By leveraging the transformative potential of in vivo CAR-T technology, Creative Biolabs offers comprehensive In vivo Viral-based CAR-T Development Services aimed at overcoming the major limitations of conventional ex vivo approaches. Our platform is designed to bypass the lengthy, costly, and logistically complex manufacturing processes by directly engineering patient T cells in vivo using targeted lentiviral and AAV delivery systems. This enables sustained CAR expression, enhanced T-cell persistence, and reduced immunotoxicity, accelerating therapeutic development while improving clinical accessibility and scalability for next-generation cell therapies.
The ex vivo CAR-T relies on isolating T cells from patients or healthy donors, followed by genetic modification and expansion outside the body before reinfusion, a process that is complex, time-consuming, and costly. In contrast, the in vivo strategy employs systemic delivery vehicles to directly reprogram circulating T cells within the patient, thereby bypassing extensive ex vivo manipulation, streamlining the production workflow, and significantly lowering the barriers to therapeutic access. Viral vector-based platforms, particularly those utilizing lentivirus and adeno-associated virus, have emerged as a major focus of current research due to their high transduction efficiency and sustained CAR expression.
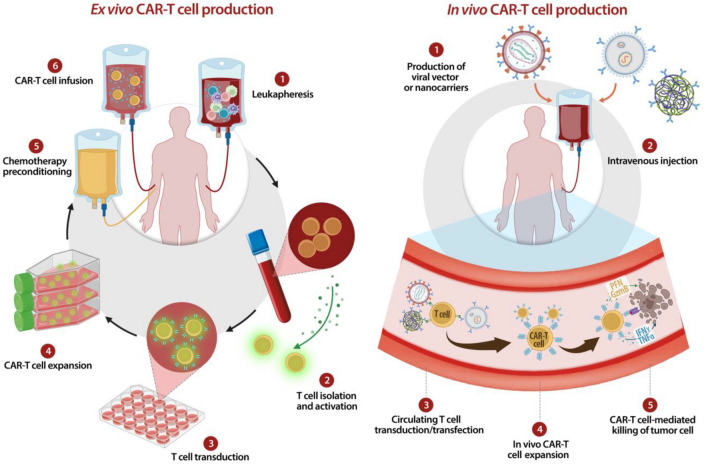 Fig.1 Comparative analysis: ex vivo and in vivo CAR-T generation.1
Fig.1 Comparative analysis: ex vivo and in vivo CAR-T generation.1
Creative Biolabs' In vivo Viral-based CAR-T Development Services genetically engineers specific T cell subsets directly within the patient to generate functional CAR-T cells in situ. Our strengths lie in integrating clinically validated CAR designs, highly specific viral targeting systems, and comprehensive in vivo efficacy and safety assessment platforms. Together, these capabilities establish a more efficient, safe, and scalable pathway for next-generation CAR-T therapy development.
We provide two distinct, cutting-edge delivery approaches, AAV and Lentivirus vector technologies, designed to enable efficient and durable in situ engineering of patient T cells. Each system offers unique advantages in terms of safety, persistence, and manufacturing scalability.
This service utilizes recombinant AAV vectors for the targeted delivery of CAR constructs directly into circulating T cells in vivo. Its advantages include high transduction efficiency in non-dividing cells, low immunogenicity, and sustained episomal expression, enabling durable CAR-T responses with reduced manufacturing time and complexity.
Through engineered lentiviral vectors pseudotyped with T cell-targeting ligands, we achieve stable genomic integration and persistent CAR expression in vivo. This approach offers high transduction efficiency, scalability, and the ability to maintain long-term T cell functionality, providing a robust platform for "off-the-shelf" in vivo CAR-T therapy development.
Required starting materials:
Key Steps:
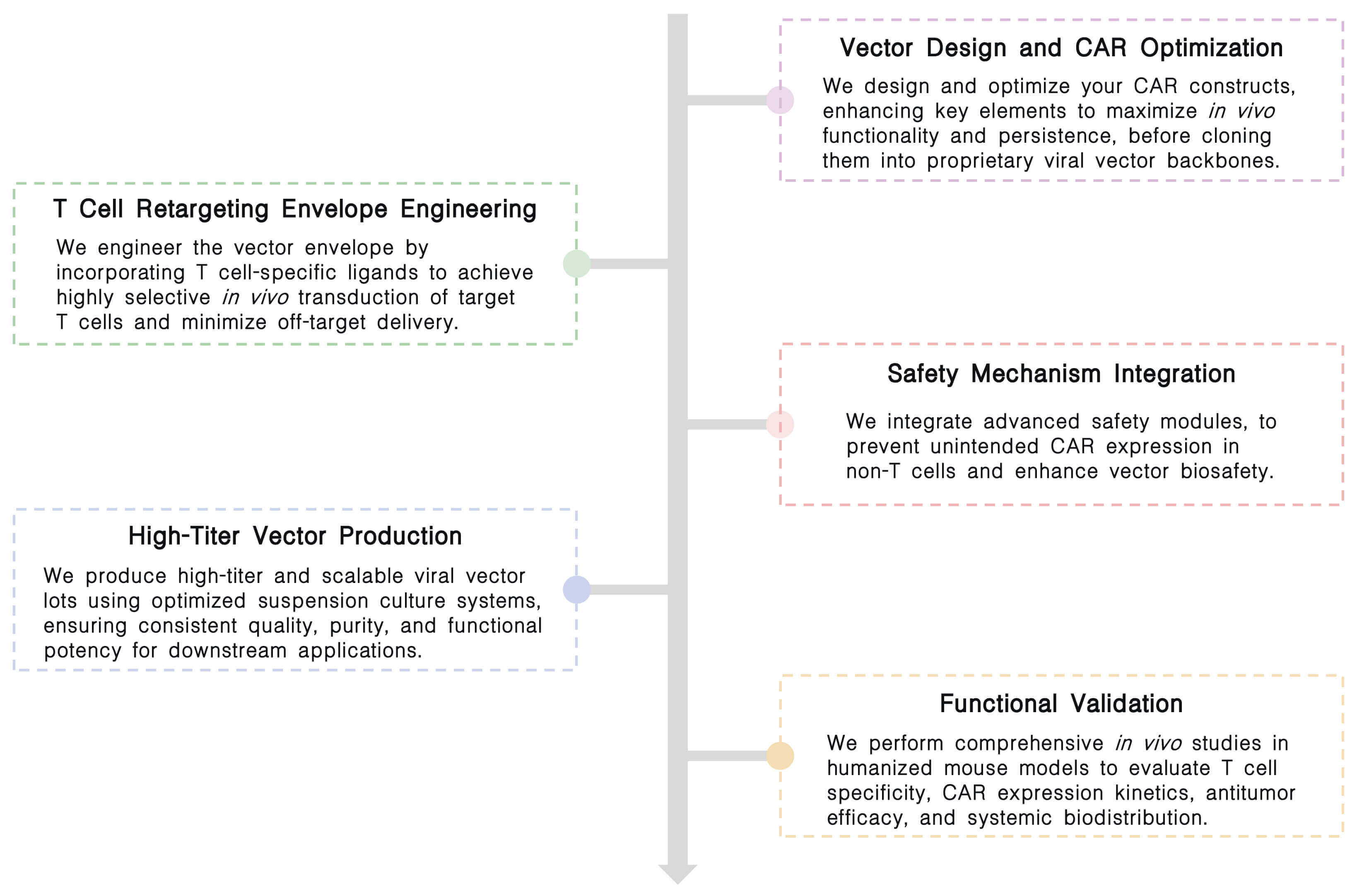
Final Deliverables:
How does the "ready-to-use" in vivo approach compare to traditional ex vivo CAR-T therapy for solid tumors?
