Creative Biolabs is a CRO solution provider created and operated by a team of scientists and trained professionals with considerable expertise and experience in cell therapy research and development. We dedicate ourselves to delivering top-notch products and services to customers globally in the scientific and industrial sectors, promoting the pursuit of breakthrough discoveries in science, and effectively boosting preclinical and clinical research.
Receptor tyrosine kinase-like orphan receptor 1 (ROR1) is a surface antigen that belongs to the orphan receptor tyrosine kinase-like family. It consists of three main sections: an extracellular domain, a transmembrane segment, and a cytoplasmic region. The extracellular section contains an immunoglobulin-like domain (IG), a cysteine-rich domain (CRD), and a Kringle domain (KD). On the other hand, ROR1's intracellular side has two serine/threonine-rich domains (Ser/Thr), a proline-rich domain (PRD), and a tyrosine kinase domain (TKD).
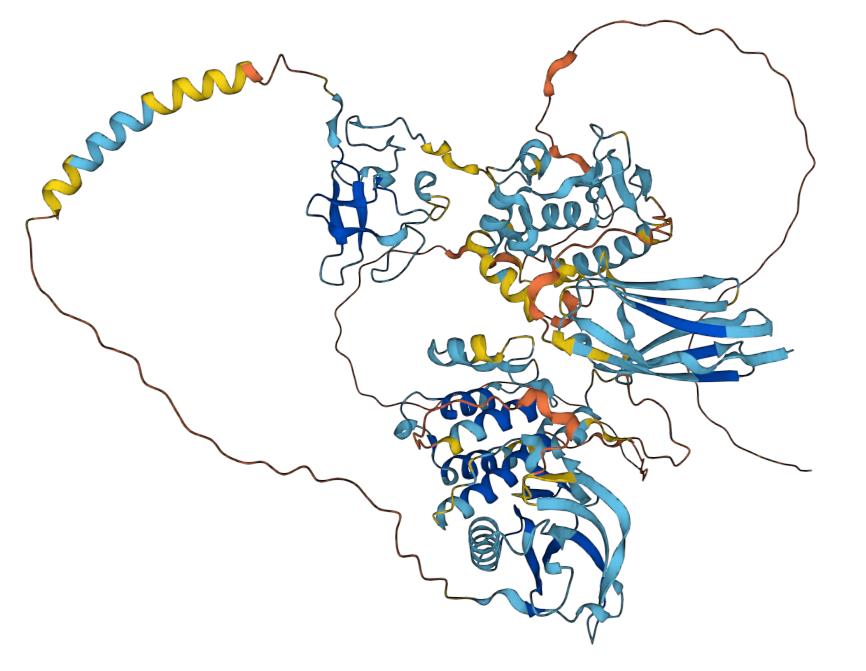 Fig.1 Structure of ROR1
Fig.1 Structure of ROR1
ROR1 is expressed in undifferentiated embryonic stem (ES) cells, but its expression decreases upon differentiation in conditioned medium. ROR1-positive B cell non-Hodgkin's lymphomas (B-NHL), including mantle cell lymphoma (MCL) and diffuse large B cell lymphoma, are identified. Despite previous findings, ROR1 is also expressed on several normal tissues such as parathyroid, pancreatic islets, and regions of the esophagus, stomach, and duodenum. The Human Protein Atlas project confirms that ROR1 is expressed in various tissues like bone marrow, lymphoid tissues, and testis.
The biological function of ROR1 is not fully understood, but it may play a role in embryonic development and the regulation of cell migration and survival. As a result, ROR1 has been identified as a potential therapeutic target for cancer treatment.
Studies have shown that ROR1 activates cell survival signaling events, particularly the non-canonical WNT pathway, which is important in oncogenesis. Specifically, ROR1 binds to Wnt5a to activate this pathway, which has been linked to cancer progression. However, further research is needed to fully comprehend the role of ROR1 in the Wnt pathway and develop effective treatments targeting it.
RORs are highly expressed in tumor cells but not in normal tissues, making them ideal targets for treating minimal residual disease, the final obstacle to curing many cancers, including solid tumors like neuroblastoma.
ROR1 is a surface receptor that can be targeted by monoclonal antibodies and T-cells modified to express a chimeric antigen receptor (CAR) specific for ROR1. This approach overcomes the low receptor density of ROR1 and retargets T-cell-mediated cytotoxicity against tumor cells.
Currently, there are two ongoing CAR cell therapies targeting ROR1 mainly for hematologic malignancies (see Table 1). Preclinical studies have shown promising results in using ROR1-targeted CAR cells to treat B-cell malignancies such as chronic lymphocytic leukemia (CLL) and mantle cell lymphoma (MCL).
Table 1. Ongoing ROR1-Targeted CAR Cell Therapy Clinical Trials
| NCT Number | Title | Status | Conditions | Phases | Sponsor/Collaborators |
| NCT02706392 | Genetically Modified T-Cell Therapy in Treating Patients With Advanced ROR1+ Malignancies | Terminated | Hematopoietic and Lymphoid Cell Neoplasm|Malignant Solid Neoplasm|Metastatic Lung Non-Small Cell Carcinoma|Metastatic Triple-Negative Breast Carcinoma|Recurrent Acute Lymphoblastic Leukemia|Recurrent Mantle Cell Lymphoma|Refractory Chronic Lymphocytic Leukemia|Stage III Lung Non-Small Cell Cancer AJCC v7|Stage IIIA Lung Non-Small Cell Cancer AJCC v7|Stage IIIB Lung Non-Small Cell Cancer AJCC v7|Stage IV Breast Cancer AJCC v6 and v7|Stage IV Lung Non-Small Cell Cancer AJCC v7|Unresectable Lung Non-Small Cell Carcinoma | Phase 1 | Fred Hutchinson Cancer Center|National Cancer Institute (NCI) |
| NCT02194374 | Autologous ROR1R-CAR-T Cells for Chronic Lymphocytic Leukemia (CLL) | Withdrawn | Leukemia | Phase 1 | M.D. Anderson Cancer Center|CLL Global Research Foundation Alliance |
References
For any technical issues or product/service related questions, please leave your information below. Our team will contact you soon.
All products and services are For Research Use Only and CANNOT be used in the treatment or diagnosis of disease.
 NEWSLETTER
NEWSLETTER
The latest newsletter to introduce the latest breaking information, our site updates, field and other scientific news, important events, and insights from industry leaders
LEARN MORE NEWSLETTER NEW SOLUTION
NEW SOLUTION
CellRapeutics™ In Vivo Cell Engineering: One-stop in vivo T/B/NK cell and macrophage engineering services covering vectors construction to function verification.
LEARN MORE SOLUTION NOVEL TECHNOLOGY
NOVEL TECHNOLOGY
Silence™ CAR-T Cell: A novel platform to enhance CAR-T cell immunotherapy by combining RNAi technology to suppress genes that may impede CAR functionality.
LEARN MORE NOVEL TECHNOLOGY NEW SOLUTION
NEW SOLUTION
Canine CAR-T Therapy Development: From early target discovery, CAR design and construction, cell culture, and transfection, to in vitro and in vivo function validation.
LEARN MORE SOLUTION