The advancement of cell-based immunotherapies into clinical practice frequently encounters major obstacles, including resource-intensive ex vivo production processes, limited in vivo longevity of therapeutic cells, and the challenge of tumor antigen diversity. Creative Biolabs' CellRapeutics™ in vivo PBMC engineering platform overcomes these fundamental limitations by facilitating the direct in vivo genetic modification of a patient's own circulating immune cells. Utilizing proprietary, cell-targeted delivery vectors, we achieve efficient and specific gene transfer without the need for external cell manipulation. Our strategy eliminates the lengthy and costly ex vivo manufacturing step, significantly accelerating development timelines and reducing production expenses.
Peripheral blood mononuclear cells (PBMCs) represent a heterogeneous population comprising critical immune cells such as lymphocytes and monocytes. Beyond serving as an accessible peripheral blood sample, PBMCs can reflect the systemic immune status of the body. They are widely utilized to assess disease-associated immune response features, screen for predictive biomarkers, and develop personalized immunotherapy strategies. Currently, the isolation of PBMCs predominantly relies on density gradient centrifugation methods, which effectively separate whole blood into layers and enrich mononuclear cells. Further purification steps, including red blood cell lysis, enhance cellular purity, thereby providing a reliable cell source for subsequent functional, molecular, and genomic analyses.
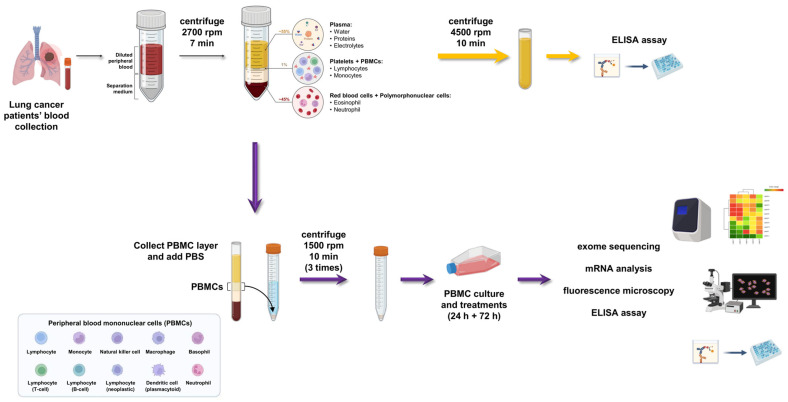 Fig.1 PBMC isolation workflow.1
Fig.1 PBMC isolation workflow.1
Creative Biolabs' CellRapeutics™ In Vivo Peripheral Blood Mononuclear Cell (PBMC) Engineering Service transitions CAR T-cell therapy from a complex, costly manufacturing burden to a precise, streamlined biological process occurring within the native environment. We solve the core logistical and biological challenges of ex vivo therapy by delivering the therapeutic payload directly to the patient's PBMCs in vivo, which ensures maximum cell integrity, superior T cell fitness, and a predictable path to clinical translation.
Our platform offers an integrated, end-to-end solution for in vivo PBMC development, encompassing high precision cell isolation technologies, targeted in vivo gene delivery systems, and intelligent CAR designs with built-in polarization signals to generate potent and durable cell therapies directly within the patient.

Required starting materials:
Key Steps:
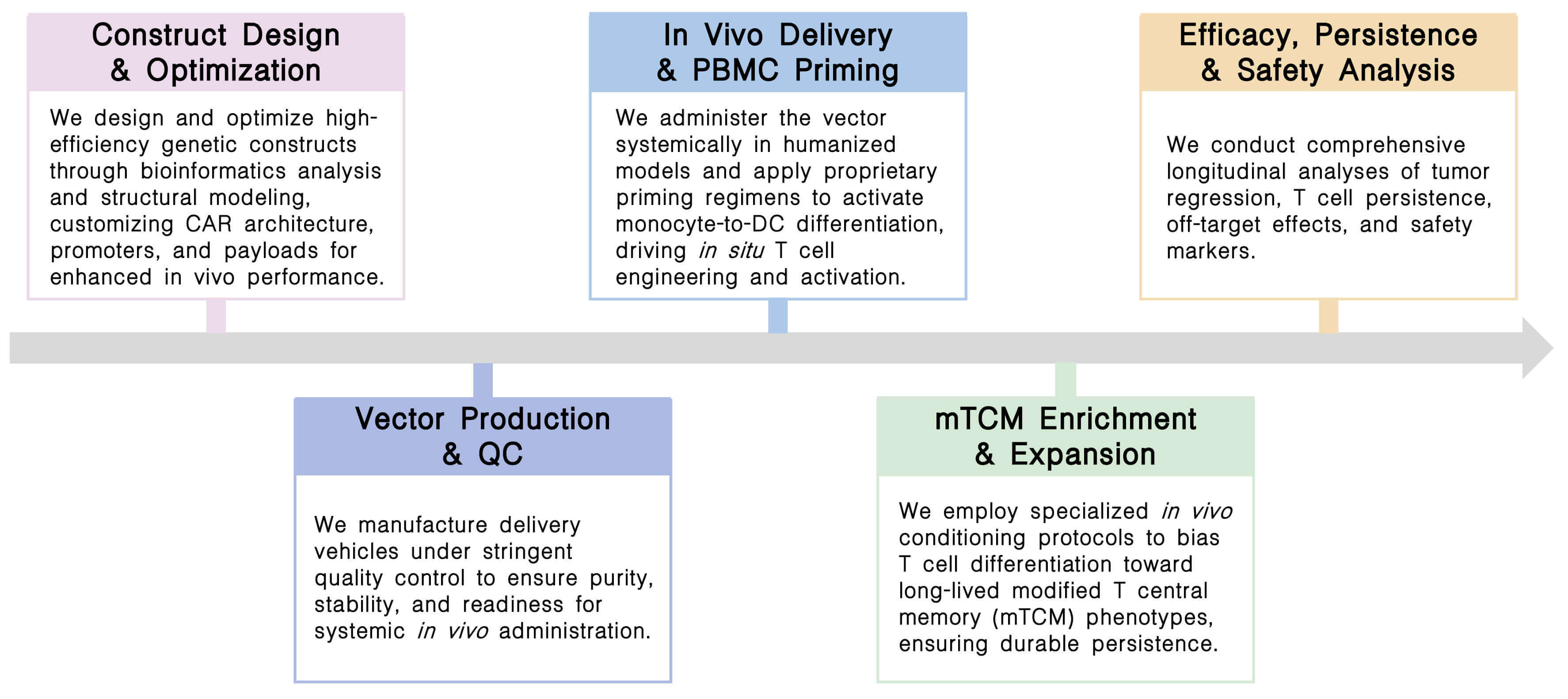
Final Deliverables:
What type of genetic cargo can your In vivo PBMC Development platform deliver?
Our proprietary delivery vectors are highly versatile and can deliver various payloads, including full-length CAR sequences, TCRs, specialized co-stimulatory molecules, and non-integrative gene editing components. We tailor the vector to your specific therapeutic and safety requirements.