The in vivo approach is more efficient because it avoids T cell exhaustion common during ex vivo expansion, yielding a more potent cell population. We also design next-generation CARs and utilize co-delivery strategies to help engineered T cells resist the immunosuppressive microenvironment of solid tumors.
What steps are taken to ensure the in vivo viral vectors target only T cells and avoid off-target toxicity?
We ensure high specificity through two key strategies: Retargeted Viral Envelopes and the Safety System, which prevents the expression of the CAR protein in non-target cells, minimizing the risk of inadvertent tumor cell transduction.
We provide an advanced viral delivery platform specifically engineered to generate CAR-T cells directly within the patient. Our expertise integrates clinically validated CAR designs with targeted viral envelope engineering and proprietary technology to ensure precise T cell transduction, enhanced functional persistence, and superior safety. This approach eliminates ex vivo manufacturing, offering a scalable and efficient path to next-generation cell therapies.
In the evolving field of in vivo CAR-T engineering, we are dedicated to pioneering novel platforms that significantly improve therapeutic efficacy and accessibility in oncology.
This service leverages in vitro-transcribed (IVT) mRNA encapsulated in advanced lipid nanoparticles (LNPs) to enable transient yet potent CAR expression directly within circulating T cells in vivo, allowing for rapid, dose-tunable CAR-T generation without genomic integration and reducing long-term safety risks.
Utilizing engineered circular RNA (circRNA) delivered via targeted non-viral carriers, this platform facilitates extended CAR protein expression compared to linear mRNA due to its inherent resistance to exonucleases. The service offers enhanced translational durability and stability in vivo, supporting sustained T cell activity while circumventing the need for viral vectors or repeated dosing.
To explore tailored solutions for your project, obtain a customized workflow proposal, or learn more about our proprietary platforms, please reach out to our scientific team for a detailed consultation.
Reference
For any technical issues or product/service related questions, please leave your information below. Our team will contact you soon.
All products and services are For Research Use Only and CANNOT be used in the treatment or diagnosis of disease.
 NEWSLETTER
NEWSLETTER
The latest newsletter to introduce the latest breaking information, our site updates, field and other scientific news, important events, and insights from industry leaders
LEARN MORE NEWSLETTER NEW SOLUTION
NEW SOLUTION
CellRapeutics™ In Vivo Cell Engineering: One-stop in vivo T/B/NK cell and macrophage engineering services covering vectors construction to function verification.
LEARN MORE SOLUTION NOVEL TECHNOLOGY
NOVEL TECHNOLOGY
Silence™ CAR-T Cell: A novel platform to enhance CAR-T cell immunotherapy by combining RNAi technology to suppress genes that may impede CAR functionality.
LEARN MORE NOVEL TECHNOLOGY NEW SOLUTION
NEW SOLUTION
Canine CAR-T Therapy Development: From early target discovery, CAR design and construction, cell culture, and transfection, to in vitro and in vivo function validation.
LEARN MORE SOLUTION