In vitro transcribed mRNA CAR-T (IVT mRNA CAR T) cells act as a safe tumor therapy capable of addressing off-target tumor toxicity, in which T cells are briefly reprogrammed with mRNA encoding chimeric membrane antigen receptor proteins to combat TSA or TAA. In addition, IVT-mRNA CAR-T reduces side effects associated with off-target tumor toxicity due to mRNA instability. Currently, with the development of in situ cell engineering technology, in vivo IVT mRNA-based CAR-T engineering has been gradually developed for cancer immunotherapy.
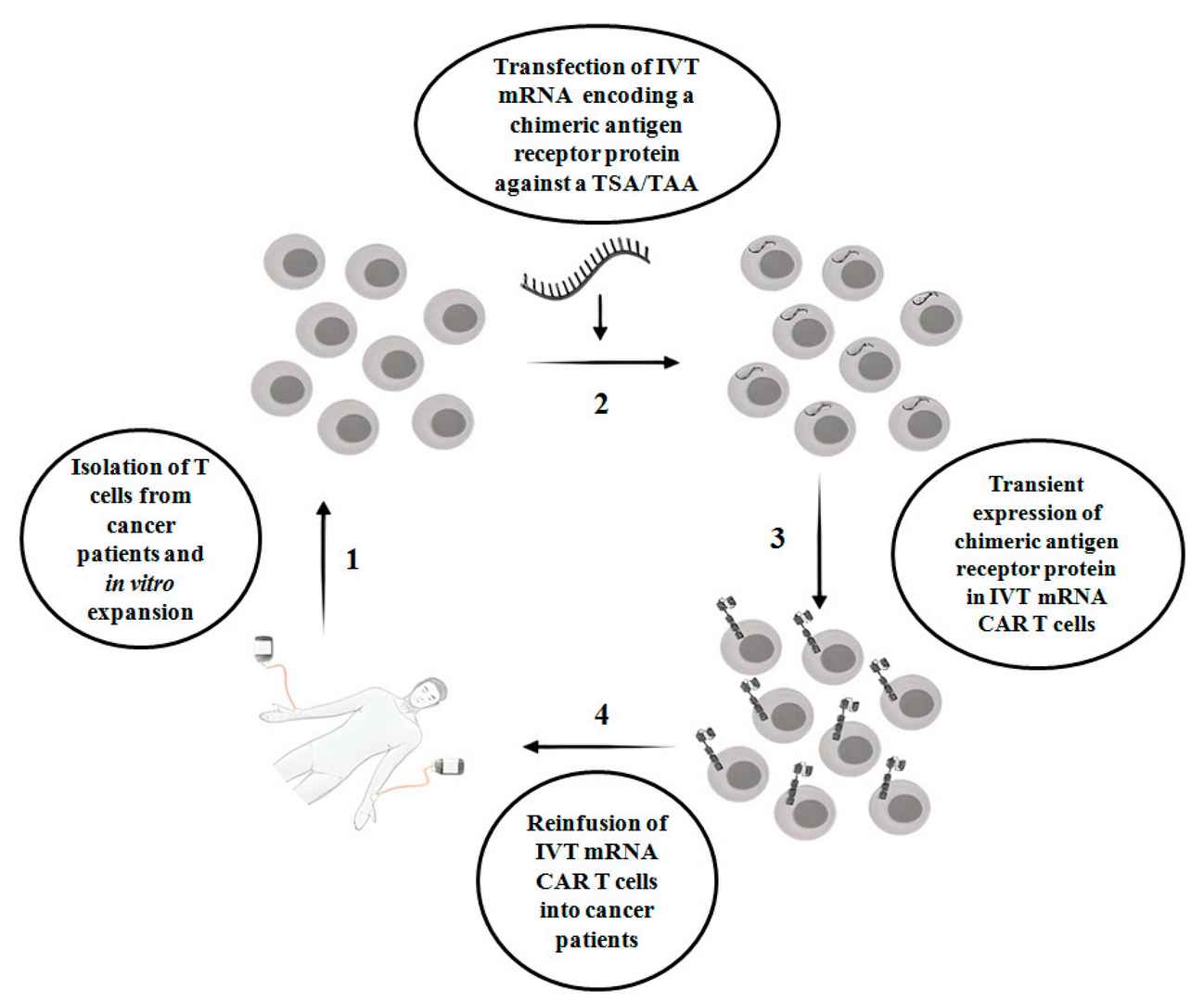 Fig.1 IVT mRNA CAR-T therapy strategy. (Soundara Rajan, 2020)
Fig.1 IVT mRNA CAR-T therapy strategy. (Soundara Rajan, 2020)
Creative Biolabs provides an end-to-end solution for generating potent CAR-T cells directly within the body using optimized mRNA and next-generation lipid nanoparticle systems. This service enables clients to bypass complex ex vivo manufacturing, reduce timelines, and achieve controlled, transient CAR expression ideal for early-stage evaluation, mechanistic studies, and preclinical programs.
