Pseudotyping Service of Lentiviral Vectors with Gibbon Ape Leukemia Virus
Pseudotyping lentivirus vectors with different viral glycoprotein is a useful strategy to change the host range of the vectors. Nowadays, the gammaretrovirus has been widely used for gene therapy because of its wide host cell tropism and nonpathogenicity. Lentiviral vectors pseudotyped with the envelope (Env) glycoprotein derived from gibbon ape leukemia virus (GaLV) have been successfully used in recent preclinical gene therapy studies. Creative Biolabs provides comprehensive glycoprotein-pseudotyped lentivirus vectors for basic research and preclinical applications. Here, with the help of our advanced technique and experienced staff, Env derived from GaLV is fully utilized to provide customized lentivirus vectors design and construction services for our global clients.
Gibbon Ape Leukemia Virus
The origin of GaLV has been assumed to be a murine retrovirus derived from a local South East Asian rodent host. The host cell receptor for GaLV Env has been identified as a sodium-dependent phosphate transporter protein. Similar to other retroviruses, GaLV encodes a single transmembrane surface glycoprotein (GaLV Env), which is cleaved into surface (SU) and transmembrane (TM) subunits. The TM domain of GaLV Env contains a short 30-amino-acid C-terminal cytoplasmic tail. The Env protein from GaLV has been shown to be incompatible with HIV-1 in the production of infectious pseudotyped particles. This incompatibility has been mapped to the C-terminal cytoplasmic tail of GaLV Env and the HIV-1 accessory protein Vpu modulates this incompatibility. Glycoproteins containing the cytoplasmic tail from GaLV Env are prevented from being incorporated into HIV-1 particles by Vpu, effectively reducing infectious particle production by 50- to 100-fold.
Pseudotyping of Lentiviral Vectors
Rationale for Pseudotyping
The GaLV Env allows the lentiviral core to bypass traditional entry barriers. In many primary human cells, the amphotropic or ecotropic receptors are either lowly expressed or quiescent. GaLV provides a high-affinity alternative that is particularly effective in dividing cells, making it a cornerstone for ex vivo expansion protocols.
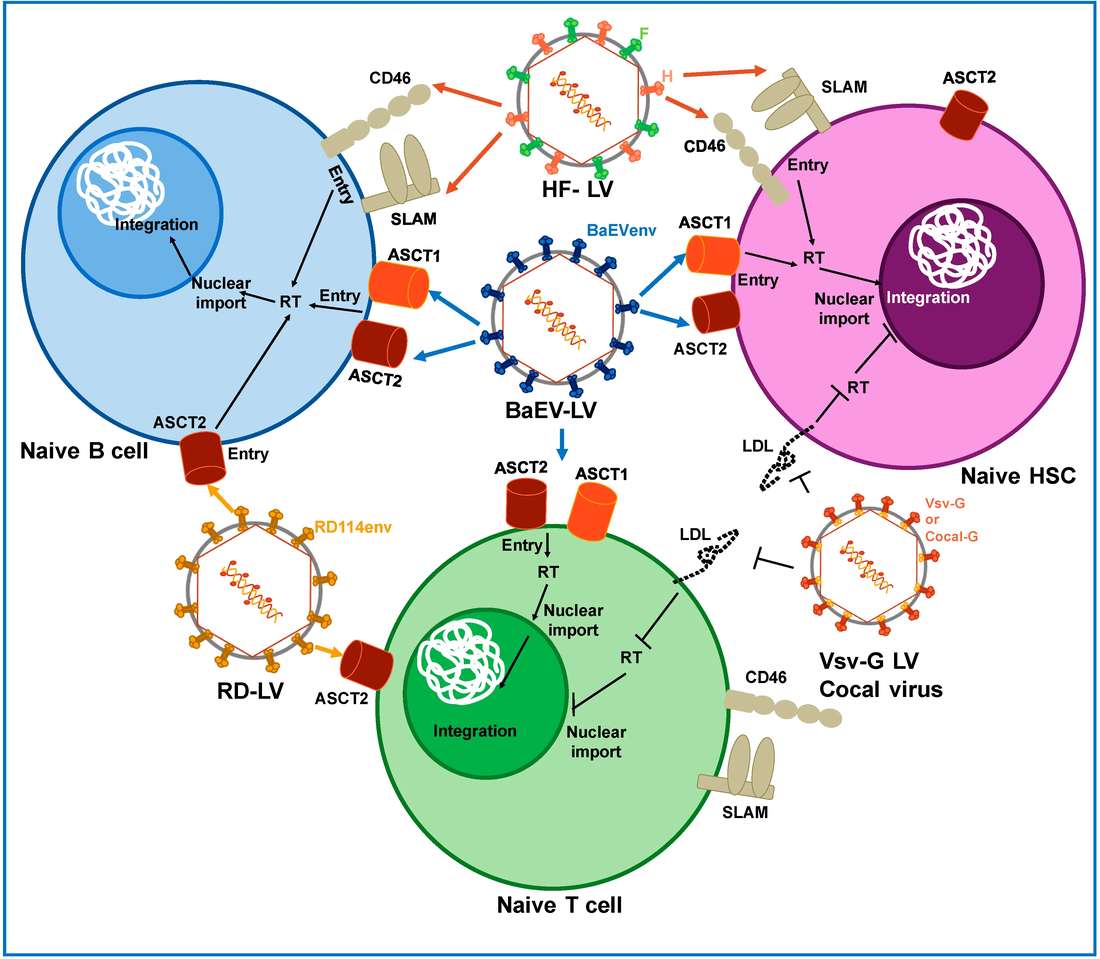 Figure 1. Pseudotyping of lentiviral vectors.1
Figure 1. Pseudotyping of lentiviral vectors.1
Pseudotyping Strategy with GaLV Env
