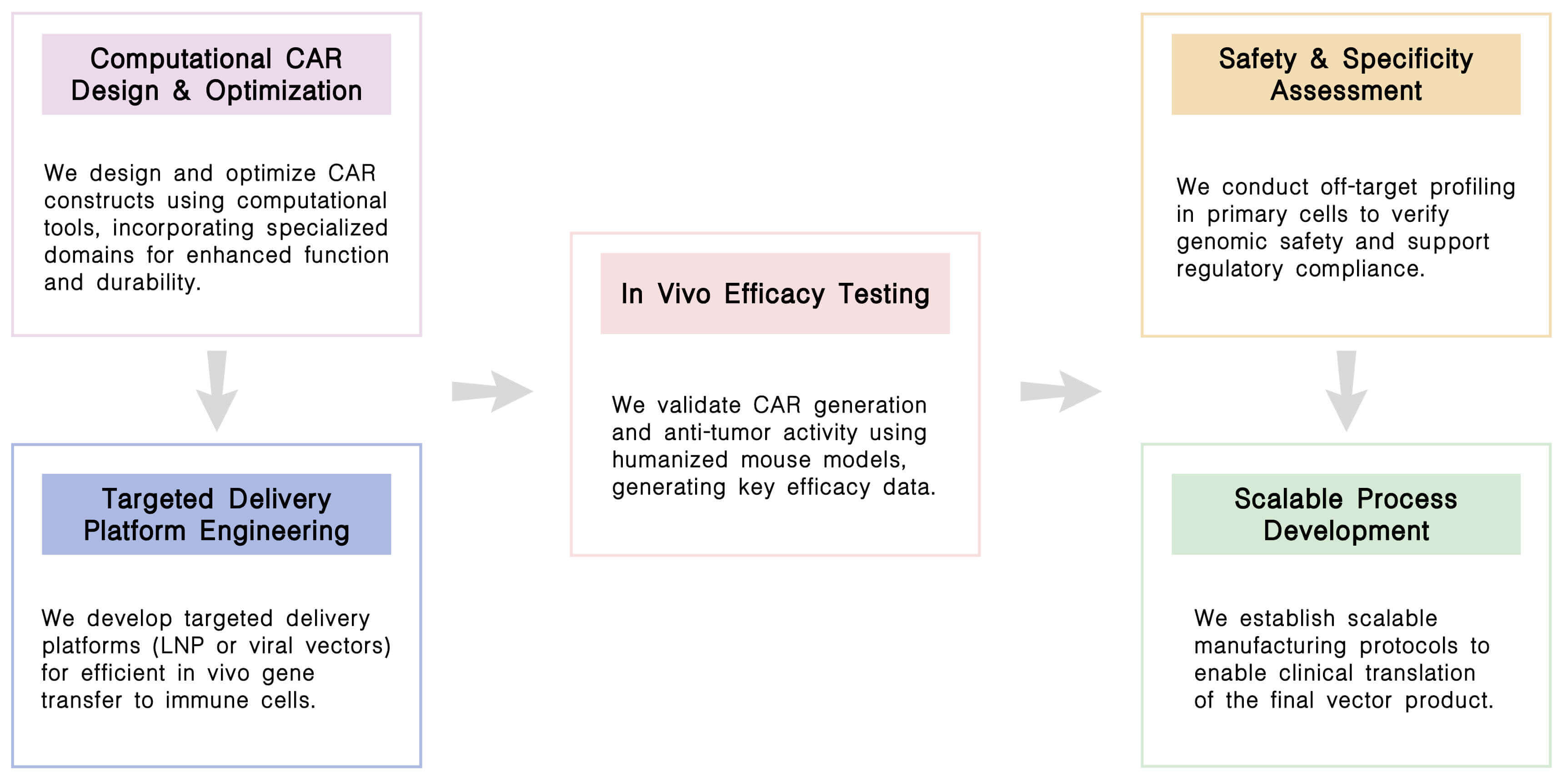
Final Deliverables:
How does in vivo CAR-NK development compare to traditional ex vivo manufacturing for solid tumors?
Ex vivo processing is costly and complex, but in vivo CAR-NK offers a highly scalable, off-the-shelf solution that bypasses the need for patient cell harvesting. Furthermore, our targeted in vivo approach is specifically engineered to overcome the physical barriers, like the Extracellular Matrix (ECM), encountered by cell therapies in solid tumors, leading to improved infiltration.
Is the elimination of lymphodepletion safe and effective?
Yes. Traditional CAR-T relies on lymphodepletion to create space for the infused cells. Our advanced in vivo delivery systems are designed to selectively target and transduce sufficient immune cells without requiring preconditioning chemotherapy, significantly enhancing patient safety and reducing toxicity risk. Preclinical data validate the robust generation and expansion of functional CAR-NK cells under these conditions.
We pioneer safe and potent in vivo CAR-NK solutions. Our core non-viral mRNA-LNP platform eliminates the risks of genomic integration and avoids lymphodepletion, while leveraging NK cells' dual natural and ADCP-mediated cytotoxicity. This is validated by rigorous off-target profiling to ensure clinical-grade safety and efficacy from concept to clinic.
Customer Reviews
"By integrating Creative Biolabs' In vivo CAR-NK Development platform into our study, we achieved a marked enhancement in therapeutic cell longevity in vivo, primarily through their advanced IL-15 armoring strategy. The resulting functional persistence proved to be twice as durable compared to conventional CAR designs."— Dr. L***a S.
"We selected Creative Biolabs based on their emphasis on preserving the innate cytotoxicity of NK cells beyond CAR-directed targeting, a key advantage for addressing highly heterogeneous tumors. Their comprehensive tumor microenvironment assessment, provided as part of the service, played a pivotal role in shaping our combined treatment approach."— Prof. J***n H.
"The rigorous off-target profiling integrated into Creative Biolabs' In vivo CAR-NK Development process represented a decisive benefit for our program. It directly alleviated our safety concerns related to genomic integration and delivered the precise dataset required to advance our pre-IND submission without delay."— M***a K.
How to Contact Us?
Unlock your therapeutic potential by partnering with Creative Biolabs. We specialize in pioneering in vivo CAR-NK engineering and next-generation delivery platforms to accelerate your candidate's transition into clinical development.
Connect with our experts to explore customized strategies for your program.
Reference
For any technical issues or product/service related questions, please leave your information below. Our team will contact you soon.
All products and services are For Research Use Only and CANNOT be used in the treatment or diagnosis of disease.
 NEWSLETTER
NEWSLETTER
The latest newsletter to introduce the latest breaking information, our site updates, field and other scientific news, important events, and insights from industry leaders
LEARN MORE NEWSLETTER NEW SOLUTION
NEW SOLUTION
CellRapeutics™ In Vivo Cell Engineering: One-stop in vivo T/B/NK cell and macrophage engineering services covering vectors construction to function verification.
LEARN MORE SOLUTION NOVEL TECHNOLOGY
NOVEL TECHNOLOGY
Silence™ CAR-T Cell: A novel platform to enhance CAR-T cell immunotherapy by combining RNAi technology to suppress genes that may impede CAR functionality.
LEARN MORE NOVEL TECHNOLOGY NEW SOLUTION
NEW SOLUTION
Canine CAR-T Therapy Development: From early target discovery, CAR design and construction, cell culture, and transfection, to in vitro and in vivo function validation.
LEARN MORE SOLUTION