Our platforms support mRNA construct design, LNP optimization, immune activation assessment, and preclinical evaluation, enabling rapid, programmable CAR expression directly within the body. With advanced in vivo modeling, delivery-route optimization, and robust analytical capabilities, we provide an integrated framework that streamlines concept-to-preclinical workflows. Our team ensures flexibility, scalability, and scientific rigor, empowering clients to accelerate the development of next-generation in vivo CAR-T therapeutics.
Discover How We Can Help - Request a Consultation
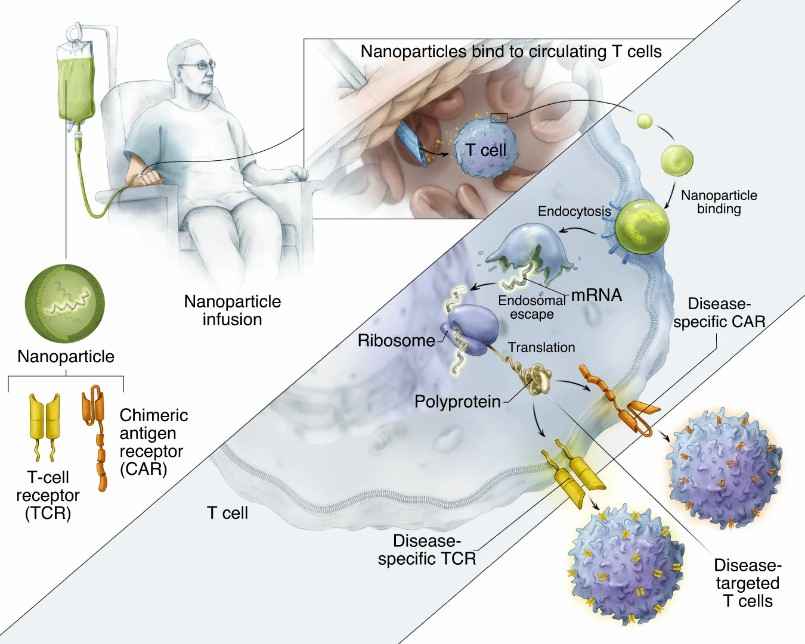 Fig.2 IVT mRNA transported by polymeric nanoparticles programming T cells in situ. (Parayath,2020)
Fig.2 IVT mRNA transported by polymeric nanoparticles programming T cells in situ. (Parayath,2020)
| mRNA Construct Engineering Platform | LNP Formulation & Screening Platform |
|---|---|
|
|
| In Vivo CAR Expression & Functional Validation Platform | |
|
|
The required starting materials include target antigen details and CAR construct outline, the intended administration route and species model for in vivo testing, and specific project objectives regarding activation profile and persistence expectations.