Retrovirus vectors are the most commonly used gene transfer vectors in current human gene therapy applications. The host ranges of such vectors are influenced in large part by the properties of the protein contained in the outer lipid Env of the vector particle. Pseudotyping allows the vectors to transduce ranges of cells and tissues different from those that would be possible with just the native Env. Three strategies have been proposed by Creative Biolabs to allow GaLV Env to pseudotype HIV particles,
- Removal of the C-terminal half of the cytoplasmic domain (R peptide)
- Replacement of the whole cytoplasmic tail with the corresponding MLV region
- Mutation of two residues K618 and I619 upstream of the R peptide cleavage site. Our perfect strategy and advanced hardware facilities will guarantee you a perfect GaLV-glycoprotein pseudotyped lentivirus vector.
Selection Guide of Comparison with Other Pseudotyping Options
| Envelope | Primary Receptor | Key Advantages | Ideal For | Considerations |
|---|---|---|---|---|
| GaLV | PiT-1 (SLC20A1) | High HSC/T-cell efficiency, low cytotoxicity, serum-resistant. | HSPCs, T/B/NK cells, ex vivo therapy, repeated transduction. | Broader tropism than ecotropic, but more selective than VSV-G. |
| VSV-G | LDL-R | Ultra-broad tropism, yields very high-titer stocks. | In vitro studies on easy-to-transduce lines, in vivo delivery (CNS). | Cytotoxic, inactivated by human serum, triggers immune responses. |
| RD114 | RDR | Excellent for canine and human HSCs, low cytotoxicity. | Canine models, clinical HSC gene therapy. | Slightly more restricted tropism than GaLV in some human cell types. |
Advantages of GaLV-Pseudotyped Lentiviral Vectors
- Enhanced Transduction Efficiency in Specific Cell Types: GaLV pseudotyping significantly boosts gene delivery into human CD34⁺ hematopoietic stem cells (HSCs), T cells, and B cells compared to standard VSV-G vectors.
- Reduced Cytotoxicity: Unlike VSV-G, which can be highly fusogenic and cytotoxic at high multiplicities of infection (MOI) or upon concentration, GaLV Env exhibits lower cell membrane disruption, preserving cell viability and function—a critical factor for therapeutic cell products.
- Improved Performance in Hematopoietic Stem Cells (HSCs): Multiple independent studies confirm that GaLV-pseudotyped lentiviral vectors achieve superior transduction rates in quiescent and activated HSCs, a cornerstone target for genetic blood disorder therapies.
