Target Background
Melanoma antigen tyrosinase (TYR) is a single membrane-spanning transmembrane glycoprotein with 13% carbohydrate content and possesses tyrosine hydroxylase, and dopa oxidase catalytic activities. Physiologically, TYR functions for the hydroxylation of a monophenol and conversion of an o-diphenol to the corresponding o-quinone, which are the rate-limiting steps of melanin and other pigments production from tyrosine via oxidation. The normal biological process sorts mature TYR into melanosomes in the skin melanocytes with catalytical domain inwards. As one of the nonmutated melanocyte differentiation antigens (MDA), TYR is expressed in nearly all melanoma with a high degree of cellular homogeneity, which manifests uncontrolled tyrosinase activity and leads to increased melanin synthesis. In addition, accumulative clinical observations prove that some degree of autoreactivity against TYR can be induced, which contributes to the prolonged survival of patients. Other reports also indicate that the tyrosinase antigen can be presented by HLA-A2. Therefore, TYR constitutes a useful target for specific immunotherapy of melanoma despite causing the possible adverse effects of the destruction of normal melanocytes. Since the safety of tissue-selectivity and extraordinary role of TYR in melanoma, TYR presents unparalleled potentials to be targeted in pre-clinical and clinical pilot studies of CAR-T cell therapy.
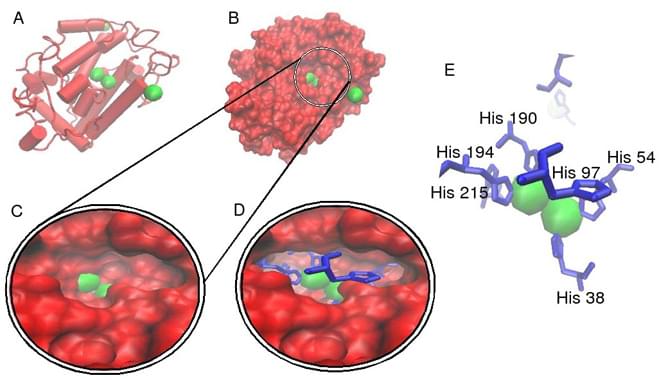 Fig.1 Crystallographic structure of a Streptomyces-derived tyrosinase in complex with a caddie protein.1
Fig.1 Crystallographic structure of a Streptomyces-derived tyrosinase in complex with a caddie protein.1
Anti-TYR CAR-T Cell Therapy
Multiple phase 1/2 clinical trials have been completed to determine the efficacy of conventional melanoma vaccines with different adjuvants, dendritic cell vaccines, DNA vaccines and monoclonal antibody treatment. Recently, a phase 1 and 2 clinical studies is accomplished to evaluate the safety of cellular adoptive immunotherapy using genetically modified and unmodified autologous CD8+ TYR-specific infiltrating T cells for patients with metastatic melanoma. The anti-TYR CAR-T cell therapy will be the next historic strategy for researchers to conquer the metastatic melanoma.
Animal Models for in vivo Study of anti-TYR CAR-T Cell Therapy
Creative Biolabs possesses almost the whole categories of mouse models for melanoma studies, including cell line xenografts (originated from cell lines: B16, 1205 Lu, 451 Lu, etc.), patient-derived tumor xenografts, syngeneic models and genetically engineered mouse models. Particularly, Creative Biolabs can use the newest strategy to generate xenografts with primary melanoma cells tittered down to single cell in NOD/SCID IL-2 receptor gamma chain knockout (NSG) mice. Beyond above, Creative Biolabs also provides UVR-induced models and chemically induced mouse models. Since each model only reveals limited insights into melanoma behavior, Creative Biolabs provides researchers with the best model combined with perfect experiment design to improve scientific understanding of coming results.
In vivo Assay Parameters and Techniques
At Creative Biolabs, we offer the most exquisite and comprehensive service platform for preclinical anti-TYR CAT-T cell therapy research.
Efficacy Test
Tumor remission monitored by tumor volume recording or bioluminescence imaging and survival curve tracking
Viability and Bio-distribution Studies
Durability, bio-distribution studies
Toxicity Evaluation
Pilot tolerability (MTD, The route of administration, Dose regimen/response/onset)
Clinical observation (body weight, feed consumption, ophthalmologic and clinical pathology)
Cytokine storm surveillance (fever, hypertension, prolonged cytopenia)
Complete necropsy, organ weight
Histopathology
Tumorigenicity study
Despite the considerable progress in the understanding of the biology of melanoma, the failure of conventional treatments and rapid resistance limit the therapy efficacy greatly. Creative Biolabs helps you optimize the animal models and assists researchers in the whole studies from the bench to the clinic.
Reference
For any technical issues or product/service related questions, please leave your information below. Our team will contact you soon.
All products and services are For Research Use Only and CANNOT be used in the treatment or diagnosis of disease.
 NEWSLETTER
NEWSLETTER
The latest newsletter to introduce the latest breaking information, our site updates, field and other scientific news, important events, and insights from industry leaders
LEARN MORE NEWSLETTER NEW SOLUTION
NEW SOLUTION
CellRapeutics™ In Vivo Cell Engineering: One-stop in vivo T/B/NK cell and macrophage engineering services covering vectors construction to function verification.
LEARN MORE SOLUTION NOVEL TECHNOLOGY
NOVEL TECHNOLOGY
Silence™ CAR-T Cell: A novel platform to enhance CAR-T cell immunotherapy by combining RNAi technology to suppress genes that may impede CAR functionality.
LEARN MORE NOVEL TECHNOLOGY NEW SOLUTION
NEW SOLUTION
Canine CAR-T Therapy Development: From early target discovery, CAR design and construction, cell culture, and transfection, to in vitro and in vivo function validation.
LEARN MORE SOLUTION