-
Solutions
- Live Attenuated & Killed Vaccine Design
- Subunit Vaccine Design
- Hapten Conjugate Vaccine Design
- Totally Synthetic Vaccine Design
- DNA & RNA Vaccine Design
- Viral Vector Vaccine Design
- Bacterial Vector Vaccine Design
- Virus-Like Particle based Vaccine Design
- Cell based Vaccine Design
- Plant based Vaccine Design
- Bacterial Vaccine Design
- Viral Vaccine Design
- Fungal Vaccine Design
- Parasitic Vaccine Design
- HBV Vaccine Design
- HPV Vaccine Design
- Diabetes Mellitus Vaccine Design
- Autoimmune Disease related Vaccine Design
- Neurodegenerative Disease related Vaccine Design
-
Allergic Disease related Vaccine Design
- Target-AgTM Platform
- Peanut Allergy Vaccine Design
- Plant Pollens Allergy Vaccine Design
- Cow's Milk Allergy Vaccine Design
- Egg Allergy Vaccine Design
- Seafood Allergy Vaccine Design
- Fruit Allergy Vaccine Design
- Bee Venom Allergy Vaccine Design
- Latex Allergy Vaccine Design
- Furry Animals Allergy Vaccine Design
- Tree Nut Allergy Vaccine Design
- House Dust Mite Induced Allergic Asthma Vaccine Design
- Exosome-based Vaccine Design
- Universal Vaccine Design
-
Services
- Vaccine Design
-
Vaccine Formulation Development
- Formulation Optimization
- Delivery System Optimization
- Adjuvant Optimization
- Adjuvant Selection
- PRR Ligand Scanning
- Custom Adjuvant Synthesis
- Mineral Salt Adjuvant related Optimization
- Oil Adjuvant related Optimization
- Saponin related Optimization
- Lipopolysaccharide related Optimization
- CpG related Optimization
- Bacteria-derived Adjuvant Optimization
- Adjuvant Emulsion Optimization
- Innate Immune Stimulator Design
- RNA-based Adjuvant Optimization
- Vaccine Analytical Development & Qualification
-
Vaccine GMP Manufacturing
- Expression System Evaluation
- Bacterial Expression System for Vaccine Production
- Yeast Expression System for Vaccine Production
- Baculovirus-Insect Expression System for Vaccine Production
- Mammalian Cell Expression System for Vaccine Production
- Plant Expression System for Vaccine Production
- Cell-free Expression System for Vaccine Production
- Small-Scale Process Development
- Scale-Up Development
- Vaccine GMP Production
- Serum-Free Suspension Culture
- GMP-grade Antigen Production
- Expression System Evaluation
-
Cancer Vaccines
-
Cancer Vaccine Development
- Cutaneous Cancer related Vaccine Development
- Genitourinary Cancer related Vaccine Development
- Gynecologic Cancer related Vaccine Development
- Gastrointestinal Cancer related Vaccine Development
- Breast Cancer related Vaccine Development
- Lung Cancer related Vaccine Development
- Head and Neck Cancer related Vaccine Development
- Blood Cancer related Vaccine Development
- Brain Tumor related Vaccine Development
- Sarcomas related Vaccine Development
- Pediatric Cancer related Vaccine Development
- Neuroendocrine Tumor related Vaccine Development
- Thyroid Cancer related Vaccine Development
-
Cancer Vaccine Type
- Neoantigen Cancer Vaccine Development
- Tumor Cell Vaccine Development
- Dendritic Cell Vaccine Development
- Antigen Vaccine Development
- Anti-Idiotype Vaccine Development
- DNA based Cancer Vaccine Development
- mRNA based Cancer Vaccine Development
- Vector based Vaccine Development
- Fully Synthetic Carbohydrate based Cancer Vaccine Development
- BCG Vaccine Development
- Ty21 based Vaccine Development
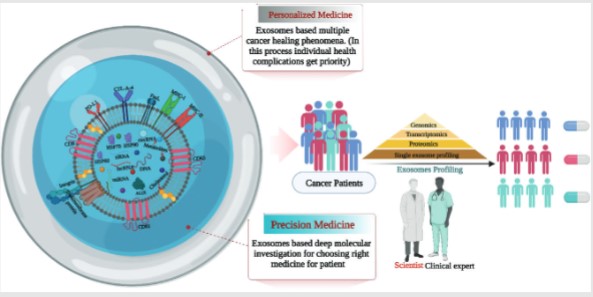
- Exosome based Therapeutic Cancer Vaccine Development
- Ag-Fc Fusion Protein based Vaccine Development
- Biomaterial-based Implantable Cancer Vaccine Development
- Cytomegalovirus based Cancer Immunotherapy Development
- Immunological Checkpoint Therapy Development
- Immunoplacental Therapy Development
- Neoantigen Cancer Vaccine Development Solution
- Personalized Neoepitope mRNA Cancer Vaccine Technology Platform
- Modular NeoVax Technology Platform
- AI-aided Analysis Technology Platform
- High-efficient Neoantigen In Vitro Evaluation Technology Platform
- NeoTCR-T Technology Platform
- SLP based Neoantigen Cancer Vaccine Development Solution
- DC based Neoantigen Cancer Vaccine Development Solution
- Biomaterial-assisted Neoantigen Cancer Vaccine Development Solution
- Adoptive Neoantigen activated T Cell Transfer Therapy Solution
- Tumor Cell Vaccine Development Solution