- Better Suitability for Repeated Transduction: The lower immunogenicity and cytotoxicity allow for sequential rounds of transduction, enabling strategies like multi-gene delivery or iterative engineering.
- Stable Gene Expression: Like all lentiviral vectors, GaLV-pseudotyped versions provide stable, long-term transgene expression in both dividing and non-dividing cells due to genomic integration.
Services at Creative Biolabs
At Creative Biolabs, we recognise that breakthroughs in gene and cell therapy research are built upon highly tailored, reliable, and efficient delivery tools. Our GaLV Pseudotyped Lentiviral Vector Service extends far beyond standard production, offering an integrated platform encompassing vector design, molecular cloning, production & purification, and comprehensive QC. We partner with you to transform your conceptual vision into a validated, high-performance vector, accelerating your path from scientific discovery to clinical translation.
Vector Design
Vector design is the critical first step determining experimental success. Our expert team provides comprehensive design strategies based on your research objectives (e.g., gene overexpression, knockout, cell labeling, or therapeutic engineering), target cell types (primary HSCs, T cells, etc.), and application scenarios (in vitro research, preclinical animal studies).
Table 1: Core Vector Design Elements and Selection Strategy
| Design Module | Options | Key Features & Advantages | Typical Application |
|---|---|---|---|
| Promoter | Strong Constitutive: EF1α, CMV, PGK, CAG | Drives robust, sustained high-level expression; PGK shows lower epigenetic silencing tendency. | General studies requiring stable, high-level reporter or therapeutic gene expression. |
| Cell-Specific: CD4, CD8a, CD19, CD34, Synapsin | Restricts expression to specific cell lineages, enhancing targeting and therapeutic safety. | CAR-T therapy (confining CAR to T cells), stem cell lineage tracing, neuroscience research. | |
| Inducible: Tet-On/Off | Enables precise spatiotemporal and dose-dependent chemical control of transgene expression. | Functional gene studies, gene therapies where controlled expression avoids toxicity. | |
| Pseudotyping Envelope | Wild-type GaLV | High transduction efficiency for hematopoietic/lymphoid cells, low cytotoxicity—the gold standard. | Research and engineering of most HSPCs, T, B, and NK cells. |
| Engineered Variants (e.g., GaLV-MTR) | Enhanced fusogenicity for superior efficiency in refractory cells. | Working with precious or hard-to-transduce primary cell samples. | |
| Chimeric Envelopes | Combines advantageous domains from different envelopes to optimize properties. | Custom needs for specialized applications, like improved in vivo delivery stability. | |
| Transgene Cassette | Reporter Genes: GFP, Luciferase | Facilitates cell tracking, sorting, and in vivo imaging analysis. | Transduction efficiency assessment, cell fate mapping, in vivo imaging in animal models. |
| Surface Markers: tNGFR, CD34, truncated EGFR | Allows antibody-based positive selection or in vivo clearance, enhancing clinical safety. | Clinical-grade cell product manufacturing, safety switch design. | |
| Multigene Expression Strategy | 2A Peptide Linkers | Co-expression of multiple proteins from a single transcript; relatively balanced expression; compact size. | Co-expression of CAR & reporter, CRISPR-Cas9 & gRNA, multiple synergistic therapeutic factors. |
| IRES or Bidirectional Promoters | Enables co-expression of two genes; IRES-mediated expression is typically weaker; bidirectional promoters allow independent regulation. | When differential expression levels of two genes are required. |
Molecular Cloning & Sequence Verification
Precision in molecular manipulation is foundational to vector function. We employ industry-leading technologies to guarantee the final plasmid sequence perfectly matches the design, free of unintended mutations.
- Gene Synthesis & Optimization: Codon-optimize your provided sequence (for human cell expression) and synthesize the full-length gene fragment.
- Plasmid Construction: Utilize high-efficiency, error-free cloning techniques (e.g., Gibson Assembly, Golden Gate Assembly) to precisely assemble the promoter, gene, and regulatory elements into our optimized third-generation lentiviral backbone.
-
Comprehensive Sequence Verification:
- Restriction Enzyme Mapping: Confirms basic vector architecture.
- Sanger Sequencing: Verifies critical regions.