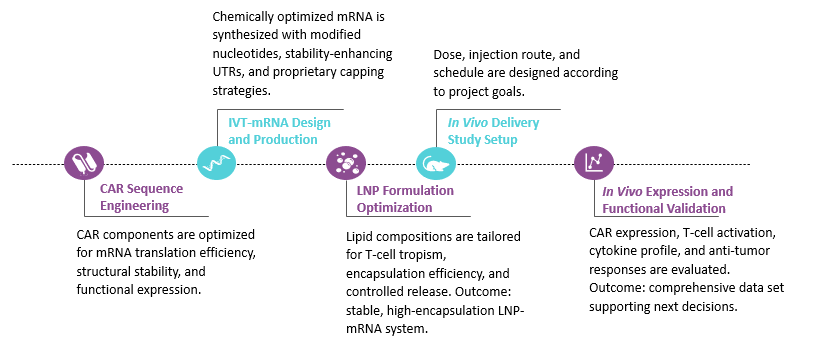 Workflow of In Vivo IVT mRNA-based CAR-T Cell Engineering. (Creative Biolabs Original)
Workflow of In Vivo IVT mRNA-based CAR-T Cell Engineering. (Creative Biolabs Original)
IVT mRNA CAR-T Cells Targeting Solid Malignancies
Summary: The study evaluated the safety and efficacy of adoptive cell therapy with IVT mRNA mesothelin-specific CAR-T cells in 6 patients with chemotherapy-refractory metastatic PDAC. After infusion, transient CAR expression was identified in patients' blood, leading to the proliferation of new immunoglobulin G proteins. The findings support the anticancer effect of mesothelin-specific IVT mRNA CAR-T Cells.
Summary: The study through the purification of mRNA encoding CAR to generate robust CAR-T cells. And in vivo investigations employing a leukemia mouse model found that T cells electroporated with pure mRNAs, regardless of nucleoside alteration, achieved the most effective 100-fold decrease of leukemic load.
If you want to learn more details about our in vivo IVT mRNA-based CAR-T engineering service, please don’t hesitate to contact us.
How stable is the CAR expression achieved through LNP-mRNA delivery?
CAR expression is typically transient yet robust, lasting long enough for functional evaluation while minimizing long-term persistence concerns. Extended expression strategies can be incorporated upon request.
Can the LNP formulation be customized for my target disease model?
Yes. Lipid composition, particle size, release behavior, and targeting strategy can all be tailored. Contact us for model-specific recommendations.
What species are compatible with this in vivo delivery approach?
The platform supports rodent and non-rodent models. Species-specific mRNA and LNP adjustments are implemented to optimize expression.
Is this technology suitable for complex CAR designs?
Multi-domain CARs, logic-gated CARs, and switchable CARs can all be encoded in IVT-mRNA. Our engineering team will evaluate feasibility and recommend design adjustments.
Creative Biolabs provides a comprehensive suite of technology platforms enabling rapid, flexible, and scalable development of next-generation in vivo CAR-T therapeutics using LNP-formulated mRNA. Our services support every stage, from construct design to in vivo functional assessment, ensuring an efficient pathway toward translational success. Contact Our Team for More Information and to Discuss Your Project.
References
For any technical issues or product/service related questions, please leave your information below. Our team will contact you soon.
All products and services are For Research Use Only and CANNOT be used in the treatment or diagnosis of disease.
 NEWSLETTER
NEWSLETTER
The latest newsletter to introduce the latest breaking information, our site updates, field and other scientific news, important events, and insights from industry leaders
LEARN MORE NEWSLETTER NEW SOLUTION
NEW SOLUTION
CellRapeutics™ In Vivo Cell Engineering: One-stop in vivo T/B/NK cell and macrophage engineering services covering vectors construction to function verification.
LEARN MORE SOLUTION NOVEL TECHNOLOGY
NOVEL TECHNOLOGY
Silence™ CAR-T Cell: A novel platform to enhance CAR-T cell immunotherapy by combining RNAi technology to suppress genes that may impede CAR functionality.
LEARN MORE NOVEL TECHNOLOGY NEW SOLUTION
NEW SOLUTION
Canine CAR-T Therapy Development: From early target discovery, CAR design and construction, cell culture, and transfection, to in vitro and in vivo function validation.
LEARN MORE SOLUTION