How does the in vivo approach specifically ensure better T-cell durability than ex vivo methods?
The ex vivo process involves forced expansion, often resulting in exhausted T effector cells. Our in vivo priming uses the natural, endogenous immune cues to drive differentiation toward the highly desired T Central Memory (mTCM) phenotype. This results in cells with superior proliferative capacity and long-term survival, which is the key to preventing tumor relapse.
We provide an integrated platform that transforms peripheral blood mononuclear cells into potent in vivo CAR therapies. Our approach combines computational CAR design, targeted LNP or viral delivery, rigorous humanized model validation, mandatory genome safety profiling, and scalable manufacturing protocols to deliver a safe, effective, and clinically translatable therapeutic strategy.
"Using Creative Biolabs' In vivo PBMC Development in our research has significantly improved the long-term persistence and T central memory profile of our engineered cells compared to our own ex vivo products."— D. T****n, Research Director.
"The multi-targeting delivery mechanism was essential for validating our strategy against highly heterogeneous solid tumor models, an area where single-antigen ex vivo products consistently failed."— Dr. A. P***k, Immuno-Oncology Lead.
"Creative Biolabs' commitment to understanding the intrinsic immune status, like the cGAS-STING activation pathway, allowed us to refine our therapeutic construct for maximized patient response, greatly informing our selection criteria."— S. M***r, Translational Scientist.
To explore a tailored in vivo PBMC development strategy, receive project-specific technical details, or schedule a comprehensive consultation for your immunotherapy program, please contact our scientific team to initiate a collaborative discussion.
Reference
For any technical issues or product/service related questions, please leave your information below. Our team will contact you soon.
All products and services are For Research Use Only and CANNOT be used in the treatment or diagnosis of disease.
 NEWSLETTER
NEWSLETTER
The latest newsletter to introduce the latest breaking information, our site updates, field and other scientific news, important events, and insights from industry leaders
LEARN MORE NEWSLETTER NEW SOLUTION
NEW SOLUTION
CellRapeutics™ In Vivo Cell Engineering: One-stop in vivo T/B/NK cell and macrophage engineering services covering vectors construction to function verification.
LEARN MORE SOLUTION NOVEL TECHNOLOGY
NOVEL TECHNOLOGY
Silence™ CAR-T Cell: A novel platform to enhance CAR-T cell immunotherapy by combining RNAi technology to suppress genes that may impede CAR functionality.
LEARN MORE NOVEL TECHNOLOGY NEW SOLUTION
NEW SOLUTION
Canine CAR-T Therapy Development: From early target discovery, CAR design and construction, cell culture, and transfection, to in vitro and in vivo function validation.
LEARN MORE SOLUTION