- Next-Generation Sequencing (NGS): Provides deep sequencing of the complete plasmid, ensuring 100% coverage of the expression cassette. This offers the highest level of sequence accuracy assurance, crucial for IND-enabling studies.
Viral Production & Purification
We utilize scalable transient transfection systems coupled with downstream purification processes to deliver viral preparations suitable for research through preclinical stages.
Table 2: Production Scale and Delivery
| Service Tier | Production Scale | Primary Purification Method | Key QC Parameters | Application Stage |
|---|---|---|---|---|
| Research Grade | Small-scale | Ultracentrifugation or simple concentration | Functional Titer, Sterility | Feasibility testing, protocol optimization |
| Standard Grade | Mid-scale | TFF combined with Ion-Exchange/Size-Exclusion Chromatography | Full QC Panel: Functional/Physical Titer, Infectivity Potency, RCL, Endotoxin, Sterility | In vitro functional studies, small animal experiments |
| Preclinical Grade | Large-scale | TFF & Chromatography | Extended Characterization + Standard QC | IND-enabling studies, GLP toxicology |
GaLV Pseudotyping Technology Platform at Creative Biolabs
Envelope Engineering and Optimisation
Our platform utilises proprietary GaLV Env expression plasmid designs. We employ codon optimisation for enhanced expression in human producer cells and strategic truncation of the cytoplasmic tail to improve pseudotyping efficiency with lentiviral cores. These optimisations maximise viral incorporation efficiency, resulting in higher functional titers.
Lentiviral Vector Production
We employ a robust third-generation lentiviral packaging system, split across four plasmids (vector, gag/pol, rev, and envelope) to ensure safety by minimising recombination risk. Production is typically via transient transfection of HEK-293T cells, optimised for yield and consistency. For clients requiring clinical-grade material, we offer a scalable, production workflow.
Frequently Asked Questions
Q: What cell types are most suitable for GaLV pseudotyped lentiviral vectors?
A: They are exceptionally effective for human CD34⁺ hematopoietic stem and progenitor cells (HSPCs), primary T lymphocytes, B cells, and NK cells, as well as many established hematopoietic cell lines.
Q: How does GaLV compare to VSV-G for HSC transduction?
A: GaLV often shows equal or superior transduction efficiency with significantly less toxicity, better preservation of stem cell multipotency, and engraftment potential in immunodeficient mouse models.
Q: Can GaLV pseudotyped vectors be used for in vivo studies?
A: While primarily optimised for ex vivo use, GaLV pseudotyped vectors can be used for in vivo delivery, particularly in immunodeficient models. However, their tropism is more restricted than VSV-G.
Q: What biosafety level is required?
A: GaLV-pseudotyped third-generation lentiviral vectors are typically handled at BSL-2 containment, as they are replication-incompetent. Always follow your institution's guidelines.
Q: Can this service be combined with tissue-specific promoters?
A: Absolutely. We routinely clone custom promoters to restrict transgene expression to specific lineages (e.g., T-cell-, myeloid-, or erythroid-specific promoters), thereby enhancing safety and specificity.
Why Choose Creative Biolabs' GaLV Lentiviral Pseudotyping Service?
- Extensive Experience: Over a decade of specialized work in lentiviral vector engineering and alternative envelope pseudotyping.
- Technical Expertise: Deep understanding of envelope-receptor biology and vector optimization.
- Reproducibility: Stringent protocols ensure high consistency and batch-to-batch reproducibility.
- Efficiency: Fast turnaround time complemented by dedicated project management and technical support.
We're Here to Help—Contact Us!
The properties of the GaLV Env enable lentivirus vectors pseudotyped with GaLV Env be potentially useful for human gene therapy applications. If you are interested in our GaLV EnV pseudotyping services, please feel free to contact us for more information.
Reference
- Gutierrez-Guerrero A, Cosset F L, Verhoeyen E. Lentiviral vector pseudotypes: precious tools to improve gene modification of hematopoietic cells for research and gene therapy. Viruses, 2020, 12(9): 1016. https://doi.org/10.3390/v12091016 (Distributed under Open Access license CC BY 4.0, without modification.)