- Hematopoietic Stem Cell Transplantation Technology Platform
- COVB Technology Platform
- Allogeneic Cell Vaccine Design & Modification Solution
- Costimulatory Molecule Development Solution
- IL-2-Secreting Tumor Cell Vaccine Development Solution
- GM-CSF-Secreting Tumor Cell Vaccine Development Solution
- Cancer Stem Cell targeted Vaccine Development Solution
- Dendritic Cell Vaccine Development Solution
- Antigen Vaccine Development Solution
- Anti-Idiotype Vaccine Development Solution
- DNA based Cancer Vaccine Development Solution
- mRNA based Cancer Vaccine Development Solution
- Vector based Vaccine Development Solution
- Fully Synthetic Carbohydrate based Cancer Vaccine Development Solution
- BCG Vaccine Development Solution
- Ty21 based Vaccine Development Solution
- Ag-Fc Fusion Protein based Vaccine Development Solution
- Biomaterial-based Implantable Cancer Vaccine Development Solution
- Cytomegalovirus based Cancer Immunotherapy Development Solution
- Immunological Checkpoint Therapy Development Solution
- Immunoplacental Therapy Development Solution
- Immune Monitoring Assay Platform for Cancer Vaccine
- Cytokine Development Platform for Cancer Vaccine
- Exosome based Therapeutic Cancer Vaccine Development Solution
-
Cancer Vaccine Technology
- Tumor-APC Fusion Technology
- Immune Monitoring Assay
- Cytokine Development
- Anti-Treg Technology
- Vaccine Combination Immunotherapy
- Biomarker Discovery Technology
- Microfluidic Cell Squeezing Technology
- Antibody-Antigen Fusion Technology
- MHC Class II associated li Protein Suppression Technology
- Innovative Tumor-APC Fusion Platform for Cancer Vaccine
- Anti-Treg Technology Platform for Cancer Vaccine
- Vaccine Combination Immunotherapy Platform for Cancer Vaccine
- Biomarker Discovery Platform for Cancer Vaccine
- Microfluidic Cell Squeezing Platform for B Cell based Vaccine
- Antibody-Antigen Fusion Platform for Cancer Vaccine
-
Cancer Vaccine Development
- Products
-
Supports
- FAQs
- Literature Center
- Download Resources
- Podcast
-
Scientific Frontier
- Murine vs. Human Norovirus Models in Vaccine Research
- Norovirus Immune Evasion and the Challenge of Durable Vaccine Protection
- Norovirus Vaccine Development – Where Are We Now in 2025
- Virus-Like Particle Vaccines for Norovirus – A Promising Immunological Strategy
- Why Rotavirus Vaccines Don't Protect Against Norovirus
- Understanding the Molecular Mechanisms of Three RSV Vaccine Construction Strategies
- Respiratory Syncytial Virus F Protein in Next-Generation Vaccine Design
- RSV Infection Life Cycle and Vaccine Target Discovery
- The Translational Value of BRSV Models in Human RSV Vaccine
- Deciphering the Molecular Mechanisms of Respiratory Syncytial Virus Infection
- Rabies Virus Vaccine:Revealing Potential Vaccine Targets
- Biological Research on mRNA Rabies Vaccines
- Designing mRNA Ebola Vaccines: Delivery Systems and Immunogenicity Analysis
- Ebola Virus Glycoprotein: Structure, Entry Mechanism, and Therapeutic Implications
- Ebola Virus Replication Cycle: Host Factors, RdRp Kinetics, and Vaccine Targets
- The Molecular Architecture of HPV: Decoding the L1 Capsid Protein
-
Featured Project Solutions
- Fungal Vaccine Design
- Viral Vaccine Development Solution
- Parasitic Vaccine Solution
- HBV Vaccine Solution
- HPV Vaccine Solution
- Diabetes Mellitus Vaccine Solution
- Autoimmune Disease related Vaccine Solution
- Neurodegenerative Disease related Vaccine Solution
- Allergic Disease related Vaccine Solution
- Exosome-based Vaccine Solution
- Universal Vaccine Solution
- Monkeypox Virus (MPXV) Vaccine Solution
- Vaccine Design Solution
- Vaccine Formulation Development Solution
- Vaccine Analytical Development & Qualification Solution
- Vaccine GMP Manufacturing Solution
- Cancer Vaccine Development Solution
- Featured Cancer Vaccine Type
- Featured Cancer Vaccine Technology
- Bacterial Vaccines Solution
- Subunit Vaccine Solution
- Live Attenuated and Killed Vaccine Solution
- Bacterial Vector Vaccine Solution
- Cell based Vaccine Solution
- DNA and RNA Vaccine Solution
- Hapten Conjugate Vaccine Solution
- Plant based Vaccine Solution
- Totally Synthetic Vaccine Solution
- Viral Vector Vaccine Solutions
- Virus-Like Particle based Vaccine Solution
- Company