CAR-γδ T cells show anti-cancer properties, as well as acquire additional antigen-specific cytotoxicity and proliferative response, without being affected by prolonged culture or transduction with CAR gene. Creative Biolabs provides one-stop in vitro assay services that best suit your requirements which can accelerate your research.
It is well accepted that CAR-T therapy is efficacious in the treatment of hematological malignancies but is limited to success in solid tumors. γδ T cells come into focus as potential vehicles for CAR therapy. Unlike conventional αβ T cells, allogeneic γδ T cells do not have the risk of graft versus host disease (GvHD). The unique features of γδ T cells, including their major histocompatibility complex (MHC)-independent anti-cancer activity, tissue tropism, the antigen cross-presenting ability, and multivalent response against a broad spectrum of the tumors, render them ideal for designing universal 'third-party' cell products, with the potential to overcome the challenges of allogeneic cell therapy.
Given the natural tropism of γδ T cells for the tumor microenvironment, their transduction with CARs might enhance cytotoxicity while retaining their ability to migrate to the tumor and act as antigen-presenting cells to prolong the intratumoral immune response. Based on the functions of γδ T cells, we provide professionally perform multiple evaluations on the post-transfection CAR-γδ T cells to verify the solid success of CAR-γδ T construction and superior function.
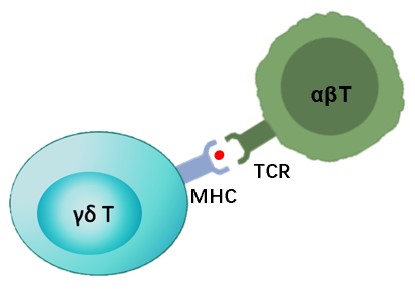
Tumor Antigen Cross-presentation Assay
Upon activation by γδ TCR ligation, γδ T cells become capable of taking up antigens and mediate professional antigen presentation to naïve αβ T cells. Activated γδ T cells might be sufficient to achieve a degree of cross-presentation that is equivalent to that of mature dendritic cells. We provide services you needed to understand and assess the ability of CAR-γδ T cells to take up tumor antigens and cross presented the processed peptide to responder αβ T cells.
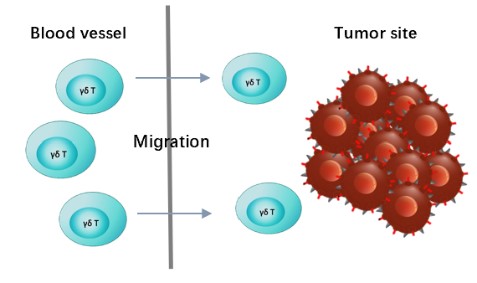
γδ T cells provide a migratory advantage over αβ T cells and a higher ability to infiltrate and function in tumors hypoxic environments. To verify the retaining cell migration function of the CAR-γδT cells, we can provide migration assays with reproducible results in vitro. Popular cell migration assays typically are carried out using Transwell.
We also provide other common in vitro assays to evaluate the CAR functions.
If you are interested in our services, please don't hesitate to contact us.
For any technical issues or product/service related questions, please leave your information below. Our team will contact you soon.
All products and services are For Research Use Only and CANNOT be used in the treatment or diagnosis of disease.
 NEWSLETTER
NEWSLETTER
The latest newsletter to introduce the latest breaking information, our site updates, field and other scientific news, important events, and insights from industry leaders
LEARN MORE NEWSLETTER NEW SOLUTION
NEW SOLUTION
CellRapeutics™ In Vivo Cell Engineering: One-stop in vivo T/B/NK cell and macrophage engineering services covering vectors construction to function verification.
LEARN MORE SOLUTION NOVEL TECHNOLOGY
NOVEL TECHNOLOGY
Silence™ CAR-T Cell: A novel platform to enhance CAR-T cell immunotherapy by combining RNAi technology to suppress genes that may impede CAR functionality.
LEARN MORE NOVEL TECHNOLOGY NEW SOLUTION
NEW SOLUTION
Canine CAR-T Therapy Development: From early target discovery, CAR design and construction, cell culture, and transfection, to in vitro and in vivo function validation.
LEARN MORE SOLUTION